Results from the global Diabetes Attitudes, Wishes and Needs 2 study (DAWN2TM) show that 28 per cent of Canadians living with diabetes have experienced significant emotional distress and 15 per cent have felt discriminated against due to their disease. Results from the DAWN2TM study were presented today at the 73rd Scientific Sessions of the American Diabetes Association (ADA). DAWN2TM represents opinions from more than 15,000 people living with, or caring for people with diabetes in 17 countries across four continents. The study assessed a wide range of psychosocial indicators of diabetes care, including discrimination and the impact it has on a person’s emotional wellbeing. Key Canadian results from the DAWN2TM study include: More than 12 per cent of Canadians with diabetes had possible depression. Twenty-seven per cent of family members reported a significant burden on the family related to diabetes. Eighty-nine per cent of Canadian respondents living with diabetes had attended a diabetes education program or activity. More than 35 per cent of family members had ever attended a diabetes education program or activity. DAWN2TM will help stimulate much-needed discussion between healthcare providers, patients and their families, and lead to significant improvements in the management of diabetes.
Rotary and Gates Foundation extend fundraising agreement to end polio
Rotary International and the Bill & Melinda Gates Foundation announced an extension of their existing fundraising partnership that could generate up to US$525 million in new money for polio eradication as the global effort to end this crippling disease enters its critical endgame phase. Under the new agreement, announced before an audience of more than 20,000 Rotary members from 160 countries gathered in Lisbon for the humanitarian group’s annual convention, the Gates Foundation will match 2 for 1 every new dollar Rotary commits to polio eradication up to $35 million per year through 2018. All funds raised will support crucial immunization activities in polio-affected countries. These are part of a comprehensive six-year plan to eradicate both wild poliovirus and vaccine-derived virus announced by the eradication initiative during the Global Vaccine Summit in Abu Dhabi. At the Summit, global leaders and individual philanthropists signaled their confidence in the endgame plan by pledging $4 billion, nearly three-quarters of the plan’s projected $5.5 billion cost. They also called upon additional donors to commit the additional $1.5 billion needed to ensure eradication. Since then, the government of Australia, and now Rotary, are committing funding toward the remaining $1.5 billion gap through 2018.
Mushrooms in Cancer Care
Introduction
Many patients with cancer turn to complementary and alternative medicine as an adjunct to their care. Historically, mushrooms have been used across various cultures for their health-promoting effects and for their effects on the immune system (Jin 2012). Much research has been conducted on the effects of mushrooms for various cancer types, including both laboratory research and clinical trials. The most common mushrooms that have been studied are Agaricus blazei, Coriolus versicolor, Grifolia frondosa, and Ganoderma lucidum. Agaricus has immunological effects on the complement and innate immune systems which could potentially lead to pro-apoptotic and anti-angiogenic effects (Lima 2011). Coriolus contains two polysaccharides (PSP and PSK) that appear to improve overall survival rates in patients with multiple cancers, including gastric, colon/ rectal, breast carcinomas (Wong 2012). Grifolia appears to stimulate the function of various immune cells and may improve overall quality of life, reduce tumor burden and act synergistically with chemotherapy, especially in patients with hepatocellular, breast and lung cancer (Kodama 2002). Ganoderma has the potential of reducing tumor burden and stimulating immunity in various cancer types (Jin 2012). This article will individually review the evidence available for these four mushrooms and clearly summarize the most pertinent research findings in the hopes that they can be effectively utilized in a clinical setting. Suggested dosing protocols and safety concerns are also explored for this purpose.
Agaricus blazei
Agaricus blazei is a medicinal mushroom of Brazilian origin. It has been used traditionally to treat a variety of conditions, including diabetes, hypercholesterolemia, chronic hepatitis and cancer (Hetland 2008). Agaricus contains several biologically active constituents, including beta-glucans, which are known activators of macrophages, polymorphonuclear cells and NK cells. The beta-glucans component of Agaricus blazei have been found to be the main source of antitumor activity (Hetland 2008).
More recent in-vitro and in-vivo evidence has demonstrated the immunological effects Agaricus blazei can have, via the complement and innate immune systems. A review paper of fourteen studies was recently published to assess Agaricus blazei’s action on the immune system (Lima 2011). In ten of the studies a stimulatory effect was observed, whereas in one study an inhibitory effect was described. In the remaining three studies, both effects were observed. The inhibitory effects included down-regulation of cytokines IL-2, IL-4, IFN-gamma and TNF-alpha. The stimulatory effects observed included stimulation of macrophages via CD receptors and of NK cells. Furthermore, in-vivo studies reviewed demonstrated a stimulatory effect of antibody synthesis and activation of the complement system. Thus Agaricus blazei’s mechanism of action includes acting as an anti-inflammatory agent, while stimulating both innate and adaptive immunity (Lima 2011).
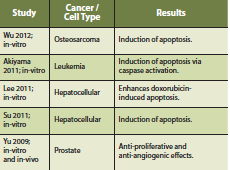
Furthermore, a phase I clinical study of Agaricus blazei was conducted on seventy-eight patients with a history of cancer who were in remission (Ohno 2011). Adverse events (AE’s) were observed in 12% of patients studied, with reports of nausea being the most common. Importantly, the authors make note that none of the AE’s occurred in a dose-dependent manner and that Agaricus blazei is considered safe in this patient population. Most of the anti-cancer research for Agaricus blazei consists of in-vitro and in-vivo studies. A pro-apoptotic effect has been observed in-vitro and whether this effect is maintained in human studies remains to be seen. Anti-angiogenic effects have also been studied in a rat model (Kimura 2004). Further human trials for Agaricus blazei are needed to fully understand its utility and safety.
Dosage: 1,500-3,000mg of hot-water extract daily, containing a minimum of 40% polysaccharides.
Safety: Caution with patients diagnosed with an autoimmune condition, and/or using immunosuppressive medications.
Coriolus versicolor (Trametes versicolor)
The mushroom Coriolus versicolor (Trametes versicolor) is a macrofungi belonging to the Basidiomycetes class, which consists of at least 22,000 known species (Chu 2002). Traditionally, it has been used to increase energy and treat pulmonary and upper respiratory tract infections, hepatitis and cancer. The fruiting body and mycelium provides its most effective components (Wong 2012).
Coriolus versicolor is one of the better-studied mushrooms …
Coriolus contains several different polysaccharides, however, most research has focused on polysaccharide peptide (PSP) and polysaccharide krestin (PSK) (Ng 1998). PSP and PSK are two chemically similar structures isolated from Coriolus versicolor from the mycelia of two different strains. Both extracts consist of similar compounds, with PSK predominantly containing fucose while PSP contains arabinose and rhamnose (Ng 1998). Coriolus versicolor is one of the better-studied mushrooms, with several randomized control trials. Most of the research is focused on quality of life outcomes and its use adjunctively with conventional cancer treatments. PSP and PSK are both used as interventions, however, PSK is more commonly used in randomized control trials.
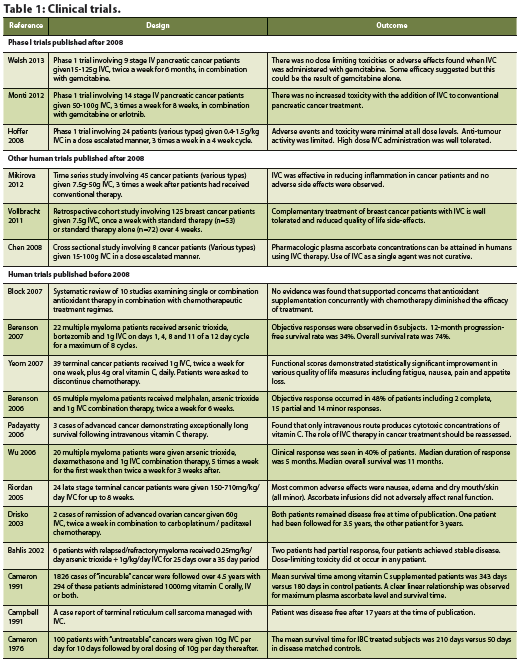
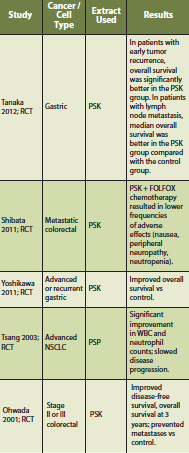 A systematic review and meta-analysis was conducted to assess the efficacy of Coriolus versicolor on overall survival in patients with cancer (Wong 2012). Thirteen randomized trials were included. Types of cancer (encompassing stage I to IV) in these studies included esophageal, gastric, colon/rectal, breast and nasopharyngeal carcinoma. All of the trials compared Coriolus versicolor with conventional cancer treatment versus conventional cancer treatment alone (with or without placebo). The dose range was 1-3.6g daily, and the duration of therapy was 1-36 months. In total, 1,284 patients with cancer were studied who received Coriolus versicolor versus 1,303 in the conventional cancer treatment group. There was a statistically significant difference in overall survival at 5 year (P < 0.00001; RR = 1.14 95% CI = 1.09, 1.20). AE’s were not significantly increased as a result of Coriolus versicolor intervention. Of patients randomized to Coriolus versicolor, there was a 9% absolute reduction in 5-year mortality, resulting in one additional patient alive for every 11 patients treated. The authors state that in patients with breast, gastric, or colorectal cancer treated with conventional cancer treatments and Coriolus versicolor, the overall 5-year survival rate was more profound (Wong 2012).
A systematic review and meta-analysis was conducted to assess the efficacy of Coriolus versicolor on overall survival in patients with cancer (Wong 2012). Thirteen randomized trials were included. Types of cancer (encompassing stage I to IV) in these studies included esophageal, gastric, colon/rectal, breast and nasopharyngeal carcinoma. All of the trials compared Coriolus versicolor with conventional cancer treatment versus conventional cancer treatment alone (with or without placebo). The dose range was 1-3.6g daily, and the duration of therapy was 1-36 months. In total, 1,284 patients with cancer were studied who received Coriolus versicolor versus 1,303 in the conventional cancer treatment group. There was a statistically significant difference in overall survival at 5 year (P < 0.00001; RR = 1.14 95% CI = 1.09, 1.20). AE’s were not significantly increased as a result of Coriolus versicolor intervention. Of patients randomized to Coriolus versicolor, there was a 9% absolute reduction in 5-year mortality, resulting in one additional patient alive for every 11 patients treated. The authors state that in patients with breast, gastric, or colorectal cancer treated with conventional cancer treatments and Coriolus versicolor, the overall 5-year survival rate was more profound (Wong 2012).
Dosage: 3,000-6,000 mg of hot-water extract daily, containing a minimum of 20-40% beta-glucans.
Safety: Caution with patients diagnosed with an autoimmune condition, and/or using immunosuppressive medications.
Maitake (Grifola frondosa)
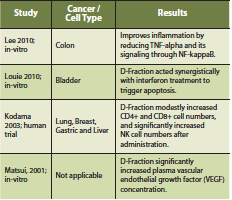
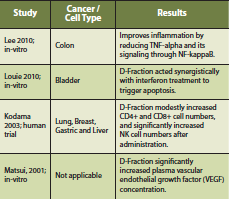
Maitake mushroom consists of a beta-glucan termed D-Fraction, which is extracted from the fruiting body (Kodama 2003). D-Fraction has been found to stimulate the function of immune cells such as macrophages, helper T cells, cytotoxic T cells and NK cells (Kodama 2003). Case series reports and clinical trials have reported an immune-stimulatory effect from Maitake mushroom, showing an increase in CD4+ cell count after administration (Kodama 2002). Furthermore, Maitake may improve overall quality of life, reduce tumor burden and act synergistically with chemotherapy. These effects were most profound in patients with hepatocellular, breast and lung cancer (Kodama 2002).
A phase I/II clinical study of Maitake mushroom was conducted on thirty-four postmenopausal breast cancer patients, free of disease after initial treatment, to detect immunological effects (Deng 2009). The authors state that no dose-limiting toxicity was encountered and that there appears to be no ‘maximum dose’, only ‘optimal dose’ depending on the immunological outcome desired. Some of the parameters measured increased with an increasing Maitake dose (CD4+ and CD8+ cells), whereas others were highest at an intermediate dose of Maitake (IL-10, IL-2, TNF-alpha) (Deng 2009).
There is a complex interaction of Maitake mushroom with the innate and adaptive immune system. Depending on the dose, there can be specific immune stimulatory and/or immune suppressing effects. Preliminary evidence also suggests that intravenous vitamin C may act synergistically with Maitake D-Fraction to improve conventional cancer treatments (Konno 2009).
Dosage: 500-3,000 mg of hot-water extract daily, containing a minimum of 20% polysaccharides.
Safety: Caution with patients diagnosed with an autoimmune condition, and/or using immunosuppressive medications.
Reishi (Ganoderma lucidum)
Ganoderma lucidum has been used to treat a variety of conditions for centuries in China, Korea and Japan (Sliva 2003). Traditionally, it has been used to treat hypertension, hyperlipidemia, viral infections, cardiovascular disease, asthma and cancer (Sliva 2003). The direct cytotoxic and anti-angiogenesis mechanisms of Ganoderma lucidum have been reproducibly demonstrated by in-vitro studies (Yuen 2005). Human clinical studies are required in order to better establish proper dosing and define the full range of activity of Ganoderma lucidum and its mechanisms of actions. The evidence available is conflicting and there are specific safety concerns with Ganoderma.
A recent Cochrane review did not find sufficient evidence to justify the use of Ganoderma lucidum as a first-line treatment for cancer (Jin 2012). However, the authors state that there is sufficient evidence to consider Ganoderma lucidum as an adjunctive treatment to conventional cancer therapies due to its potential of reducing tumor burden and stimulating immunity. Importantly, no major toxicity was noted in the studies reviewed with only a few reports of minor adverse events.
There is concern that Ganoderma lucidum may inhibit platelet aggregation and is a relative contraindication for patients on antiplatelet medication (Tao 1990). Evidence is currently conflicting, with studies stating that at doses of 1.5g Ganoderma lucidum does not impair hemostatic function and increase risk of bleeding (Kwok 2005). However, at higher doses hemostatic function may be impaired and currently, caution is advised.
Dosage: 1000-4000 mg of hot water extract daily, containing a minimum of 10% polysaccharides.
Safety: Caution with patients diagnosed with an autoimmune condition, and/or using immunosuppressive medications. Caution is also advised for patients on concomitant hypotensive and/ or antiplatelet therapy.
Conclusion
Many patients with cancer use complementary and alternative medicine as an adjunct to their care, including the use of mushrooms. Many immunological mechanisms were described. In terms of outcomes, Agaricus was shown to induce apoptosis in a number of cancer types. In clinical trials of patients with remission, it had a good safety profile with a limited number of AE’s (Ohno 2011). Coriolus is the most well-studied mushroom and in studies of 5 cancer types showed a 9% absolute reduction in 5-year mortality, resulting in one additional patient alive for every 11 patients treated (Wong 2012). Grifolia was shown to stimulate various immune cells, but had limited data with regards to patient outcomes. Ganoderma had some conflicting evidence regarding its clinical effects and had potential safety concerns due to the potential inhibition of platelet aggregation (Kwok 2005). Overall, mushrooms should be used in doses of 500-6,000mg per day, with some variation based on the specific mushroom. Additional precautions should be taken in patients diagnosed with an autoimmune condition or using immunosuppressive medications. The evidence supports the use of mushrooms as clinically useful adjuncts to conventional cancer care, especially Coriolus versicolor.
References
Akiyama H, Endo M, Matsui T, Katsuda I, Emi N, Kawamoto Y, Koike T, Beppu H. Agaritine from Agaricus blazei Murill induces apoptosis in the leukemic cell line U937. Biochem Biophys Acta. 2011 May;1810(5):519-25.
Chu K, Ho S, Chow A. Coriolus versicolor: A Medicinal Mushroom with Promising Immunotherapeutic Values. Journal of Clinical Pharmacology. 2002;42:976-984.
