This review evaluated the role of dietary components in pancreatic cancer. Pancreatic cancer has a poor prognosis with a 5-year survival rate of less than 5%. The authors performed searched in PubMed using the terms “pancreatic cancer”, together with “nutrition”, “diet”, “dietary factors”, “lifestyle”, “smoking”, “alcohol” and “epidemiology”. The results showed that fruits, particularly citrus, and vegetable consumption may be beneficial. The consumption of whole grains has been shown to reduce pancreatic cancer risk and fortification of whole grains with folate may confer further protection. Red meat, cooked at high temperatures, should be avoided, and replaced with poultry or fish. Total fat should be reduced. The use of curcumin and other flavonoids should be encouraged in the diet. There is no evidence for benefit from vitamin D supplementation. There may be benefit for dietary folate. Smoking and high Body Mass Index have both been inversely associated with pancreatic cancer risk. The authors conclude that there is evidence to encourage the use of whole grain in the staple diet and supplementation within the diet of folate, curcumin and other flavanoids. Carefully designed randomized trials are required to further elucidate these important matters. Anticancer Res. January 2014. PMID: 24403441.
EGFR mutations in Asian patients with non-smallcell lung cancer
Known as the PIONEER trial, this study was a prospective, multinational, epidemiological study of epidermal growth factor receptor (EGFR) mutations in patients from Asia with newly diagnosed advanced lung adenocarcinoma. Eligible patients had untreated stage IIIB/IV adenocarcinoma. EGFR mutation status of tumor samples was determined. The results showed that of 1482 patients from seven Asian regions, 43.4% of patients were female, median age was 60 years (range, 17-94), and 52.6% of patients were never-smokers. EGFR mutation status was evaluable in tumors from 1450 patients (97.8%) (746 [51.4%] positive; 704 [48.6%] negative). Country, sex, ethnicity, smoking status, pack-years (all p < 0.001), disease stage (p = 0.009), and histology type (p = 0.016) correlated significantly with EGFR mutation frequency. Mutation frequency was 61.1% in females, 44.0% in males; lower in patients from India (22.2%) compared with other areas (47.2%- 64.2%); highest among neversmokers (60.7%); and decreased as pack-year number increased (>0-10 pack-years, 57.9%; >50 pack-years, 31.4%) (similar trend by sex). Ethnic group (p < 0.001) and packyears (p < 0.001) had statistically significant associations with mutation frequency (multivariate analysis); sex was not significant when adjusted for smoking status. The authors conclude that there is high EGFR mutation frequency (51.4% overall) in tumors from Asian patients with adenocarcinoma. J Thorac Oncol. February 2014. PMID: 24419411.
Inflammationassociated pigmentation in psoriasis mediated by IL-17 and TNF
The etiology of the underlying inflammation-associated pigmentation changes in psoriasis was studied in this article. Previously, it was unclear how the myriad of cytokines known to be involved in inflammatory skin processes affect epidermal melanocytes. The authors sought to determine how IL-17 and tumor necrosis factor (TNF) influence normal human melanocytes, as these two cytokines have been implicated in various skin diseases. IL-17 and TNF jointly stimulated broad inductions of cytokines, including melanoma mitogens CXCL1 and IL-8. Moreover IL-17 and TNF synergistically inhibited pigmentation-related signaling and melanin production, and induced keratinocyte production of β-defensin 3, an antagonist for melanocortin 1 receptor. When analyzing psoriasis lesions that are known to overexpress IL-17 and TNF, the authors observed an increase in melanocyte number and a simultaneous decrease in pigmentation signaling. Furthermore, therapeutic neutralization of TNF and IL-17 with mAbs resulted in a rapid recovery of pigment gene expression in psoriasis lesions. They conclude that IL-17 and TNF can affect both the growth and pigment production of melanocytes, which may contribute to the pigmentation changes associated with psoriasis. These findings may allow the development of novel therapeutics for pigmentary disorders and bring new insights into the immune milieu surrounding melanocytes and related neoplasms. J Invest Dermatol. December 2013. PMID: 23732752.
The Case for Blood Donation
The Case for Blood Donation
Why Blood Donation is a Larger Health Issue than Generally Appreciated
Abstract
Blood donation is probably more often motivated by altruism than by the notion that it carries