Deng G, Lin H, Seidman A, Fornier M, D’Andrea G, Wesa K, Yeung S, Cunningham- Rundles-S, Vickers A, Cassileth B. A phase I/ II trial of a polysaccharide extract from Grifola frondosa (Maitake mushroom) in breast cancer patients: immunological effects. J Cancer Res Clin Oncol. 2009;135:1215-1221.
Hetland G, Johnson E, Lyberg T, Bernardshaw S, Tryggestad A, Grinde B. Effects of the Medicinal Mushroom Agaricus blazei Murill on Immunity, Infection and Cancer. Scandinavian Journal of Immunology. 2008 July;68:363-370.
Jin X, Ruiz J, Sze DMY, Chan GCF. Ganoderma lucidum (Reishi mushroom) for cancer treatment (Review). The Cochrane Collaboration. 2012;6:1-38.
Kimura Y, Kido T, Takaku T, Sumiyoshi M, Baba K. Isolation of an anti-angiogenic substance from Agaricus blazei Murill: its antitumor and antimetastatic actions. Cancer Sci. 2004 Sept;95(9):758-64.
Kodama N, Komuta K, Nanba H. Can Maitake MD-Fraction Aid Cancer Patients? Alternative Medicine Review. 2002;7(3):236-9.
Kodama N, Komuta K, Nanba H. Effect of Maitake (Grifola frondosa) D-Fraction on the Activation of NK cells in Cancer Patients. J Med Food. 2003;6(4):371-77.
Konno S. Synergistic potentiation of D-Fraction with vitamin C as possible alternative approach for cancer therapy. Int J Gen Med. 2009 Jul;2:91-108.
Kwok Y, Ng K, Li C, Lam C, Man R. A Prospective, Randomized, Double-Blind, Placebo Controlled Study of the Platelet and Global Hemostatic Effects of Ganoderma lucidum (Ling-Zhi) in Healthy Volunteers. Anesth Analg. 2005;101:423-6.
Lee JS, Hong EK. Agaricus blazei Murill enhances doxorubicin-induced apoptosis in human hepatocellular carcinoma cells by NFkB-mediated increase of intracellular doxorubicin accumulation. Int J Oncol. 2011 Feb;38(2):401-8.
Lee JS, Park SY, Thapa D, Choi MK, Chung IM, Park YJ, Yong CS, Choi HG, Kim JA. Grifola frondosa water extract alleviates intestinal inflammation by suppressing TNF-alpha production and its signaling. Exp Mol Med. 2010 Feb;42(2):143-54.
Lima C, Cordova C, Nobrega O, Funghetto S, Karnikowski M. Does the Agaricus blazei Murill Mushroom Have Properties That Affect the Immune System? An Integrative Review. Journal of Medicinal Food. 2011 March;14(1/2):2-8.
Louie B, Rajamahanty S, Won J, Choudhury M, Konno S. Synergistic potentiation of interferon activity with maitake mushroom d-fraction on bladder cancer cells. BJU Int. 2010 Apr;105(7):1011-5.
Martinez-Montemayor MM, Acevedo RR, Otero-Franqui E, Cubano LA, Dharmawardhane SF. Ganoderma lucidum (Reishi) inhibits cancer cell growth and expression of key molecules in inflammatory breast cancer. Nutr Cancer. 2011;63(7):1085-94.
Matsui K, Kodama N, Nanba, H. Effects of maitake (Grifola frondosa) D-Fraction on the carcinoma angiogenesis. Cancer Lett. 2001 Oct;172(2):193-8.
Ng T. A Review of Research on the Protein-Bound Polysaccharide (Polysaccharide, PSP) from the Mushroom Coriolus versicolor (Basidiomycetes: Polyporaceae). Gen. Pharmac. 1998;30(1):1-4.
Ohno S, Sumiyoshi Y, Hashine K, Shirato A, Kyo S, Inoue M. Phase I Clinical Study of the Dietary Supplement, Agaricus blazei Murill, in Cancer Patients in Remission. Evid Based Complement Alternat Med. 2011.
Ohwada S, Kawate S, Ikeya T, Yokomori T, Kusaba T, Roppongi T, Takahashi T, Nakamura S, Kawashima Y, Nakajima T, Morishita Y. Adjuvant Therapy With Protein-Bound Polysaccharide K and Tegafur Uracil in Patients with Stage II or II Colorectal Cancer: Randomized, Controlled Trial. Dis Colon Rectum. 2003 Aug;46(8):1060-8.
Oka S, Tanaka S, Yoshida S, Hiyama T, Ueno Y, Ito M, Kitadai Y, Yoshihara M, Chayama K. A water-soluble extract from culture medium of Ganoderma lucidum mycelia suppresses the development of colorectal adenomas. Hiroshima J Med Sci. 2010 Mar;59(1):1-6.
Shibata M, Shimura T, Nishina Y, Gonda K, Matsuo S, Abe H, Yajima Y, Nakamura I, Ohki S, Takenoshita S. PSK decreased FOLFOX4-induced peripheral neuropathy and bone marrow suppression in patients with metastatic colorectal cancer. Gan To Kagaku Ryoho. 2011 May;38(5):797-801.
Sliva D. Ganoderma lucidum (Reishi) in Cancer Treatment. Int Cancer Therapies. 2003;2(4):358-364.
Su ZY, Tung YC, Hwang LS, Sheen LY. Blazeispirol A from Agaricus blazei fermentation product induces cell death in human hepatoma Hep 3B cells through caspase-dependent and caspase-independent pathways. J Agric Food Chem. 2011 May;59(9):5109-16.
Tanaka H, Muguruma K, Ohira M, Kubo N, Yamashita Y, Maeda K, Sawada T, Hirakawa K. Impact of adjuvant immunochemotherapy using protein-bound polysaccharide-K on overall survival of patients with gastric cancer. Anticancer Res. 2012 Aug;32(8):3427-33.
Tao J, Feng KY. Experimental and clinical studies on inhibitory effect of ganoderma lucidum on platelet aggregation. J Tongji Med Univ. 1990;10:240-3.
Thyagarajan A, Zhu J, Sliva D. Combined effect of green tea and Ganoderma lucidum on invasive behavior of breast cancer cells. Int J Oncol. 2007 Apr;30(4):963-9.
Tsang KW, Lam CL, Mak JC, Ooi GC, Ho JC, Lam B, Man R, Shan JS, Lam WK. Coriolus versicolor polysaccharide peptide slows progression of advanced non-small cell lung cancer. Respir Med. 2003 Jun;97(6):618-24.
Weng CJ, Chau CF, Yen GC, Liao JW, Chen DH, Chen KD. Inhibitory effects of ganoderma lucidum on tumorigenesis and metastasis of human hepatoma cells in cells and animal models. J Agric Food Chem. 2009 Jun;57(11):5049-57.
Wong E, Fai C, Chung L. Efficacy of Yun Zhi (Coriolus versicolor) on Survival in Cancer Patients: Systematic Review and Meta-Analysis. Recent Patents on Inflammation and Allergy Drug Discovery. 2012;6:78-87.
Wu B, Cui J, Zhang C, Li Z. A polysaccharide from Agaricus blazei inhibits proliferation and promotes apoptosis of osteosarcoma cells. Int J Biol Macromol. 2012 May; 50(4): 1116-20.
Yoshikawa T, Tsuburaya A, Saze Z, Aoyama T, Hasegawa S, Kanemoto A, Terashima M, Tahara H. Randomized phase II trial to compare S-1 and S-1/PSK for advanced or recurrent gastric cancer-lessons from the results. Gan To Kagaku Ryoho. 2011 Nov;38(12):1909-11.
Yu CH, Kan SF, Shu CH, Lu TJ, Sun-Hwang L, Wang PS. Inhibitory mechanisms of Agaricus blazei Murill on the growth of prostate cancer in vitro and in vivo. J Nutr Biochem. 2009 Oct; 20(10):753-64.
Yuen J, Gohel M. Anticancer Effects of Ganoderma lucidum: A Review of Scientific Evidence. Nutrition and Cancer. 2005;53(1):11-17.
Zhao S, Ye G, Fu G, Cheng JX, Yang BB, Peng C. Ganoderma lucidum exerts anti-tumor effects on ovarian cancer cells and enhances their sensitivity to cisplatin. Int J Oncol. 2011 May;38(5):1319-27
The Pear Tree Clinic
Leaders in integration – promoters of regulation
We at IHP are pleased to cover The Pear Tree Clinic, located in Dieppe, New Brunswick. Dr. Melissa Blake, ND is the owner of the clinic. She graduated from the Canadian College of Naturopathic Medicine in 2006 and knew at that time that she wanted to return to the Maritimes. She first opened The Pear Tree in Sackville and then decided after 2 years to move to the larger Moncton area with hopes of growing The Pear Tree into a multi-disciplinary medical center. Dr. Blossom Bitting, ND joined Dr. Blake in 2009 and they have been practicing together successfully for the last 4 years. Both NDs have recently become mothers and impressively still manage to service approximately 200 patient visits per week.
The clinic is 3000 square feet and includes 6 treatment rooms, an infrared sauna, and a yoga studio. The other members of the clinic include Andrée Surette Poirier, RMT and Sari LaBelle (HSI), CYT. Andrée focuses her practice on stress reduction and stress management. She is also a Reiki Master Teacher and utilizes many different techniques in personalizing her treatments to her patient’s needs. Sari is a Life Coach, Yoga teacher, and Meditation instructor. She leads group sessions and helps her patients actualize life goals through selfexploration, body and mind awareness, and creative expression. The Pear Tree Clinic is unique in that they are the only integrative clinic in their area that offers naturopathic services and other alternative health care services. Their team approach is reflected in the clinic slogan: “Pearing together, for your health and wellness.”
Staying connected is important to the clinic team members. The team meets once a week to discuss clinic operations and upcoming events. During these meetings, they bring up challenging cases and work together to develop comprehensive, individualized plans. Their mission is to make an impact in the wellness of each person who walks through their door. They believe a team-based approach is the best way of achieving this. As a result, they put a significant effort towards networking and continually contact other local health care providers, in hopes of improving the health service options available for their patients. Naturopathic Doctors are not regulated in New Brunswick. Dr. Blake and Dr. Bitting try to bridge the gap between NDs and MDs by attending and hosting networking events, educating the public and other health care practitioners, and by providing high-quality holistic care to their patients. They are both members of the New Brunswick association of Naturopathic Doctors (NBAND). Dr. Blake is also a member of the Nova Scotia Association.
New patients usually find the clinic through their website or online through the yellow pages. The practitioners also see a large amount of patients from word-of-mouth referrals and from external referrals. For new patients, the clinic’s receptionist is responsible for filtering primary health goals and then directing the patient to the appropriate practitioner. Although both NDs enjoy general family practice, Dr. Blake’s practice focuses on cardiovascular health and oncology, while Dr. Bitting’s practice focuses on pediatrics and fertility. The NDs are usually the primary contact for patients and manage the addition of other practitioners as needed. If the patients present with musculoskeletal complaints or a high degree of stress, they usually refer to the massage therapist. If weight loss is a concern, they also suggest tailored yoga programs and will meet with the patients on an individual basis as well as in groups.
The clinic has a natural health product dispensary. It includes custom tinctures using herbs from Viriditas and St-Francis Herb Farm. They carry supplements from professional product lines, including AOR, Genestra, CanPrev, Vitazan, Thorne, and Restorative Formulations. They carry homeopathics from Boiron. Dr. Blake does all blood draws for the clinic and then sends out to labs such as Gamma-Dynacare Laboratories, Rocky Mountain Analytical, Doctor’s Data (especially for heavy metals), and Neuroscience. The clinic’s most popular lab test is Rocky Mountain Analytical’s IgG test for food allergies, followed by Vitamin D testing.
Due to the lack of regulation, the public still has misconceptions about the ‘ND’ designation and other competing designations that non-regulated health care practitioners use, such as ‘nd’, ‘naturopath’, and ‘naturotherapist’. Many health plans cover services by nonregulated practitioners. One concern for the NDs is that many people assume they can’t afford naturopathic services. Another concern the practitioners have is that patients sometimes think that because they are seeing a medical doctor, seeing an ND may be counterproductive. The medical doctors can also perpetuate this idea because they may have had a bad experience with an unregulated practitioner. To remedy this, the clinic does a lot of community outreach work. They send letters to MPs and they try to work with local MDs by initiating and maintaining communication. A lot of education also occurs within the community. Both NDs write articles for local papers and send out monthly newsletters to their mailing list. Dr. Blake believes that by offering IV therapy, it has helped expand the clinic’s reach and credibility.
Legislation is a big challenge and is a hot topic at provincial meetings. The largest obstacle faced by Naturopathic Doctors is the low number of practitioners. Unfortunately, this is a self-perpetuating problem. Because the province is unregulated, fewer NDs may want to practice there. In turn, because there are not a lot of NDs practicing, it becomes difficult to justify regulation! However, Dr. Blake has seen a large shift over the last several years.
The public is more educated and the number of NDs has doubled provincially over the last 6 years. In an effort to promote awareness, the team has been actively involved in many wellness and health expos that occur in the area and have hosted successful networking events for health care practitioners. Future plans for the clinic include expanding the team. They are actively seeking other practitioners who would fit well within their integrative model. They would especially like to find a health care practitioner that is linked to the conventional system, such as a Nurse Practitioner or an MD. The clinic hosts health programs and workshops for the public in an effort to continue integrating into the healthcare system. They are also looking forward to implementing a community acupuncture program, whereby multiple acupuncture treatments could occur simultaneously. IHP would like to thank The Pear Tree Clinic for allowing us to showcase their accomplishments.
The clinic hosts health programs and workshops for the public in an effort to continue integrating into the healthcare system.
Dr Tris Trethart, MD
Dr Tris
30 years of integrative medicine and counting…
It is always a special thing when the IHP team has the privilege of showcasing a true pioneer of integrative medicine; Dr Trethart’s 30 year tenure of delivering world- class integrative care to the Edmonton, Alberta community is indeed such an occasion. In 2013, anyone interested in educating themselves in integrative medicine has dozens of accredited academic institutions, thousands of PubMed indexed clinical trials and review articles, and a lifetime of web- based resources to call upon. As Dr Trethart wrapped up his medical residency in 1982, such well- developed resources of education simply did not exist.
Trethart, MD
Named during his medical training “most likely to administer IV carrot juice and bran muffins”, his vision of applying integrative medical techniques was no mystery to his colleagues. Traveling such a path was invariably an infinitely greater challenge in 1982 than it is today. Thanks to the tireless efforts of Dr Trethart and others, so many of us today have the relatively easy ability to enter the discipline of integrative medicine and thrive.
While operating a truly eclectic practice, Dr Trethart describes his facility as “anti aging focused – the practice of aging with dignity”.
 As Dr Trethart worked on his undergraduate training, he found himself suffering from a long list of common, chronic ailments fatigue, aches/ pains, insomnia, depression. He came across William Dufty’s book Sugar Blues and decided to attempt to eliminate sugar from his diet. After routing through all his cupboards and throwing away any food containing sugar, he realized he had no food left! The “radical” change in his diet to eliminate sugar quickly, within a handful of weeks, resolved many of the chronic concerns he was experiencing. An inclination towards diet and lifestyle as medicine, as opposed to a reliance on pharmacotherapy, was firmly entrenched in Dr Trethart upon his arrival at the University of Manitoba school of medicine.
As Dr Trethart worked on his undergraduate training, he found himself suffering from a long list of common, chronic ailments fatigue, aches/ pains, insomnia, depression. He came across William Dufty’s book Sugar Blues and decided to attempt to eliminate sugar from his diet. After routing through all his cupboards and throwing away any food containing sugar, he realized he had no food left! The “radical” change in his diet to eliminate sugar quickly, within a handful of weeks, resolved many of the chronic concerns he was experiencing. An inclination towards diet and lifestyle as medicine, as opposed to a reliance on pharmacotherapy, was firmly entrenched in Dr Trethart upon his arrival at the University of Manitoba school of medicine.
Dr Trethart embraces all tenants encompassing the discipline of integrative medicine. Truly patient-centred, individualized, integrated care is delivered. The facilities name? As unassuming as the facilities only physician; Dr Trethart’s Clinic.
The system of care delivery is impressive, and certainly worth noting. An initial visit is utilized to provide a classic intake with physical exam, and any appropriate conventional labs are ordered. The second visit reviews the findings of the physical and basic lab work, integrative lab tests are discussed, and a basic diet/ supplement program is initiated. Dr Trethart is a firm believer in the use of a myriad of integrative diagnostic tests, sighting them as key in the process of doctor – as – detective, necessary to achieve truly individualized patient care. Food sensitivity panels are ordered on virtually every patient. Thereafter, comprehensive stool analysis, provocative heavy metal assessment, detailed nutrition profiles, and others are utilized frequently.
Dr Trethart described his practice as a “resort practice”. By this he meant patients come to him as a last resort. Integrative healthcare providers across the country are no doubt familiar with such a scenario. In his own words “I see more interesting, unique, and difficult cases in one day than a GP sees in six months”. I wholeheartedly agree Dr Trethart, and that isn’t something they teach us in ND school, either. While operating a truly eclectic practice, Dr Trethart describes his facility as “anti aging focused – the practice of aging with dignity”.
Dr Trethart is the only clinician in the facility, yet the facility services over 125 patients per week, in addition to 30-40 IV therapies per week. This impressive patient volume is co-managed by an incredible and dedicated team of professionals; two executive administrators act as office managers, yet also involve themselves as patient coaches, fielding phone questions from patients, running patients through the process of specimen collection for integrative diagnostic tests, etc… There are also two receptionists and an RMT. The on- site dispensary utilizes a manager and three support staff. A nurse and nurse assistant round out the team, principally managing the IV room of the facility.
Common IV therapies employed include EDTA, DMPS, Plaquex, IV-C, Meyer’s, bio-oxidative therapies, glutathione, and others. Dr Trethart is also a firm supporter of breast thermography, running the technique in-house, and utilizing a Toronto-based lab for interpretation of outcomes.
We asked Dr Trethart what it was like starting an integrative practice so many years ago, where he turned for training, if he has experienced scrutiny from regulators along the way, and if he has noticed a change in perception of his work from colleagues and/ or regulators over the years. He explained that even from the days of his residency there were “watchful eyes” looking over his shoulder, because he would provide lifestyle advice while on rotation. Thereafter he described “extensive scrutiny”

John Trethart, David Carr; front row: Karen Fech, Nelly Velasco, Monique Da Costa,
Jade Gionet, Lesley Erskine.
Dr Trethart described his practice as a “resort practice”. By this he meant patients come to him as a last resort. Integrative healthcare providers across the country are no doubt familiar with such a scenario. In his own words “I see more interesting, unique, and difficult cases in one day than a GP sees in six months”.
from the provincial regulator, beginning in 1983 with an inquiry because he referred a war vet with back pain to see a chiropractor. In honesty, I expressed to Dr Trethart that relative to other integrative healthcare providers across Canada, his interactions with regulators in fact seem minimal; he was somewhat relieved to hear such a comment. From a perspective of his colleagues, he did note that over the years there has been a consistent increase in referrals he obtains from local conventional practitioners. He still feels however the gap between conventional and integrative providers remains very large.
I undertake these interviews with leading Canadian integrated healthcare providers to create awareness among doctors across Canada of the important, tireless, selfless, and immensely valuable work people like Dr Trethart perform daily. Never have I been so directly impacted by an interview as I was by Dr Trethart. For every point or two I jotted down about him and his practice, I was furiously writing pages of notes to immediately implement in my private practice. Dr Trethart is a wealth of knowledge and experience. He is truly among the most gifted of integrated healthcare providers in the country. He made a tragic mistake during our interview, however; he informed me that his facility is not visited by ND preceptors! Rest assured Dr Trethart, I am confident you will be swarmed with them in the years to come! You have been keeping yourself a secret for far too long… Thank you for allowing us to showcase your efforts to our readership. Thank you for traveling down this path in a time when it more resembled a brick wall than a path.
And thank you in advance for opening your doors to aspiring ND’s…
You will be seeing them soon.
Dr Trethart is a firm believer in the use of a myriad of integrative diagnostic tests, sighting them as key in the process of doctor – as – detective, necessary to achieve truly individualized patient care.
Iron & Diabetes
Iron & Diabetes
A review
Abstract
Both anemia and iron overload are commonly encountered problems in clinical practice. In between is a huge gray area of iron levels, measured by various markers, that fall in the normal reference ranges and are viewed as not a cause for concern. However, to take as an example the risk of acute cardiovascular events in women, the prevalence is very small prior to menopause and then increases to approach the risk level where postmenopausal women have a prevalence similar to adult men. This has been attributed to the very low systemic iron burden in premenopausal women and has encouraged the questioning of the optimum levels in both men and postmenopausal women. This is not a frivolous suggestion since the iron levels, or more properly the iron stores, are easily adjustable by blood donation or phlebotomy and blood iron stores of premenopausal women are easily achieved by adult men, apparently with negligible risk of side effects including anemia. Rather like the argument of those who believe in statins that the lower the LDL the better, a valid concern can be raised as to the appropriate range of iron stores in both adult men and postmenopausal women. Elevated iron stores lead to elevated iron available for redox reactions which act as generators of reactive oxygen species and highly significant inflammation, both of which are associated with a number of chronic diseases having inflammation as a part of their etiology. In this review, the connection between iron overload and diabetes will be discussed.
Introduction
It is well established that humans need a certain level of iron for health, but higher levels are potentially toxic (Kell 2009). From the point of view of evolution, normal stores of iron during reproductive years provided reserves for hemorrhage or periods of severe dietary restrictions or starvation. However there is no homeostatic mechanism for excreting excess iron to maintain a certain level.
Hemochromatosis and thalassaemia involve very high pathological iron levels, but even mildly elevated iron stores over time which are implicated in excessive redox (oxidation-reduction) reactions have been associated with the pathogenesis of cancer, neurodegenerative disorders, atherosclerosis, cardiovascular disease, peripheral artery disease and diabetes (Kell 2009). Iron is mostly sequestered as ferritin, a ubiquitous intracellular protein that both stores iron and releases it in a controlled manner in a safe form. Transferrin also ties up iron but can be saturated. Its function is to transport iron through the blood to the liver, spleen and bone marrow, with the iron in part coming from diet and erythrocyte breakdown. Significant serum levels of non-transferrin bound iron can also be present. It is redox active, and appears to be present even when transferrin saturation is absent (Lee 2006). The blood level of ferritin is the most commonly used marker for the magnitude of iron stores and iron overload.
Elevated ferritin levels are not always a true indicator of iron stores since it is an acute phase reactant. In most studies involving diabetes, this does not appear to be an issue. Mean ferritin levels in the US population study NHANES III were for men about 150 ng/mL, for menstruating women ages 17-49, 25-35 ng/mL, and for menopausal women ages 50-59, 60 ng/mL and for ages > 60, about 90-100 ng/mL (Zacharski 2000). Low levels of stored iron in premenopausal women have been frequently evoked as an explanation for the well known low risk of cardiovascular disease. These numbers raise concerns that the normal male level or the level found in older postmenopausal women may be associated with elevated risk of morbidity or mortality.
Toxicity of Iron
The toxicity of iron is related to its role in producing oxidative damage and the ease with which it is reversibly oxidized and reduced. Ferrous iron catalyzes the production of the highly toxic and reactive hydroxyl radical from hydrogen peroxide, whereas superoxide dismutase serves to equilibrate superoxide and peroxide. Oxidized iron regenerates reduced iron by reacting with superoxide with redox cycling. This results in reactive oxygen species (ROS) responsible for oxidative stress. When these overwhelm the antioxidant capacity, damage occurs which has been related to diseases of aging and inflammation (Brewer 2007). One view is that the dangers of excess iron operate through inadequately liganded (tied up) iron ions. When the ions are tightly liganded they are unreactive but weak ligand formation leaves some reactive free iron. The pathophysiology of iron is thus related to the availability of so-called catalytic iron, or iron that is available to participate in free radical reactions. Due to their weak antioxidant defences the pancreatic β-cells are particularly susceptible to oxidative damage such as can be caused by iron-generated reactive oxygen species (Tiedge 1997). In fact, iron has been used to induce diabetes in animals. Correlations with ferritin levels are an indirect measure of risk from iron-induced oxidative stress, and high ferritin levels correlate with elevated inflammatory cytokine levels (Depalma 2010).
The Association between Iron Levels, Diabetes and the Metabolic Syndrome
The potential role of iron in the pathogenesis of diabetes is suggested by several observations (Rajpathak 2009): (1) Increased incidence of diabetes is seen in patients with diverse causes of iron overload. (2) The reduction in iron load by either chelation or phlebotomy can improve glycemic control or reverse diabetes. (3) Dietary intake of heme iron (e.g. red meat) and concomitant increases in iron stores is associated with the risk of developing diabetes. (4) Insulin sensitivity and insulin secretion are increased by frequent blood donation. The molecular mechanisms are numerous and incompletely understood but include oxidative stress, modulation of adipokines and intracellular singling transduction pathways (Simcox 2013, Swaminathan 2007).
It has been argued that the modest elevations of ferritin observed in diabetes may be a consequence of the disorder rather than a causal factor impacting insulin resistance and β-cell function and apoptosis (Jehn 2007). However, evidence suggests otherwise. Excessive amounts of nontransferrin- bound iron, the form most susceptible to redox activity, are found in diabetic patients with a strong gradient for disease severity (Lee 2006). Furthermore, as will be discussed below, phlebotomy in type 2 diabetes results in improvements in glycemic control and insulin sensitivity which also supports the hypothesis that iron plays a pathogenic role.
Two recently published systematic reviews plus metaanalyses have examined the association of diabetes incidence with ferritin levels. One study (Zhao 2012) reports a meta-analysis of 12 prospective or crosssectional studies which analyzed ferritin levels and involved 4366 type 2 diabetes patients and over 41,000 controls plus four studies that measured heme-iron intake involving 9246 type 2 diabetics and about 180,000 controls. It was found that for the highest vs. the lowest category of ferritin level, the risk of diabetes was increased 66% in prospective studies and 130% in cross-sectional studies. A similar comparison for heme-iron intake yielded a 31% risk increase.
A second study (Bao 2012) examined the association between the risk of diabetes and dietary iron intake in prospective studies. A meta-analysis of five studies gave a pooled relative risk increase of 33% in a comparison of the lowest vs. the highest heme-iron intake. For elevated ferritin levels, they found a 70% increase in relative risk in multivariableadjusted models (seven studies) and a 63% increase in multivariable-adjusted models which included inflammatory markers (five studies). There was no significant association with dietary intake and risk for non-heme or supplemental iron intake, a result consistent with the high bioavailability only of heme iron. Incidentally, another study found a correlation between ferritin levels and the risk of diabetic retinopathy (Canturk 2003).
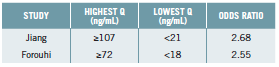
For postmenopausal women (56-62 years of age) who typically have ferritin levels between 70 and 90 ng/dL (Zacharski 2000), data taken from one of the above studies (Bao 2012) for the risk of developing type 2 diabetes are given in the table below according to the highest vs. the lowest ferritin quartiles.
It would seem that for women in this age group, levels similar to the NHANES average already carry significant risk.
A recent review and meta-analysis relates to the above. At issue is the association between red meat intake and risk of developing type 2 diabetes. A 19% increase was found per 100 g/day of red meat and a 51% increase per 50 g/day of processed red meat (Micha 2012).
The metabolic syndrome (MetS) is recognized as a risk factor for diabetes. Population studies find an association between ferritin levels and the risk of MetS in the US (Jehn 2004), Korea (Lee 2011), and Germany (Wrede 2006). Ferritin levels are associated with the MetS in postmenopausal but not premenopausal Korean women (Cho 2011), High levels of ferritin in a Chilean population correlated not only with a three-fold increase in developing MetS but for high levels of oxidative stress indicated by serum markers, there was a 21 fold increase in the development of this syndrome when the highest vs. the lowest quartiles were compared (Leiva 2013).
Clinical use of Phlebotomy to reduce Body Iron Stores
Both blood donation and phlebotomy can dramatically reduce iron stores (Houschyar 2012, Zacharski 2011). Repeated blood letting very efficiently lowers ferritin levels even if the initial values are very high such as seen in hemochromatosis. Furthermore, no induced anemia has been reported. The following studies are of interest:
• In a randomized controlled trial with metabolic syndrome patients, reduction of mean ferritin levels from 183 to 105 ng/mL using phlebotomy resulted in significant reductions in blood glucose, HbA1c, and systolic blood pressure. Changes in blood pressure and the HOMA-IR insulin resistance index correlated with ferritin reduction (Houschyar 2012).
• In a study comparing lacto-ovo vegetarians and meat eaters, the former were found to have mean ferritin levels of 35 ng/mL compared to 72 ng/mL for meats eaters and to have higher insulin sensitivity. When body stores of iron were lowered by phlebotomy in the meat eating group there was a 40% increase in insulin mediated glucose disposal (Hua 2001).
• The effect of phlebotomy on insulin resistance in a group of patients with non-alcoholic fatty liver disease and strongly elevated ferritin levels found a significant reduction in the HOMA-IR from 4.81 to 3.12 when ferritin levels were reduced from 438 to 52 ng/mL (Valenti 2007).
• In a study designed to examine the pathogenesis of diabetes associated with mutations of the hemochromatosis gene, 17 carriers comprising eight patients with diabetes and nine with normal glucose tolerance (NGT) were subjected to phlebotomy and the impact on insulin sensitivity and secretion investigated. Baseline ferritin levels were 942 and 1148 in the NGT and diabetic subjects, respectively. Ferritin targets were ≤ 100 ng/mL or ≤ 50 ng/mL depending of negative or positive evidence for iron deposits in the liver. The target levels were maintained and at 24 months the endpoint parameters measured. In both the NGT and diabetic groups, insulin secretion and insulin sensitivity increased. In the diabetic patients, fasting glucose declined from 137 to 105 mg/dL (7.6 to 5.8 mmol/L), the latter being close to normal (Equitani 2008).
• Earlier studies also found that reduction of iron stores consistently produces an improvement in insulin sensitivity and β-cell function (Fernandez- Real 2002, Fernandez-Real 2005).
Chelation, the Alternative to Phlebotomy or Blood Donation
Advanced glycation end products are thought to play a role in the complications of diabetes and the basic biochemistry involves reactive oxygen species including those attributed to iron activity (Nagai 2012). Oral chelation has been the traditional mainstream approach to iron overload for patients having pathological levels, and several prescription drugs are available. However, iron overloads involved in most of the studies discussed above are nowhere near those encountered in pathological iron overload. Furthermore, while studies, both interventional and observational, suggest target ranges for both men and women, optimum levels are clearly debatable. Chronic low-dose oral chelation therapy may be an important tool for the prevention and treatment of diabetic complications.
If one wishes to use non-pharmaceutical interventions to lower ferritin levels to, for example, to the upper end of the normal range for premenopausal women even if one is male, there are a number of “natural” iron chelators. N-acetyl cysteine is in fact a standard therapy for treating pediatric pathological iron overload even in infants. Green tea extract, curcumin, silymarin, alpha-lipoic acid (or R-lipoic acid) and quercetin all have documented success in iron chelation (Anderson 2012). These chelators also act to eliminate other toxic metals although for mercury it may help to add selenium to N-acetyl cysteine and lipoic acid. Curcumin was recently found to be a very good iron chelator (Jiao 2006). A recent randomized controlled trial demonstrated the effectiveness of curcumin in significantly improving markers of glucose metabolism in diabetics (Chuengsamarn 2012).
The results of the Trial to Access Chelation Therapy (TACT), a randomized, placebo controlled intravenous EDTA chelation trial, recently reported. Included were the results that for diabetics, there was a 39% relative risk reduction and a number needed to treat of eight (median follow-up 55 months) found for the composite primary endpoint of total mortality, recurrent MI, stroke, coronary revascularization or hospitalization for angina (Lamas 2013). EDTA chelation generally results in large increases in urine iron immediately post infusion (Cranton 2001).
The TACT result is particular interesting since intensive glycemic control with multiple drugs and even the addition of insulin fails to impact cardiovascular or total mortality or almost all other complications associated with diabetics (Boussageon 2012, Hemmingsen 2011, Turnbull 2012, Turnbull 2009). The dramatically lowered fasting blood glucose or HbA1c produced by intensive drug therapy may be viewed as successful control and treatment, but engenders false optimism while the pathogenesis of complications relentlessly progresses to ultimately yield clinical manifestations.
Iron Stores Reductions and Diabetic Complications
Studies on humans are limited. A nine-month study among patients with diabetes using deferiprone oral iron chelation reduced ferritin levels from 144 to 59 ng/mL and decreased the mean albumin/creatinine ratio from 187 to 25 mg/L (Rajapurkar 2013). In addition, a study involving the progression of diabetic nephropathy used a polyphenol–enriched, low-iron carbohydrate-restricted diet over four years. There was no significant change in HbA1c, but there was an absolute decrease in the incidence of serum creatinine doubling of 18% and a decrease in mortality or end-stage kidney disease of 18% over four years (number needed to treat over four years for either was six) (Facchini 2003). Iron chelation due to the polyphenols (from red wine, tea and polyphenol enhanced olive oil) was probably partly responsible for reduced ferritin from 325 to 53 ng/mL and may have been a major contributor to these results.
Conclusions
Studies are clearly needed to establish optimum iron stores in both men and women in the context of not only diabetes incidence, progression and complications, but also for all inflammation-related chronic diseases, in particular those associated with aging. Furthermore, large clinical studies are needed to examine the impact of lowering iron stores on the incidence of clinical manifestations of the complications of diabetes, not just markers of glucose metabolism. Can aggressive iron stores reduction, perhaps with severe carbohydrate restriction, cure type 2 diabetes, i.e. eliminate need for any medication? This question should have high priority. There are health issues associated with iron where one of the potentially effective interventions, blood donation, is free with almost no side effects. Iron stores as measured by ferritin can be dramatically lowered in most individuals without inducing anemia.
References:
Anderson, K., 2012. Excess Iron and Brain Degeneration. http://www.lef.org/magazine/mag2012/mar2012_ Excess-Iron-Brain-Degeneration_01.htm.
Bao, W., Rong, Y., Rong, S. and Liu, L. Dietary iron intake, body iron stores, and the risk of type 2 diabetes: a systematic review and meta-analysis. BMC Med 2012; 10: 119.
Boussageon, R., Supper, I., Bejan-Angoulvant, T., Kellou, N., Cucherat, M., Boissel, J.P., Kassai, B., Moreau, A., Gueyffier, F. and Cornu, C. Reappraisal of metformin efficacy in the treatment of type 2 diabetes: a meta-analysis of randomised controlled trials. PLoS Med 2012; 9(4): e1001204.
Brewer, G.J. Iron and copper toxicity in diseases of aging, particularly atherosclerosis and Alzheimer’s disease. Exp Biol Med (Maywood. ) 2007; 232(2): 323-335.
Canturk, Z., Cetinarslan, B., Tarkun, I. and Canturk, N.Z. Serum ferritin levels in poorly- and well-controlled diabetes mellitus. Endocr. Res 2003; 29(3): 299-306.
Cho, G.J., Shin, J.H., Yi, K.W., Park, H.T., Kim, T., Hur,J.Y. and Kim,S.H. Serum ferritin levels are associated with metabolic syndrome in postmenopausal women but not in premenopausal women. Menopause. 2011; 18(10): 1120-1124.
Chuengsamarn, S., Rattanamongkolgul, S., Luechapudiporn, R., Phisalaphong, C. and Jirawatnotai, S. Curcumin extract for prevention of type 2 diabetes. Diabetes Care 2012; 35(11): 2121-2127.
Cranton, E.M., 2001. A Textbook on EDTA Chelation Therapy. Hamptom Roads Publishing Co., Inc., Charlottesville, VA.
Depalma, R.G., Hayes, V.W., Chow, B.K., Shamayeva, G., May, P.E. and Zacharski, L.R. Ferritin levels, inflammatory biomarkers, and mortality in peripheral arterial disease: a substudy of the Iron (Fe) and Atherosclerosis Study (FeAST) Trial. J Vasc Surg 2010; 51(6): 1498-1503.
Equitani, F., Fernandez-Real, J.M., Menichella, G., Koch, M., Calvani, M., Nobili, V., Mingrone, G. and Manco, M. Bloodletting ameliorates insulin sensitivity and secretion in parallel to reducing liver iron in carriers of HFE gene mutations. Diabetes Care 2008; 31(1): 3-8.
Facchini, F.S. and Saylor, K.L. A low-iron-available, polyphenol-enriched, carbohydrate-restricted diet to slow progression of diabetic nephropathy. Diabetes 2003; 52(5): 1204-1209.
Fernandez-Real, J.M., Lopez-Bermejo, A. and Ricart, W. Iron stores, blood donation, and insulin sensitivity and secretion. Clin Chem 2005; 51(7): 1201-1205.
Fernandez-Real, J.M., Penarroja, G., Castro, A., Garcia-Bragado, F., Hernandez-Aguado, I. and Ricart, W. Blood letting in high-ferritin type 2 diabetes: effects on insulin sensitivity and beta-cell function. Diabetes 2002; 51(4): 1000-1004.
Hemmingsen, B., Lund, S.S., Gluud, C., Vaag, A., Almdal, T., Hemmingsen, C. and Wetterslev, J. Intensive glycaemic control for patients with type 2 diabetes: systematic review with meta-analysis and trial sequential analysis of randomised clinical trials. BMJ 2011; 343: d6898.
Houschyar, K.S., Ludtke, R., Dobos, G.J., Kalus, U., Broecker- Preuss, M., Rampp, T., Brinkhaus, B. and Michalsen, A. Effects of phlebotomy-induced reduction of body iron stores on metabolic syndrome: results from a randomized clinical trial. BMC Med 2012; 10: 54.
Hua, N.W., Stoohs, R.A. and Facchini, F.S. Low iron status and enhanced insulin sensitivity in lacto-ovo vegetarians. Br J Nutr 2001; 86(4): 515-519.
Jehn,M., Clark, J.M. and Guallar, E. Serum ferritin and risk of the metabolic syndrome in U.S. adults. Diabetes Care 2004; 27(10): 2422-2428.
Jehn,M.L., Guallar, E., Clark, J.M., Couper, D., Duncan, B.B., Ballantyne, C.M., Hoogeveen, R.C., Harris, Z.L. and Pankow, J.S. A prospective study of plasma ferritin level and incident diabetes: the Atherosclerosis Risk in Communities (ARIC) Study. Am J Epidemiol 2007; 165(9): 1047-1054.
Jiao,Y., Wilkinson, J., Christine, P.E., Buss, J.L., Wang, W., Planalp, R., Torti, F.M. and Torti, S.V. Iron chelation in the biological activity of curcumin. Free Radic Biol Med 2006; 40(7): 1152-1160.
Kell, D.B. Iron behaving badly: inappropriate iron chelation as a major contributor to the aetiology of vascular and other progressive inflammatory and degenerative diseases. BMC Med Genomics 2009; 2: 2.
Lamas, G.A., Goertz, C., Boineau, R., Mark, D.B., Rozema, T., Nahin, R.L., Lindblad, L., Lewis, E.F., Drisko, J. and Lee, K.L. Effect of disodium EDTA chelation regimen on cardiovascular events in patients with previous myocardial infarction: the TACT randomized trial. JAMA 2013; 309(12): 1241-1250.
Lee, B.K., Kim, Y. and Kim, Y.I. Association of serum ferritin with metabolic syndrome and diabetes mellitus in the South Korean general population according to the Korean National Health and Nutrition Examination Survey 2008. Metabolism 2011; 60(10): 1416-1424.
Lee, D.H., Liu, D.Y., Jacobs, D.R., Jr., Shin, H.R., Song, K., Lee, I.K., Kim, B. and Hider, R.C. Common presence of nontransferrin- bound iron among patients with type 2 diabetes. Diabetes Care 2006; 29(5): 1090-1095.
Leiva, E., Mujica, V., Sepulveda, P., Guzman, L., Nunez, S., Orrego, R., Palomo, I., Andrews, M. and Arredondo, M.A. High levels of iron status and oxidative stress in patients with metabolic syndrome. Biol Trace Elem. Res 2013; 151(1): 1-8.
Micha, R., Michas, G. and Mozaffarian, D. Unprocessed red and processed meats and risk of coronary artery disease and type 2 diabetes–an updated review of the evidence. Curr Atheroscler. Rep 2012; 14(6): 515-524.
Nagai, R., Murray, D.B., Metz, T.O. and Baynes, J.W. Chelation: a fundamental mechanism of action of AGE inhibitors, AGE breakers, and other inhibitors of diabetes complications. Diabetes 2012; 61(3): 549-559.
Rajapurkar, M.M., Hegde, U., Bhattacharya, A., Alam, M.G. and Shah, S.V. Effect of deferiprone, an oral iron chelator, in diabetic and non-diabetic glomerular disease. Toxicol Mech Methods 2013; 23(1): 5-10.
Rajpathak, S.N., Crandall, J.P., Wylie-Rosett, J., Kabat, G.C., Rohan, T.E. and Hu, F.B. The role of iron in type 2 diabetes in humans. Biochim Biophys Acta 2009; 1790(7): 671-681.
Simcox, J.A. and McClain, D.A. Iron and diabetes risk. Cell Metab 2013; 17(3): 329-341.
Swaminathan, S., Fonseca, V.A., Alam, M.G. and Shah, S.V. The role of iron in diabetes and its complications. Diabetes Care 2007; 30(7): 1926-1933.
Tiedge, M., Lortz, S., Drinkgern, J. and Lenzen, S. Relation between antioxidant enzyme gene expression and antioxidative defense status of insulin-producing cells. Diabetes 1997; 46(11): 1733-1742.
Turnbull, F. and Zoungas, S. Intensive glucose-lowering therapy in people with type 2 diabetes: what do we learn from a new meta-analysis of randomised controlled trials? Evid. Based. Med 2012; 17(3): 98-99.
Turnbull, F.M., Abraira, C., Anderson, R.J., Byington, R.P., Chalmers, J.P., Duckworth, W.C., Evans, G.W., Gerstein, H.C., Holman, R.R., Moritz, T.E., Neal, B.C., Ninomiya,T., Patel, A.A., Paul, S.K., Travert, F. and Woodward, M. Intensive glucose control and macrovascular outcomes in type 2 diabetes. Diabetologia 2009; 52(11): 2288-2298.
Valenti, L., Fracanzani, A.L., Dongiovanni, P., Bugianesi, E., Marchesini, G., Manzini, P., Vanni, E. and Fargion, S. Iron depletion by phlebotomy improves insulin resistance in patients with nonalcoholic fatty liver disease and hyperferritinemia: evidence from a case-control study. Am J Gastroenterol 2007; 102(6): 1251-1258.
Wrede, C.E., Buettner, R., Bollheimer, L.C., Scholmerich, J., Palitzsch, K.D. and Hellerbrand, C. Association between serum ferritin and the insulin resistance syndrome in a representative population. Eur J Endocrinol 2006; 154(2): 333-340.
Zacharski, L.R., Ornstein, D.L., Woloshin, S. and Schwartz, L.M. Association of age, sex, and race with body iron stores in adults: analysis of NHANES III data. Am Heart J 2000; 140(1): 98-104.
Zacharski, L.R., Shamayeva, G. and Chow, B.K. Effect of controlled reduction of body iron stores on clinical outcomes in peripheral arterial disease. Am Heart J 2011; 162(5): 949-957.
Zhao, Z., Li, S., Liu, G., Yan, F., Ma, X., Huang, Z. and Tian, H. Body iron stores and heme-iron intake in relation to risk of type 2 diabetes: a systematic review and meta-analysis. PLoS One 2012; 7(7): e41641.
Vitamin E
Vitamin E
Obstacles and opportunities in cancer prevention and treatment
Abstract
Vitamin E is one of the most researched compounds in the medical community because of what is believed to be its reduced risk association with a multitude of diseases, including cancer. There are eight forms of vitamin E, four tocopherols and four tocotrienols. Alpha tocopherol is by far the most studied form because it is the most biologically active (Aggarwal 2010). Gamma tocopherol and tocotrienols have shown the greatest promise with respect to recent cancer research. Gamma tocopherol has been found to prevent cancer cell growth both in vitro and in animal models by decreasing the formation of mutagens (Stone 2004). Cell-based studies have shown that tocotrienols exhibit even stronger anticancer activities than other forms of vitamin E, including gammatocopherol (Jiang 2000). For all forms of vitamin E, additional research is needed that involves human trials to understand the relationship between dietary supplementation, required dosages, and the long-term risks and benefits.
Introduction
Since Herbert M. Evans discovered vitamin E in his laboratory at the University of California, Berkley in 1922, vitamin E has become one of the most researched compounds within the medical community (Packer 2001). Gamma tocopherol and tocotrienols are often discussed within the medical literature as providing the greatest opportunities for disease prevention and treatment. The results of several studies have indicated that the tocotrienols may have stronger bioactivity than the tocopherols. Both types have shown anti-proliferative, pro-apoptotic and cyclooxygenase-2-inhibiting effects in in vitro studies (Wada 2012). These types of vitamin E have shown promise in treatment of a variety of cancers, including prostate cancer in men and breast cancer in women (Nesaretnam 2010, Sylvester 2005). The purpose of this paper is to examine some of the existing research regarding the potential benefits of gamma tocopherol and tocotrienols in relation to cancer prevention and treatment.
Vitamin E and Cancer
Before discussing the research that has shown the benefits of gamma tocopherol and tocotrienols with respect to cancer, it is important to briefly note the somewhat negative findings associated with supplemental alpha tocopherol intake and cancer. Alpha tocopherol is the most biologically active form of vitamin E in the human body and all other forms of vitamin E are measured in alpha tocopherol equivalents (National Academies Press 2000). A large randomized clinical trial, the SELECT trial, began in 2001 to determine whether daily supplementation with vitamin E (400 IU, as alpha tocopherol), with or without selenium (200 mcg), reduced the number of new prostate cancers in 35,533 healthy men over 50 years old. The trial was discontinued in 2008 when an analysis found that the supplements, taken alone or together for about five years, did not prevent prostate cancer. Results from an additional 1.5 years of follow-up from this trial showed that the men who had taken the alpha tocopherol had a 17 percent increased risk of prostate cancer compared placebo (Klein 2011). Other research has suggested that taking too much vitamin E can have pro-oxidant instead of anti-oxidant effects on cells (Brown 1997). Due to this effect, alpha tocopherol taken in high dosages may create a set of conditions in the body resulting in cancer cell formation that would not have otherwise occured. The warning that seems appropriate is that supplemental alpha tocopherol should not be viewed as a way of reducing cancer risk or cancer cell growth.
Gamma Tocopherol in Cancer Treatment
Research on human epithelial cells has indicated that gamma tocopherol, one of the forms of vitamin E and the most prevalent in the North American diet, has antiinflammatory properties as a COX-2 inhibitor that may be important in preventing a variety of diseases including cancer and cardiovascular disease (Jiang 2000). Research has shown that increased intake of gamma tocopherol was associated with a reduced risk of prostate cancer (Moyad 1999). Gamma tocopherol is superior to alpha tocopherol in the trapping of reactive nitrogen species, inhibition of COX-2 activity, activation of PPAR-γ and suppression of inflammation. Jihyeung et al. (2010) propose that a mixture of tocopherols, at ratios similar to those in our diet, could be a better cancer chemopreventive agent, based on their lab results demonstrating that gamma tocopherol inhibited colon, mammary, prostate and lung carcinogenesis in animal models. Human cell line study findings indicate that gamma tocopherol has potent anti-inflammatory activity and that at physiological concentrations, it may play an important role in human disease prevention (Jiang 2000). Well-designed human intervention trials with gamma tocopherol are needed to yield more definitive information on its cancer-preventive activities, and the most effective route of administration (ie. oral vs. injectable).
Tocotrienols in Cancer Treatment
As compared to tocopherols, tocotrienols have been found to be much more effective in prevention and inhibition of cancer cell growth in the body (Sylvester 2005). Part of the reason for this is that tocotrienols are more likely than tocopherols to achieve high cellular concentrations. The potential for higher cellular concentration of tocotrienols results in mitochondrial toxicity and apoptosis, a reduction of cancer cell reproduction, and the prevention of new cancer cell formation (Viola 2012). It has been noted that tocotrienols target different molecular structures than tocopherols. Specifically, the anti-inflammatory properties of tocotrienols have been found to suppress the transcription factor NF-kB, which has been linked to tumorigenesis and inhibition of HMG-CoA reductase, mammalian DNA polymerases, and some protein tyrosine kinases (Aggarwal 2010). Because of the unique anti-inflammatory properties and cell line targeting that occurs with tocotrienols, patients suffering from various types of cancers, specifically breast and prostate cancer, may be able to take a lower dosage of tocotrienols as compared to tocopherols, and achieve better results. This is important considering that research has indicated that high levels of vitamin E supplementation, in the form of alpha tocopherol, has been associated with increased levels of hemorrhagic stroke (Schurks 2010).
Research suggests that for women, taking tocotrienols as a dietary supplement may reduce the risk of developing breast cancer (Sylvester 2010). Part of the benefit of tocotrienols is indeed related to the lower dosages that are required in order to achieve the anti-inflammatory and anti-cancer properties. Other research has indicated that tocotrienols may increase immune function and response in the body (Nesaretnam 2012). The rationale is that tocotrienols bind to estrogen receptor beta ER-beta) leading to alteration of cell morphology and apoptosis of breast cancer cells (Nesaretnam 2012). Tamoxifen is currently one of the standard treatment in patients with estrogen receptor positive tumors. New research indicates that a combination of tamoxifen with tocotrienols may prolong the life of women with breast cancer (Nesaretnam 2012). A common problem with tamoxifen therapy is that patients develop resistance to the drug. Using a combination of tocotrienols and tamoxifen may reduce the problems that occur once patients develop tamoxifen resistance while also acting synergistically to extend breast cancer-specific survival (Nesaretnam 2012).
Therapeutic Dosing
The eight forms of vitamin E are not interconvertible within the human body. Instead, serum and intracellular concentrations of tocopherols and tocotrienols each rely upon the affinity of the hepatic alpha-tocopherol transfer protein (alpha-TTP) for their selective retention in plasma (National Academies Press 2000). As well, in vivo concentrations of vitamin E rely on the body’s ability to absorb each form. Typically, this absorption is below 80% and for some forms of vitamin E can be as low as 10% absorption from the gastrointestinal tract.
Although it is common for supplemental vitamin E products to measure doses in International Units (IU), this format is outdated. Most commonly, milligrams of alpha-tocopherol equivalents (alpha-TE) are used to show dosage and this more clearly represents the ability of the body to absorb and utilize different forms of vitamin E (National Academies Press 2000). The conversion factor is based on 0.67mg (alpha-TE)/IU. 1000mg/day (1500IU) is considered the Upper Limit (UL) for vitamin E supplementation based on statistical analysis of adverse effects in high dose studies as well as other factors (National Academies Press 2000). However, research by Johns Hopkins University showed that vitamin E supplementationin excess of 400IU daily for one year in the alpha tocopherol form resulted in increased all-cause mortality and should be avoided (Miller 2005). In terms of therapeutic dosing for maximal cancer prevention and apoptotic potential, there are no standardized protocols. Since many of the studies showing benefit for cancer treatment have been in vitro or in animal models, adult human dosing is difficult to determine. Nesaretnam et al. (2010) found that during a five year randomized controlled trial on women undergoing tamoxifen therapy, risk of mortality due to breast cancer was 60% lower in the intervention group supplemented with 400mg tocotrienols in the form of a tocotrienol rich fraction (TRF) daily versus the controls. This dosing provides a guideline for further investigation. Generally, dietary sources are considered safe due to the bodies inefficient absorption mechanisms for all forms of vitamin E. At higher levels, vitamin E may inhibit absorption of other fat soluble vitamins (A, D, and K). In particular, vitamin K may be decreased in some individuals supplementing with vitamin E and so caution should be taken at higher doses to monitor blood coagulation.
During cancer chemotherapy and radiotherapy, a research review (Lawenda 2008) suggests that supplementing any antioxidant (including vitamin E) should be avoided due to the potential for anti-oxidants to protect any cell regardless of whether it is a cancer cell or not. Most definitely, dosing protocols specific to cancer treatment should be researched further once clinical trials elucidating effectiveness have been undertaken. Common dosages on supplements range from a daily dose of 60mg mixed tocopherols in professional brand multivitamins to 200-400mg daily of tocotrienols in the form of a tocotrienol rich fraction (TRF) from palm oil.
Conclusion
The research that has been reviewed suggests that both gamma tocopherol and tocotrienols have antiinflammatory properties that can reduce cancer cell growth in the body. Tocotrienols may have a greater ability to reduce the risk of cancer development and spread, especially with estrogen receptor positive breast cancer in women. Much more research regarding the specific supplemental dosages of the sup-types of vitamin E is required. Clinical trials are necessary to establish safe and effective dosing protocols. What does seem clear is that gamma tocopherol, tocotrienols, and mixed tocopherols are safer forms of supplementation and provide a higher level of benefit than alpha tocopherol.
References:
Aggarwal, B. B., Sundaram, C., Prasad, S. & Kannappan, R. Tocotrienols, the vitamin E of the 21st century: its potential against cancer and other chronic diseases. Biochemical Pharmacology. 2010. 80(11), 1613-1631.
Dietrich, M., Traber, M. G., Jacques, P. F., Cross, C. E., Hu, Y. & Block, G.Does γ-Tocopherol play a role in the primary prevention of heart disease and cancer? A review. Journal of the American College of Nutrition. 2006. 25(4), 292-299.
Jiang, Q., Elson-Schwab, I., Courtemanche, C. & Ames, B. N. Gamma-tocopherol and its major metabolite, in contrast to alpha-tocopherol, inhibit cyclooxygenase activity in macrophages and epithelial cells. Proceedings of the National Academy of Sciences. 2000. 97(21), 11494-11499.
Jihyeung, Ju, Picinich S, Yang Z, et al. Cancer-preventive activities of tocopherols and tocotrienols. J Carcinogenesis. 2010 April; 31(4): 533–542.
Klein EA, Thompson Jr. IM, Tangen CM, Crowley JJ, Lucia MS, Goodman PJ, et al. Vitamin E and the risk of prostate cancer: the Selenium and Vitamin E Cancer Prevention Trial (SELECT). JAMA 2011;306:1549-1556.
Lawenda, B.D., Kelly, K.M., Ladas, E.J., Sagar, S.M., Vickers, A., Blumberg, J.B. Should Supplemental Antioxidant Administration Be Avoided During Chemotherapy and Radiation Therapy? J Natl Cancer Inst. 2008. 100(11), 773-83.
Miller, E.R.3rd, Pastor-Barriuso, R., Dalal, D., Riemersma, R.A., Appel, L.J., Guallar, E. Meta-analysis: High Dosage Vitamin E Supplementation May Increase All-cause Mortality. Ann Int Med. 2005. 142:37-46.
Moyad, M. A., Brumfield, S. K. & Pienta, K. J. Vitamin E, alpha- and gamma-tocopherol, and prostate cancer. Seminars in Urologic Oncology. 1999. 17(2), 85-90.
National Institutes of Health. Dietary Supplement Fact Sheet: Vitamin E. 2011. Retrievedfrom: http://ods.od.nih.gov/ factsheets/vitamine/.
National Academies Press. Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium, and Carotenoids. Washington, DC, 2000.
Nesaretnam, K., Meganthan, P., Veerasenan, S. D. & Selvadurary, K. R. Tocotrienols and breast cancer: the evidence to date. Genes Nutr. 2012. 7, 3-9.
Nesaretnam, K., Selvaduray, K.R., Razak, G.A., Veerasenan, S.D., Gomez, P.A. Effectiveness of Tocotrienol Rich Fraction Combined with Tamoxifen in the Management of Women with Early Breast Cancer: A Pilot Clinical Trial. Breast Cancer Research. 2010. 12; R81.
Packer, L. Weber, S. U. & Rimbach, G. Molecular aspects of alpha-tocotrienol antioxidant action and cell signaling. Journal of Nutrition.2001. 131(2), 369S-373S.
Schurks, M., Glynn, R.J., Rist, P.M., Tzourio, C., Kurth, T. Effects of Vitamin E on Stroke Subtypes: Meta-analysis of Randomized Controlled Trials. BMJ. 2010. 341.
Stone W, Krishnan K, Campbell S, Qui M, Whaley S, Yang H. Tocopherols and the Treatment of Colon Cancer. Annals of the New York Academy of Sciences Volume 1031, Vitamin E and Health pages 223–233, December 2004
Sylvester, P. W. & Shah, S. J. Mechanisms mediating the antiproliferative and apoptotic effects of vitamin E in mammary cancer cells. Frontiers in Bioscience.2005. 10, 699-709.
Sylvester, P. W., Kaddoumi, A., Nazzal, S. & El Sayed, K. A. The value of tocotrienols in the prevention and treatment of cancer. Journal of the American College of Nutrition. 2010. 29(3), 3245-3335.
Viola, V., Pilolli, F., Piroddi, M., Pierpaoli, E., Orlando, F., Provinciali, M., Betti, M., Mazzini, F. &Galli, F. Why tocotrienols work better: insights into the in vitro anti-cancer mechanism of vitamin E. Genes Nutr. 2012. 7, 29-41.
Wada, S. Cancer preventive effects of vitamin E. Curr Pharm Biotechnol. 2012 Jan;13(1):156-64.
Nigella Sativa
Nigella Sativa
A Panacea for Human Disease
Abstract
Nigella sativa, also known as black cumin, is a highly-regarded and prominent herb that has been used in traditional Middle Eastern and Indian systems of medicine. The Prophet Muhammad was recorded as stating that the herb can cure any disease, other than death. Many therapeutic activities have been ascribed to Nigella, including astringent, bitter, stimulant, anthelminthic, carminative, anodyne, expectorant and other effects. In modern research, Nigella has been shown to possess potent anti-inflammatory activity, lipid, glucose, and blood pressure lowering effects, anti-allergic effects, as well as neuroprotective, antimicrobial, and smooth muscle relaxing effects. Preclinical research has indicated potentially important anticancer effects, though this has yet to be investigated in human studies. These effects have been attributed in large part to the constituent thymoquinone. This long list of therapeutic effects demonstrated by scientific research reinforces the traditional view of this impressive herb.
Introduction
Nigella sativa, also known as black cumin, is part of the Ranunculaceae family and an herb with a long history of traditional use in southeast Asia, Egypt, Greece, and the Middle East (Khan 2011). Nigella was regarded as a panacea by the ancients, and played a central role in the medical practice of Middle Eastern and South Asian societies for over 2000 years, where it was used for the treatment of a range of conditions including asthma, headache, dysentery, infection, back pain, dermatological and gastrointestinal conditions (Isik 2010). In traditional Indian medicine, Nigella seeds are astringent, bitter, stimulant, diuretic, emmenagogue, anthelmintic, galactogogue, carminative, anodyne, sudorific, febrifuge, expectorant, purgative, and abortifacient (Parrakh 2010). The medieval Islamic philosopher Avicenna wrote of Nigella in his Canon of Medicine, that it stimulates the body’s energy and helps recovery from fatigue and dispiritedness (Parrakh 2010). Mohammed is reported to have said that the seed has the power to cure every disease except death (Parrakh 2010).
Medicinally, Nigella has been used in the form of crushed seed, oil, and water extract. The activity of Nigella has been attributed in to its thymoquinone content, which is present in both the fixed and essential oils of Nigella; thymoquinone has been extracted and used therapeutically in isolation (Akhondian 2011, Paarakh 2010).
Nigella oil also contains plant sterols, as well as 25% oleic acid/ 55% linoleic acid, nigellone, and volatile oils including thymol, limonene, and carvacrol (Isik 2010, Paarakh 2010). It is also possible that fibre content may mediate part of the metabolic effects of Nigella.
Mechanism of Action: Preclinical Evidence
There is a wealth of information on Nigella and thymoquinione derived from preclinical research. Thymoquinone appears to be capable of protecting tissues and organs from various types of damage due to antiinflammatory and antioxidant activity, and has also been investigated for its anticancer effects (Woo 2012). In animal models of diabetes, thymoquinone was shown to inhibit cellular expression of the COX-2 enzyme and increased levels of the antioxidant enzyme superoxide dismutase (Al Wafai 2013). In models of hyperlipidemia, Nigella sativa volatile oil and methanol extract have both been shown to reduce triglyceride and increase HDL, as well as decrease hepatic HMG-CoA reductase activity (Ahmad 2013). In smooth muscle tissue, including intestinal and bronchial tissue, thymoquinone has been shown to inhibit spontaneous contraction via inhibition of voltage-gated calcium channels (Ghayur 2012). Thymoquinone has also been shown to inhibit liver fibrosis (Bai 2013); attenuate lung injury and fibrosis (El-Khouly 2012); attenuate diabetic nephropathy (Sayed 2012); prevent E. coli induced tissue damage in bacterial prostatitis (Inci 2013); and prevent dextran sulfate sodium-induced colitis (Lei 2012).
In an animal model of epilepsy, thymoquinone was shown to have antioxidant effects in attenuating malondialdehyde (a marker of lipid peroxidation), reduced neurodegenerative changes and neuron loss, and decreased the number of reactive astrocytes (astrogliosis) (Dariani 2013). Thymoquinone has also been shown to have protective effects against amyloid β-induced neurotoxicity, leading to the suggestion that Nigella may have therapeutic potential in Alzheimer’s disease (Alhebshi 2013).
Nigella and its constituent thymoquinone have also been extensively studied in lab models for its anticancer effects. Thymoquinone has been shown to have pro-apoptotic effects in breast cancer, including synergistically with tamoxifen (Rajput 2013); and has been shown to inhibit cancer cell growth and invasiveness in lung, liver, colon, melanoma, and breast cancer cells (Attoub 2012). This is an area where further research is needed to investigate these promising activities.
Clinical Trials
There is a well-developed body of evidence investigating Nigella in humans. Nigella has been shown to have anti-diabetic, blood pressure and cholesterol lowering effects. It also has activity against specific types of infection, possesses anti-allergic effects, and improves rheumatoid arthritis as well as pediatric seizures. Table 1 summarizes all available evidence from human intervention trials concerning the herb as identified through Pubmed and Google Scholar.
In metabolic syndrome, Nigella seed has been shown to significantly decrease total cholesterol, LDL, and triglyceride (Dehkordi 2008, Sabzghabaee 2012), improve fasting and post-prandial glucose and HbA1C (Bamosa 2010), decrease systolic blood pressure (Datau 2010, Dehkordi 2008) by almost 10 points in one study (Datau 2010), as well as promote reduction of body weight and waist circumference (Datau 2010). In the area of allergy and asthma, Nigella oil or extract has been shown to significantly reduce symptoms of allergic rhinitis (Kalus 2003,Nikakhlagh 2011), as well as reduce the severity and frequency of asthma symptoms, improve lung function tests, and reduce the need for asthma medications (Boskabady 2007). Also related to respiratory function are two studies showing that Nigella can act as an antimicrobial agent in acute pharyngitis (Dirjomuljono 2008), and improve lung function, respiratory symptoms, as well as medication requirements in patients who were victims of chemical warfare (Boskabady 2008). One study found that Nigella seed in combination with omeprazole was equal to triple therapy in the eradication of H. pylori (Salem 2010). Finally, one study each showed that Nigella was effective in reducing disease activity of rheumatoid arthritis (Gheita 2012), and reduce the frequency of seizures in patients with refractory epilepsy (Akhondian 2011).
Dosage
The effective dosage forms and quantities of Nigella used include 2g of ground seeds in metabolic syndrome or syndrome components (Sabzghabaee 2012); 1-4g seed oil for inflammatory conditions such as allergy and arthritis (Gheita 2012, Kalus 2003); 1mg/kg thymoquinone in pediatric seizure (Akhondian 2011); and varying doses of a boiled aqueous extract (Boskabady 2007, Boskabady 2008).
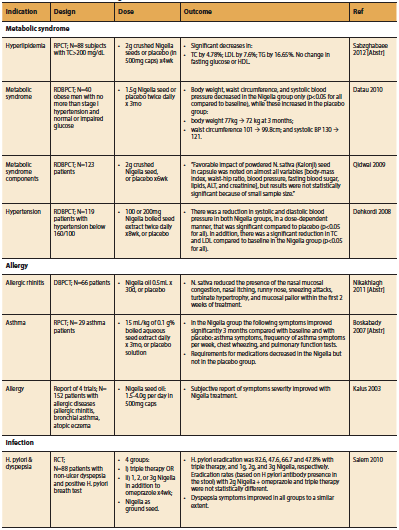
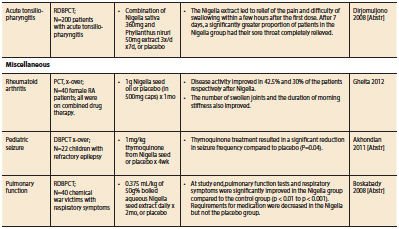
Safety
Based on the studies in Table 1 as well as the impressive range of activities demonstrated in preclinical research, Nigella appears to possess powerful disease modifying potential in a range of conditions. The only adverse events reported with any consistency among the trials were mild gastrointestinal upset (Akhondion 2011, Bamosa 2010, Salem 2010). Overall, Nigella and/ or thymoquinone appears to have a good safety profile (Al-Ali 2008). In animals, toxicity was demonstrated only at dosages of 250 mg/kg thymoquinone by oral route (maximum tolerated dose) (Abukhader 2012). When given by intraperitoneal injection, toxicity occurred at a dose of between 15-20mg/kg thymoquinone, more than 10-fold the dose used in human trials (Abukhader 2012). Signs of toxicity were acute pancreatitis and peritonitis (Abukhader 2012). In another study, the LD50 of thymoquinone by oral administration was 870.9 mg/kg, more than 100 to 150-fold higher than the therapeutic dose (Al-Ali 2008).
Conclusion
Nigella sativa is a highly regarded herb with a long tradition of use for infection, conditions of inflammation, respiratory and gastrointestinal conditions, and more. Human trials have demonstrated benefits from Nigella seed in metabolic conditions such as dyslipidemia, type 2 diabetes, and hypertension; and Nigella oil or extract in allergy and asthma, inflammatory conditions, infections including pharyngitis and H. pylori, as well as refractory epilepsy. Further research is needed to elucidate the potential anticancer effects of Nigella suggested by preclinical findings.
References
Abukhader MM. The effect of route of administration in thymoquinone toxicity in male and female rats. Indian J Pharm Sci. 2012 May;74(3):195-200.
Ahmad S, Beg ZH. Elucidation of mechanisms of actions of thymoquinone-enriched methanolic and volatile oil extracts from Nigella sativa against cardiovascular risk parameters in experimental hyperlipidemia. Lipids Health Dis. 2013 Jun 13;12(1):86. [Epub ahead of print]
Akhondian J, Kianifar H, Raoofziaee M, Moayedpour A, Toosi MB, Khajedaluee M. The effect of thymoquinone on intractable pediatric seizures (pilot study). Epilepsy Res. 2011 Jan;93(1):39-43.
Al-Ali A, Alkhawajah AA, Randhawa MA, Shaikh NA. Oral and intraperitoneal LD50 of thymoquinone, an active principle of Nigella sativa, in mice and rats. J Ayub Med Coll Abbottabad. 2008 Apr-Jun;20(2):25-7.
Alhebshi AH, Gotoh M, Suzuki I. Thymoquinone protects cultured rat primary neurons against amyloid β-induced neurotoxicity. Biochem Biophys Res Commun. 2013 Apr 19;433(4):362-7.
Al Wafai RJ. Nigella sativa and thymoquinone suppress cyclooxygenase-2 and oxidative stress in pancreatic tissue of streptozotocin-induced diabetic rats. Pancreas. 2013 Jul;42(5):841-9.
Attoub S, Sperandio O, Raza H, Arafat K, Al-Salam S, Al Sultan MA, Al Safi M, Takahashi T, Adem A. Thymoquinone as an anticancer agent: evidence from inhibition of cancer cells viability and invasion in vitro and tumor growth in vivo. Fundam Clin Pharmacol. 2012 Jun 11.
Bai T, Lian LH, Wu YL, Wan Y, Nan JX. Thymoquinone attenuates liver fibrosis via PI3K and TLR4 signaling pathways in activated hepatic stellate cells. Int Immunopharmacol. 2013 Feb;15(2):275-81.
Bamosa AO, Kaatabi H, Lebdaa FM, Elq AM, Al-Sultanb A. Effect of Nigella sativa seeds on the glycemic control of patients with type 2 diabetes mellitus. Indian J Physiol Pharmacol. 2010 Oct-Dec;54(4):344-54.
Boskabady MH, Farhadi J. The possible prophylactic effect of Nigella sativa seed aqueous extract on respiratory symptoms and pulmonary function tests on chemical war victims: a randomized, double-blind, placebo-controlled trial. J Altern Complement Med. 2008 Nov;14(9):1137-44.
Boskabady MH, Javan H, Sajady M, Rakhshandeh H. The possible prophylactic effect of Nigella sativa seed extract in asthmatic patients. Fundam ClinP harmacol. 2007 Oct;21(5):559-66. Erratum in: Fundam Clin Pharmacol. 2008 Feb;22(1):105.
Dariani S, Baluchnejadmojarad T, Roghani M. Thymoquinone Attenuates Astrogliosis, Neurodegeneration, Mossy Fiber Sprouting, and Oxidative Stress in a Model of Temporal Lobe Epilepsy. J Mol Neurosci. 2013 Jun 23. [Epub ahead of print]
Datau EA, Wardhana, Surachmanto EE, Pandelaki K, Langi JA, Fias. Efficacy of Nigella sativa on serum free testosterone and metabolic disturbances in central obese male. Acta Med Indones. 2010 Jul;42(3):130-4.
Dehkordi FR, Kamkhah AF. Antihypertensive effect of Nigella sativa seed extract in patients with mild hypertension. Fundam Clin Pharmacol. 2008 Aug;22(4):447-52.
Dirjomuljono M, Kristyono I, Tjandrawinata RR, Nofiarny D. Symptomatic treatment of acute tonsillopharyngitis patients with a combination of Nigella sativa and Phyllanthus niruri extract. Int J Clin Pharmacol Ther. 2008 Jun;46(6):295-306.
El-Khouly D, El-Bakly WM, Awad AS, El-Mesallamy HO, El-Demerdash E. Thymoquinone blocks lung injury and fibrosis by attenuating bleomycin-induced oxidative stress and activation of nuclear factor Kappa-B in rats. Toxicology. 2012 Dec 16;302(2-3):106-13.
Ghayur MN, Gilani AH, Janssen LJ. Intestinal, airway, and cardiovascular relaxant activities of thymoquinone. Evid Based Complement Alternat Med. 2012;2012:305319.
Gheita TA, Kenawy SA. Effectiveness of Nigella sativa oil in the management of rheumatoid arthritis patients: a placebo controlled study. Phytother Res. 2012 Aug;26(8):1246-8.
Inci M, Davarci M, Inci M, Motor S, Yalcinkaya FR, Nacar E, Aydin M, Sefil NK, Zararsiz I. Anti-inflammatory and antioxidant activity of thymoquinone in a rat model of acute bacterial prostatitis. Hum Exp Toxicol. 2013 Apr;32(4):354-61.
Işik H, Cevikbaş A, Gürer US, Kiran B, Uresin Y, Rayaman P, Rayaman E, Gürbüz B, Büyüköztürk S. Potential adjuvant effects of Nigella sativa seeds to improve specific immunotherapy in allergic rhinitis patients. Med Princ Pract. 2010;19(3):206-11.
Kalus U, Pruss A, Bystron J, Jurecka M, Smekalova A, Lichius JJ, Kiesewetter H. Effect of Nigella sativa (black seed) on subjective feeling in patients with allergic diseases. Phytother Res. 2003 Dec;17(10):1209-14.
Khan MA, Chen HC, Tania M, Zhang DZ. Anticancer activities of Nigella sativa (black cumin). Afr J Tradit Complement Altern Med. 2011;8(5 Suppl):226-32.
Lei X, Liu M, Yang Z, Ji M, Guo X, Dong W. Thymoquinone prevents and ameliorates dextran sulfate sodium-induced colitis in mice. Dig Dis Sci. 2012 Sep;57(9):2296-303.
Nikakhlagh S, Rahim F, Aryani FH, Syahpoush A, Brougerdnya MG, Saki N. Herbal treatment of allergic rhinitis: the use of Nigella sativa. Am J Otolaryngol. 2011 Sep-Oct;32(5):402-7.
Paarakh PM. Nigella sativa Linn. – a comprehensive review. Indian Journal of Natural Products and Resources. 2010 Dec;1(4):409-29.
Qidwai W, Hamza HB, Qureshi R, Gilani A. Effectiveness, safety, and tolerability of powdered Nigella sativa (kalonji) seed in capsules on serum lipid levels, blood sugar, blood pressure, and body weight in adults: results of a randomized, doubleblind controlled trial. J Altern Complement Med. 2009 Jun;15(6):639-44.
Rajput S, Kumar BN, Sarkar S, Das S, Azab B, Santhekadur PK, Das SK, Emdad L, Sarkar D, Fisher PB, Mandal M. Targeted apoptotic effects of thymoquinone and tamoxifen on XIAP mediated Akt regulation in breast cancer. PLoS One. 2013 Apr 17;8(4):e61342.
Sabzghabaee AM, Dianatkhah M, Sarrafzadegan N, Asgary S, Ghannadi A. Clinical evaluation of Nigella sativa seeds for the treatment of hyperlipidemia: a randomized, placebo controlled clinical trial. Med Arh. 2012;66(3):198-200.
Salem EM, Yar T, Bamosa AO, Al-Quorain A, Yasawy MI, Alsulaiman RM, Randhawa MA. Comparative study of Nigella Sativa and triple therapy in eradication of Helicobacter Pylori in patients with non-ulcer dyspepsia. Saudi J Gastroenterol. 2010 Jul-Sep;16(3):207-14.
Sayed AA. Thymoquinone and proanthocyanidin attenuation of diabetic nephropathy in rats. Eur Rev Med Pharmacol Sci. 2012 Jun;16(6):808-15.
Woo CC, Kumar AP, Sethi G, Tan KH. Thymoquinone: potential cure for inflammatory disorders and cancer. Biochem Pharmacol. 2012 Feb 15;83(4):443-51.
Effectiveness of Intravenous Vitamin C in Combination with Conventional Chemotherapy in Cancer Treatment
successful completion of the questions at the end of this paper has been approved for continuing education by the bddt-n; 1.0 credit parenteral therapy and by the cnpbc; one ce hour.
Effectiveness of Intravenous Vitamin C in Combination with Conventional Chemotherapy in Cancer Treatment
A Review
James Bao1, Chloe Haldane1, Yvonne Lee2, Brian Li1, Olesya Petrenko1, Christopher Wang1, Wendy Zhou2, Jordan Robertson ND3
¹ Bachelor of Health Sciences (Honours), McMaster University
² Bachelor of Health Sciences (Honours) Candidate, McMaster University
3 Bachelor of Health Sciences, McMaster University, Naturopathic Doctor, Canadian College of Naturopathic Medicine.
Many integrative practitioners are choosing to supplement conventional chemotherapy with intravenous vitamin C (IVC) in cancer patients. Currently, preliminary research has demonstrated the potential benefits of IVC on the length and quality of life in cancer patients. Human- level evidence at this time is limited, yet suggests a role for IV-C as an adjunct treatment for many cancers. There is a strong need for additional research to establish situations the therapy is best suited for, further develop the evidence base of safety in conjunction with conventional therapies, and more clearly delineate the specific outcomes expected from the therapy.
Introduction
The potential benefits of intravenous vitamin C (IVC) in the treatment of cancer have been debated for several decades. The landmark study conducted by Cameron and Pauling in 1976 was the first to highlight the potential use of vitamin C as a form of medication in the treatment of advanced cancer patients. Since then, clinical studies have reaffirmed that cancer patients exhibit abnormally low plasma ascorbic acid (AA) levels secondary to the disease or treatment, and it is suggested that this contributes to a decrease in survival rate (Shah 2009). Currently, IVC research has been extended to include in vitro, in vivo, and human clinical trials on several cancer types.
Preclinical evidence has provided a number of antitumourigenic mechanisms of high dose vitamin C in cancer (Mikirova 2008, Ohno 2009). Since active transporters tightly regulate intestinal absorption of vitamin C, a desirable concentration of AA can only be achieved via intravenous injection, as opposed to oral administration (Padayatty 2004) (see Figure 1). Although results from pre-clinical studies in mice have shown a decrease in tumour growth, human clinical trials, thus far, have not found similar results (Chen 2008, Hoffer 2008, Monti 2012). Rather, these studies show that IVC has limited adverse effects and is a safe and viable treatment to increase quality of life in advanced cancer patients by decreasing the side effects from chemotherapy. Mechanistic studies utilizing cell lines and animal models demonstrate promising results in terms of changing the disease progression itself (Chen 2008). This review hopes to examine any progressions on this theory through evidence provided by recent human clinical trials.
Mechanisms of action based on cell culture studies
Before vitamin C can be investigated as a possible novel cancer treatment, it is essential first to learn whether ascorbate can act as an anticancer agent in cell culture, and if so, by what mechanisms (Chen 2005). Since the 1980s, the involvement of ascorbic acid has been referenced in a number of studies examining cancer cell proliferation specifically in melanoma cells (Bram 1980), oral squamous cell carcinoma and salivary gland tumour cells (Sakagami 2000). In 2005, Chen and colleagues evaluated the use of AA on human cancer cell lines (lymphoma and three types of breast cancer cell types) and mouse cancer cells (lung, kidney, colon and melanoma) through one-hour incubations at pharmacologic concentrations of 0.3-20 mM of vitamin C (Chen 2005).The researchers observed that AA supplementation inhibited growth of breast cancer cells and caused a dose dependent cell death (Chen 2005). Furthermore, the authors showed that normal tissue cells, such as blood cells, are protected from the toxic effects of AA (Chen 2005).
A number of mechanisms have been proposed to understand how vitamin C functions in decreasing the viability of cancer cells. These include suppression of reactive oxygen species (ROS) on mitochondrial DNA, pro-oxidant function of vitamin C, inhibition of the nitric oxide (NO) pathway, degradation of hypoxia inducible factor-1 alpha (HIF-1 alpha), and maintenance of the extracellular matrix (Cha 2013, Du 2012, KC 2005, Mikirova 2008, Verrax 2008). The majority of in vitro and animal studies have focused on high dosage vitamin C acting as a pro-oxidant via the production of hydrogen peroxide (H2O2) (Chen 2008, Du 2012). By acting as an intermediate and readily donating electrons to redox-active transition metals including copper (Cu2+) or iron (Fe3+), AA reduces the transition metals to cuprous (Cu+) and ferrous (Fe2+) ions, respectively (Frei 2008). The reduced transition metals react with oxygen to produce superoxide radicals, which in turn, dismutate to form H2O2 and O2 (Frei 2008). Normal tissues have adequate blood flow which supplies anti-oxidant enzymes such as catalase and glutathione peroxidase to counter-harmful hydroxyl radicals (Ohno 2009). However, tumour cells have reduced blood flow which leads to an accumulation of H2O2 and subsequent apoptosis of these cells (Ohno 2009) (see Figure 2).
ROS can build up within cells due to intracellular oxidative events and can cause fragmentation of mitochondrial DNA (mtDNA) which is associated with a number of cancer types (Chatterjee 2006, KC 2005). AA has also been shown to protect mtDNA by directly reducing radical species and preventing the formation of reactive species, thereby inhibiting the action of ROS on nuclear DNA repair proteins (Padayatty 2003).
Hypoxia-inducible factor-1 (HIF-1) is a transcription factor that upregulates several tumour-growing mRNA transcripts (Verrax 2008). AA is a cofactor of proline hydroxylation and subsequently, the 26S proteasome degradation pathway of HIF-1, which consequently depletes necessary factors for tumour growth, resulting in growth suppression (Knowles 2003, Verrax 2008).
Another way that AA affects tumour cells is by inhibiting NO in endothelial cells (Mikirova 2008). The NO pathway has been known to be an important part of tumour angiogenesis and a high dose of AA has been observed to inhibit the production of NO, thereby decreasing blood vessel formation (Mikirova 2008) and preventing tumour growth (Folkmen 1971).
The breakdown of the extracellular matrix (ECM) has been implicated in the formation of tumours and metastasis and the aggressiveness of cancer growth (Cha 2013). Collagen makes up approximately one third of the ECM and serves as a physical barrier against invasion of cancerous cells (Du 2012, Egeblad 2010). AA is a nutrient required for the synthesis and assembly of collagen, protects against collagen degradation, and prevents proliferation of cancer (Cha 2013).
AA is also known to improve the sensitization of cancer cells to chemotherapy treatments (Heaney, 2008). Seven pathways or mechanisms have been identified to elucidate how AA enhances the effects of cancer therapy drugs. Many of these mechanisms involve the enhancement of membrane transport systems or the activation of tumour suppressor genes (Chiang 1994). As a result, AA reverses chemotherapyresistance and increases the delivery of chemotherapy into cancer cells, increases tumour cell membrane permeability to chemotherapy drugs, stabilizes p53 genes, inhibits translocation of NF-kB and AP1, inhibits Nrf2-mediated gene expression, and activates MLH1, c-Ab1 and p72-signalling cascades (Abdel-Latiff 2005, Bharti 2002, Chiang 1994, Dong 1993, Heaney, 2008, Tarumoto 2004).
In 2001, Reddy and colleagues showed that ascorbic acid stabilizes p53 genes Bax and Bcl2, involved in cancer progression. It has been shown that the absence of p53 leads to abolition of G1 arrest or apoptosis in response to ionizing radiation and DNA damaging agents (Reddy 2001). E6 is an oncoprotein that targets p53 and affect its cell-cycle regulation (Reddy 2001, Scheffner 1990). Reddy and colleagues (2001) showed that vitamin C causes a down-regulation of E6 which in turn causes the up-regulation of pro-apoptotic protein p53 and Bax as well as the down-regulation of apoptotic inhibitor Bcl-2. Accumulation of p53 and Bax then sensitizes the cells to cell-cycle arrest, cell death and apoptosis induced by chemotherapy agents such as cisplatin and etoposide (Reddy 2001).
Another proposed mechanism by which AA provides chemo-sensitization is through inhibition of nuclear factor-kB (NF-kB) and AP-1 (Abdel-Latiff 2005). NF-kB and AP-1 are nuclear transcription factors that have an effect on genes that promote cancer progression (Bharti 2002, Dong 1993). The former suppresses apoptosis and promotes tumourigenesis, while the latter activates genes involved in tumor promotion and progression (Bharti 2002, Dong 1993). Both transcription factors are activated in the presence of chemotherapy agents, thereby causing chemotherapy drug resistance (Abdel-Latiff 2005). Cell cultures studies have shown that AA helps to down-regulate the activity of NF-kB and AP-1, leading to reduced tumour cell proliferation and an improvement in the efficacy of chemotherapy drugs, though the exact mechanism of action of AA on these two factors has yet to be explored (Abdel-Latiff 2005).
Inhibit Nrf2-mediated gene expression
Ascorbic acid also inhibits Nrf2- mediated gene expression which results in greater sensitivity of cancerous cell to chemotherapeutic drugs. Nrf2 is a member of the family of basic regionleucine zipper transcription factors that migrates into the nucleus under conditions of oxidative stress (Tarumoto 2004). In the nucleus, Nrf2 regulates gene expression of a number of detoxifying enzymes such as -glutamylcysteine synthetase ( -GCS), the rate-limiting enzyme in glutathione (GSH) synthesis via the antioxidant response element (ARE)- mediated gene expression (Tarumoto 2004). GSH is involved in resistance to some anti-cancer drugs, including cisplatin and doxorubicin (Iida 1999). Ascorbic acid works to suppress the migration of Nrf2 to the nucleus, thus resulting in inhibition of GSH synthesis and restoration of sensitivity of cancerous cells to chemotherapy (Tarumoto 2004).
Lastly, ascorbic acid has been shown to activate MLH1, and p73 cascades. Mut L homologue-1 (MLH1) is a component of the DNA mismatch repair machinery which allows the removal of mismatches that occur during DNA replication, genetic recombination and DNA damage (Catani 2002). Defects in this gene lead to an accumulation of replication errors and can result in colorectal cancer (Peltomaki 2001). The response to DNA damage involves MLH1 which can activate c-Ab1 tyrosine kinase which then initiates the p53 homologue p73 which improves cellular susceptibility to apoptosis (Catani 2002). This susceptibility is crucial because chemotherapy agents work by causing DNA damage and depend on the cell’s apoptosis as a method of killing the tumour (Catani 2002).
PRE-CLINICAL STUDIES
Mouse Models
The ability of genetically engineered mice to mimic the pathophysiological and molecular features of human malignancies has allowed mouse models to become the most sophisticated animal model for human cancer (Frese 2007). A number of IVC studies have been conducted on mouse models in which decreased tumour growth and tumour volume were observed. However, since most of these studies did not investigate the effects of AA as a cotreatment to chemotherapy, the beneficial effects of AA may be underestimated.
A 1988 study by Tsao et al. reported that AA inhibited the growth of human mammary tumours in immunocompetent mice that were administered AA in drinking water and given a diet with cupric sulfate. AA as a dietary additive alone was ineffective, which supports the theory that the inhibitory effect of AA is due to H2O2 production as Cu2+ is known to catalyze the production of H2O2 in the presence of AA.
Following their in vitro study in 2005, Chen et al. in 2008 applied AA as a treatment option to ovarian, pancreatic and glioblastoma tumour xenografts in mice and found that a high dose of vitamin C (4g/ kg body weight), injected intraperitoneally, decreased tumour growth and weight of all three cancers in the range of 41-53% (Chen 2008). The authors attributed this reduction to H2O2 generation in the extracellular medium of tumour cells by the AA radical (Chen 2008).
Belin et al. (2009) conducted a study in which nude mice were injected with human colon adenocarcinoma cells and the treatment group was treated with high concentrations of AA (1000 mg/kg/day) while the control group received only 15/ mg/kg/day of AA. A decrease in tumour growth was only observed in the treatment group. Furthermore, four out of seven mice in the control group died during the course of the experiment. Thus, it was concluded that a high dose of AA given to cancerous mice will prevent invasive carcinogenic processes (Belin 2009).
Another study by Espey et al. (2011) applied IVC therapy in combination with gemcitabine, a chemotherapeutic agent used to treat pancreatic cancer. After implanting tumour cells into the flanks of mice, the authors divided the mice into four treatment groups: gemcitabine alone, AA alone, combination therapy and saline control (Espey 2011). They found that combination therapy was superior in reducing tumour volume and weight relative to gemcitabine treatment alone (Espey 2011). No adverse effects were recorded except for osmotic stress due to the sodium component of pharmacologic AA administration (Espey 2011).
Recently, Cha et al. (2013) used the collagen pathway to test the efficacy of AA on breast and melanoma cancer cell lines in knockout mice that are unable to synthesize AA. The mice were maintained on or deprived of AA in their diet for four weeks prior to and two weeks after intraperitoneal injections of melanoma and breast cancer cells (Cha 2013). AA supplemented mice showed a 28% tumour weight reduction as compared to the controls (Cha 2013). This provides further evidence that supplemented AA hinders metastasis, tumour growth and encapsulation of tumours (Cha 2013).
HUMAN TRIALS
Disease Progression
There is limited evidence exploring the efficacy of chemotherapy plus IVC in humans. Since 2008, three phase I clinical trials have been conducted, which have demonstrated the safety of IVC alongside chemotherapy (Hoffer 2008, Monti 2012, Welsh 2013). A handful of human trials conducted before 2008 are also reviewed and included in Table 1. First, Hoffer et al. (2008) conducted a dose-seeking trial in patients with advanced tumour malignancy. The study results concluded that a safe dose of 1.5 g/kg of vitamin C can be used for future phase II trials and established that there were few adverse effects in both groups – with or without vitamin C administration (Hoffer 2008). The authors postulated that these adverse effects were related to the rapid infusion of a high osmolarity solution and could have been prevented by increased fluid intake (Hoffer 2008). With regards to the treatment, however, they reported no significant difference in life expectancy and no anti-tumour activity (Hoffer 2008).
The second phase I trial, conducted by Monti et al. (2012), aimed to establish safe levels of IVC administration alongside gemcitabine and erlotinib among patients with stage IV pancreatic cancer. The purpose was to evaluate whether the predicted AA levels could be achieved and to assess any response to the treatment (Monti 2012). The safety data did not reveal any adverse events irregular to the normal progression of pancreatic cancer or use of gemcitabine and erlotinib (Monti 2012).
Most recently, Welsh et al. (2013) aimed to establish the safety and tolerability of pharmacological AA alongside conventional gemcitabine in patients with stage IV pancreatic adenocarcinoma. Using disease burden, weight, performance status, time to progression, hematologic/metabolic tests and overall survival as outcome measures, the data suggested that pharmacologic vitamin C, alongside gemcitabine administration, is well tolerated.
Since these are phase I safety and dose-escalation trials, conclusions relating to the efficacy of vitamin C as a contributor to disease recession cannot be drawn. It should be noted that current phase I studies have only examined pancreatic cancer patients, which may not be representative of all cancer types. It is also important to note the lack of power when considering the conclusions of the aforementioned studies. Currently, the published evidence on the effectiveness of IVC treatment in conjunction with chemotherapy is limited to phase I clinical trials. There are three ongoing clinical trials investigating the safety of IVC therapy (phase I) (clin trials.gov NCT01833351), progressionfree survival (phase II) (clin trials.gov NCT01555489) and toxicity (phase II) (clin trials.gov NCT01754987), to provide better evidence for the role of AA in cancer therapy for the future.
Quality of Life
For patients facing the heavy disease burden of cancer and suffering from the side effects of treatment, improvement in quality of life may be equally as important as finding a cure. Several studies have suggested that IVC therapy may play a role in improving quality of life measures. A prospective study by Yeom et al. (2007) followed 39 terminal cancer patients over a one week period. Patients were asked to stop chemotherapy treatment and were administered 10g of vitamin C intravenously twice over the study period (Yeom 2007). Quality of life was assessed after the study period using a self-administered questionnaire. Patients reported a significant increase in overall quality of life as well as significant improvement in all areas of function (physical, role, emotional, cognitive and social).
Various symptoms were also improved including fatigue, nausea/vomiting, pain, sleep disturbance and appetite loss.
A retrospective cohort study conducted by Vollbracht et al. (2011) examined how IVC affected the quality of life of breast cancer patients. The study gathered 125 patients from different cancer clinics, of which 53 formed the study group and 72 formed the control group (Vollbracht 2011). Patients in the study group received 7.5g of IVC once a week for a minimum of four weeks along with standard antineoplastic interventions (surgical treatment, chemotherapy, radiotherapy or hormone therapy). The control group did not receive adjuvant IVC therapy. The main outcome measures include intensity of complaints due to disease or therapy during treatment and after care. After adjusting for age and baseline scores, the study found statistically significant results supporting the benefits of IVC on ameliorating side effects of treatment such as loss of appetite, fatigue, sleep disorders, and nausea (Vollbracht 2011). This study also reaffirmed the safety of IVC administration in conjunction with conventional cancer treatments.
For patients undergoing chemotherapy or radiation therapy, vitamin C may help to improve quality of life measures by increasing overall anti-oxidative capacity. Since mucosal and nerve cells are particularly susceptible to oxidative stress, increased vitamin C plasma levels may protect against gastrointestinal symptoms such as diarrhea (Vollbracht 2011). Vitamin C may also protect against the degradation of neurotransmitters commonly associated with oxidative stress, possibly explaining its antidepressant-like effects (Vollbracht 2011).
CONCLUSION:
Thus far, current data on the use of IVC as an adjunct therapy to conventional cancer treatments shows promise. While preclinical studies demonstrate the potential for AA to have cytotoxic effects on cancer cells via numerous pathways, hydrogen peroxide being the primary one, recent human trials have not yet proven these effects. However, IVC has been shown to improve the quality of life of cancer patients. Vitamin C’s relatively low-cost, easy accessibility and few treatment side effects further reinforce that its benefits should not be dismissed or overlooked. This review is a call for further research into the efficacy of conventional cancer treatment versus treatment in conjunction with IVC on a larger scale. Such data will help validate IVC as an integral component of standard cancer care and encourage physicians to consider this integrative treatment for their patients.
References
Abdel-Latiff MM, Raouf AA, Sabra K, Kelleher D, Reynolds JV. Vitamin C enhances chemosensitization of esophageal cancer cells in vitro. J Chemother. 2005 Oct;17(5):539-49.
Bahlis NJ, McCafferty-Grad J, Jordan-McMurry I, Neil J, Reis I, Kharfan-Dabaja M, Eckman J, Goodman M, Fernandez HF, Boise LH, Lee KP. Feasibility and correlates of arsenic trioxide combined with ascorbic acid-mediated depletion of intracellular glutathione for the treatment of relapsed/refractory multiple myeloma. Clin Cancer Res. 2002 Dec;8(12):3658-68.
Belin S, Kaya F, Duisit G, Giacomettei S, Ciccolini J, Fonte, M. Antiproliferative effect of ascorbic acid is associated with the inhibition of genes necessary to cell cycle progression. PLoS One. 2009 4(2):e4409.doi: 10.1371/journal.pone.0004409.
Berenson JR, Boccia R, Siegel D, et al. Efficacy and safety of melphalan, arsenic trioxide and ascorbic acid combination therapy in patients with relapsed or refractory multiple myeloma: a prospective, multicentre, phase II, single-arm study. Br J Haematol. 2006 Oct;135(2):174-83.
Berenson JR, Matous J, Swift RA, Mapes R, Morrison B, Yeh HS. A phase I/II study of arsenic trioxide/bortezomib/ ascorbic acid combination therapy for the treatment of relapsed or refractory multiple myeloma. Clin Cancer Res. 2007 Mar;13(6):1762-8.
Bharti AC, Aggarwal BB. Nuclear factor-kappa B and cancer: its role in prevention and therapy. Biochem Pharm 2002 Apr;12;64:883-888.
Block KI, Koch AC, Mead MN, Tothy PK, Newman RA, Gyllenhaal C. Impact of antioxidant supplementation on chemotherapeutic efficacy: a systematic review of the evidence from randomized controlled trials. Cancer Treat Rev. 2007 Sept;33(5):407-18.
Bram S, Froussard P, Guichard M, Jasmin C, Augery Y, Sinoussi- Barre F, & Wray W. Vitamin C preferential toxicity for malignant melanoma cells. Nature. 1980 Apr;17;284(5757):629-31.
Cameron E, Pauling L. (1978). Supplemental ascorbate in the supportive treatment of cancer: reevaluation of prolongation of survival times in terminal human cancer. Proc Natl Acad Sci U S A. 1978 Sep;75(9):4538-42.
Cameron E, Campbell A. Innovation vs. quality control: an ‘unpublishable’ clinical trial of supplemental ascorbate in incurable cancer. Med Hypotheses. 1991 Nov;36(3):185-9.
Campbell A, Jack T, Cameron E. Reticulum cell sarcoma: two complete ‘spontaneous’ regressions, in response to high-dose ascorbic acid therapy. A report on subsequent progress. Oncology. 1991;48(6):495-7.
Catani MV, Costanzo A, Savini I, Levrero M, De Laurenzi V, Wang, JYJ, Melino G, Avigliano L. Ascorbate up-regulates MLH1 (Mut L homologu-1) and p73: implications for the cellular response to DNA damage. Biochem J. 2002. Jun;364(2): 441-7
Cha J, Roomi MW, Ivanov V, Kalinovksy T, Niedzwieky A, Rath M. Ascorbate supplementation inhibits growth and metastasis of B16FO melanoma and 4T1 breast cancer cells in vitamin C-deficient mice. Int J Oncol. 2013 Jan;42(1):55-64. doi: 10.3892/ ijo.2012.1712.
Chatterjee A, Mambo E, Sidransky D. Mitochondrial DNA mutations in human cancer. Oncogene. 2006 Aug 7;25(34):4663-74.
Chen Q, Espey MG, Krishna MC, Mitchell JB, Corpe CP, Buettner GR, Levine M. Pharmacologic ascorbic acid concentrations selectively kill cancer cells: action as a pro-drug to deliver hydrogen peroxide to tissues. Proc Natl Acad Sci U S A. 2005 Sept;102(38):13604-9.
Chen Q, Espey MG, Sun AY, Pooput C, Kirk KL, Krishna MC, Levine M. Pharmacologic doses of ascorbate act as a prooxidant and decrease growth of aggressive tumor xenografts in mice. Proc Natl Acad Sci U S A. 2008 Aug;105(32):11105-9. doi: 10.1073/ pnas.0804226105
Chiang CD, Song EJ, Yang VC, Chao CC. Ascorbic acid increases drug accumulation and reverses vincristine resistance of human non-small-cell lung-cancer cells. Biochemical Journal. 1994 Aug 1;301(Pt 3), 759
Dong Z, Birrer MJ, Watts RG, Matrisian LM, Colburn NH. Blocking of tumor promoter-induced AP-1 activity inhibits induced transformation in JB6 mouse epidermal cells. Proc Natl Acad Sci. 1994 Jan;91:609-613.
Drisko JA, Chapman J, Hunter VJ. The use of antioxidants with first-line chemotherapy in two cases of ovarian cancer. J Am Coll Nutr. 2003 Apr;22(2):118-23.
Du J, Cullen JJ, Buettner GR. Ascorbic acid: Chemistry, biology and the treatment of cancer. Biochim Biophys Acta. 2012 Dec;1826(2):443-57. doi: 10.1016/j.bbcan.2012.06.003.
Egeblad M, Rasch MG, Weaver VM. Dynamic interplay between the collagen scaffold and tumor evolution. Curr Opin Cell Biol. 2010 Oct;22(5):697-706. doi: 10.1016/j.ceb.2010.08.015.
Espey MG, Chen P, Chalmers B, Drisko J, Sun A, Levine M, Chen Q. Pharmacologic ascorbate synergizes with gemcitabine in preclinical models of pancreatic cancer. Free Radic Biol Med. 2011 Jun;50(12):1726-7. doi: 10.1016/j.freeradbiomed.2011.03.030.
Frei B, Lawson S. Vitamin C and cancer revisited. Proc Natl Acad Sci U S A. 2008 Aug;105(32):11037-8 doi: 10.1073/ pnas.0806433105
Folkman J. Tumor angiogenesis: therapeutic implications. New England Journal of Medicine. 1971 Nov:285(21):1182-6. doi: 10.1056/NEJM197111182852108
Frese KK, Tuveson DA. Maximizing mouse cancer models. Nat Rev Cancer. 2007 Sept;7(9): 654–658. doi:10.1038/nrc2192
Heaney ML, Gardner JR, Karasavvas N, Golde DW, Scheinberg DA, Smith EA, O’Connor, OA. Vitamin C antagonizes the cytotoxic effects of antineoplastic drugs. Cancer research. 2008 Jul 25;68(19): 8031-8038.
Hoffer LJ, Levine M, Assouline S, Melnychuk D, Padayatty SJ, Rosadiuk K, Miller WH. Phase I clinical trial of i.v.ascorbic acid in advanced malignancy. Ann Oncol. 2008 Nov;19(11):1969-74. doi: 10.1093/annonc/mdn377
Iida T, Mori E, Mori K, Goto S, Urata Y, Oka M, Kohno S, Kondo T. Co-expression of gamma-glutamycysteine synthetase subunits in response to cisplatin and doxorubicin in human cacner cells. Int J Cancer. 1999 Jul; 82(3): 405-11
KC S, Carcamo JM, Golde DW. Vitamin C enters mitochondria via facilitative glucose transporter 1 (GLUT1) and confers mitochondrial protection against oxidative injury. FASEB J. 2005 Oct;19(12):1657-67.
Knowles HJ, Raval RR, Harris AL, Ratcliffe PJ. Effect of ascorbate on the activity of hypoxia-inducible factor in cancer cells. Cancer Res. 2003;63:1764–1768.
Lemmo W. Enhancing chemotherapy effectiveness with simple vitamin C. Canadian Naturopathic Foundation. [Data file] Retried from http://www.exploreyourhealth.ca/viewpage. cfm?PageID=15
Mikirova NA, Ichim TE, Riordan NH. Anti-angiogenic effect of high doses of ascorbic acid. J Transl Med. 2008 Sep;6:50. doi: 10.1186/1479-5876-6-50.
Mikirova N, Casciari J, Rogers A, Taylor P. Effect of high-dose intravenous vitamin C on inflammation in cancer patients. J Transl Med. 2012 Sep;10:189. doi: 10.1186/1479-5876-10-189.
Monti DA, Mitchell E, Bazzan AJ, Littman S, Zabrecky G, Yeo CJ, Pillai MV, Newberg AB, Deshmukh S, Levine M. Phase I evaluation of intravenous ascorbic acid in combination with gemcitabine and erlotinib in patients with metastatic pancreatic cancer. PLoS One. 2012;7(1):e29794.
doi: 10.1371/journal. pone.0029794. Office of Dietary Supplements. Dietary Supplement Fact Sheet: Vitamin C. [Data file] Retrieved from http://ods.od.nih.gov/ factsheets/VitaminC-HealthProfessional/
Ohno S, Ohno Y, Suzuki N, Soma G, Inoue M. High-dose Vitamin C (Ascorbic Acid) Therapy in the treatment of patients with advanced cancer. Anticancer Res. 2009 Mar;29(3):809-15.
Padayatty SJ, Eck P, Kwon O, Lee J, Chen S, Corpe C, Dutta A, Dutta SK, Levine M. Vitamin C as an Antioxidant: Evaluation of Its role in Disease Prevention. J Am Coll Nutr. 2003 Feb;22(1):18- 35.
Padayatty SJ, Riordan HD, Hewitt SM, Katz A, Hoffer LJ, Lavine M. Intravenously administered vitamin C as cancer therapy: three cases. CMAJ. 2006 Mar;174(7):937-42.
Padayatty SJ, Sun H, Wang Y, Riordan HD, Hewitt SM, Katz A, Wesley RA, Levine M. Vitamin C pharmacokinetics: Implications for oral and intravenous use. Ann Intern Med. 2004 Apr;140(7):533-7.
Peltomaki P. Deficient DNA mismatch repair: a common etiologic factor for colon cancer. Hum Mol Genet. 2001 Apr; 10(7): 735-40
Reddy VG, Khanna N, Singh, N. Vitamin C augments chemotherapeutic response of cervical carcinoma HeLa cells by stabilizing p53. Biochem Biophys Res Commun. 2001 Mar;282(2): 409-15
Riordan HD, Casciari JJ, González MJ, Riordan NH, Miranda- Massari JR, Taylor P, Jackson JA.A pilot clinical study of continuous intravenous ascorbate in terminal cancer patients. P R Health Sci J. 2005 Dec;24(4):269-76.
Sakagami H, Satoh K, Hakeda Y, Kumegawa M. Apoptosisinducing activity of vitamin C and vitamin K. Cell Mol Biol (Noisy-le-grand). 2000 Feb;46(1):129-43.
Scheffner M, Werness BA, Huibregtse JM, Levine AJ, Howley PM. The E6 oncoprotein encoded by human papillomavirus types 16 and 18 promotes the degradation of p53. Cell. 1990 Dec; 63(6):1129-36
Shah FD, Patel JB, Shukla SN, Shah PM, Patel PS. Evaluation of plasma non-enzymatic antioxidants in breast cancer etiology. Asian Pac J Cancer Prev. 2009 Jan-Mar;10(1):91-6.
Tarumoto T, Nagai T, Ohmine K, Miyoshi T, Nakamura M, Kondo T, Mitsugi K, Nakano S, Muroi K, Komatsu N, Ozawa K. Ascorbic acid restores sensitivity to imatinib via suppression of Nrf2-dependent gene expression in the imatinib-resistant cell line. Exp Hematol. 2004 Apr; 32(4); 375-81
Tsao SC, Dunham WB, Leung PY. In vivo antienoplastic activity of ascorbic acid for human mammary tumor. In Vivo. 1988 Mar- Apr;2(2):147-50.
Verrax J, Calderon PB. The controversial place of vitamin C in cancer treatment. Biochem Pharmacol. 2008 Dec;76(12):1644-52. doi: 10.1016/j.bcp.2008.09.024
Vollbracht C, Schneider B, Leendert V, Weiss G, Auerbach L, Beuth J. Intravenous vitamin C administration improves quality of life in breast cancer patients during chemo-/radiotherapy and aftercare: results of a retrospective, multicentre, epidemiological cohort study in Germany. In Vivo. 2011 Nov-Dec;25(6):983-90.
Welsh JL, Wagner BA, van’t Erve TJ, Zehr PS, Berg DJ, Halfdanarson TR, Yee NS, Bodeker KL, Du J, Roberts LJ 2nd, Drisko J, Levine M, Buettner GR, Cullen JJ. Pharmacological ascorbate with gemcitabine for the control of metastatic and nodepositive pancreatic cancer (PACMAN): results from a phase I clinical trial. Cancer Chemother Pharmacol. 2013 Mar;71(3):765- 75. doi: 10.1007/s00280-013-2070-8.
Wu KL, Beksac M, van Droogenbroeck J, Amadori S, Zweegman S, Sonneveld P.Phase II multicenter study of arsenic trioxide, ascorbic acid and dexamethasone in patients with relapsed or refractory multiple myeloma. Haematologica. 2006 Dec;91(12):1722-3.
Yeom CH, Jung GC, Song KJ. Changes of terminal cancer patients’ health-related quality of life after high dose vitamin C administration. J Korean Med Sci. 2007 Feb;22(1):7-11.
Harmony Health Centre
We at IHP are pleased to introduce the Harmony Health Centre, a beautiful multi-disciplinary clinic providing high-quality alternative healthcare options. Harmony Health Centre is located in Westmount, Quebec, where Naturopathic Doctors are not currently regulated. As a result, they have faced unique challenges and have developed creative solutions to work with their community. The clinic is run by two experienced Naturopathic Doctors, Ilana Block, ND and Orna Villazan, ND. Dr. Block and Dr. Villazan met in 2005 and together founded Harmony Health Centre with the dream of providing excellent patient care. They have a general family practice, and they see a lot of gastrointestinal concerns, weight concerns, women’s health and fertility, and mental health concerns. These talented doctors are now managing one of the few integrative clinics in Quebec!
The clinic has a storefront location and the zen atmosphere helps to relieve stress from the moment patients walk in. The facility has four beautiful treatment rooms and each ND sees over 20 patient visits per week. All of the clinic operations were developed with the patient experience as the top priority, where patients can come in early, have a tea, read, relax, and if desired, see a couple of practitioners on the same day. The clinic embodies a holistic healthpromoting environment. The majority of the practitioners at the clinic are fluent in both French and English. The clinic has extended hours in evenings and on weekends (they are open seven days a week) and they are fully accessible by public transit. The marketing strategies they use include a strong online presence through their website and Facebook, and local information sessions they host for the public.
Dr. Block and Dr. Villazan were trained and are licensed in the province of Ontario (as out of province practitioners). In Quebec, there are no regulations for naturopathic medicine and without regulation, the scope of practice of Naturopathic Doctors can be quite limited. Naturopathic Medicine is currently very confusing for the average person seeking care, which makes educating the public challenging, but imperative. In Quebec, there are numerous challenges that NDs in regulated provinces do not face. From our understanding, there are no laws overseeing the practice of naturopathic medicine, including a description or regulation for what NDs are able to do. As a result, NDs, like other unregulated practitioners, are faced with the concept that they can’t practice what is considered medicine without a license. If you are not licensed as a medical doctor, you are limited in your practice of medicine. For example, the NDs do not have the authority to make a diagnosis, to perform venipuncture or intramuscular injections, to perform gynecological exams, or to perform acupuncture. As a result, the services and care that these doctors are able to provide has to be of utmost quality to balance out the regulatory limitations. They work together as a team, provide holistic care, and inter-refer to other health care practitioners.
Another limitation these NDs face is that there are approximately 5500 individuals practicing as natural therapists in Quebec. There are currently 15 Naturopathic Doctors in the province who are licensed from a regulated jurisdiction. Insurance companies do not make a distinction between licensed NDs and those who have done a few weekend courses. Since 1992, the QANM (Quebec Association of Naturopathic Medicine) has been working on behalf of the profession, attempting to get a law in place that would protect and recognize a full scope of practice for the ND as well as protecting the public. Without over-stepping their bounds or stepping on anyone’s toes, Dr. Block and Dr. Villazan have developed functioning relationships with many medical doctors and other health practitioners. This has helped them overcome some of the regulatory limitations by utilizing these other practitioners as part of their referral base, thus allowing their patients to access the care they need.
Most of the patients at the clinic come from word-of-mouth referrals. Patients see any practitioner that they originate contact with and then usually get referred appropriately to other practitioners. The other opportunity that patients have to meet other practitioners come from group workshops that are conducted at the clinic, including workshops in women’s care or a versatile session called “Conversations on Health,” where multiple practitioners discuss pain management, stress relief, depression, anxiety, and other conditions. These sessions are free to the public. The clinic also sees many referrals from outside the clinic, including from integrative medical doctors, personal trainers, and others such as psychologists.
Harmony Health Centre carries numerous reputable brands in their dispensary, including AOR, Metagenics, Seroyal, NFH, NaturePharm, St-Francis Herb Farm, and Vitazan. They carry tinctures from Mediherb and St-Francis and homeopathics from UNDA, Boiron, Pascoe, and Heel. They utilize labs from Gamma-Dynacare, Genova, Metametrics, Doctor’s Data, and Rocky Mountain Analytical. Based on their patient base, the most commonly run labs are food sensitivity tests, parasitology or digestive analysis, and heavy metal testing.
When asked what messages they would like to provide for other doctors, Dr. Block says: “Come to Quebec! The more NDs there are, the better it will be. Despite the challenges, there is an exciting shift occurring in integrative medicine in the province.” Harmony Health Centre is a dynamic work environment and they are always looking to add new members to their team. Both Dr. Block and Dr. Villazan believe there is a strong need to increase the number of NDs in Quebec to better offer quality health care options to Quebecers as well as to obtain more visibility and public awareness.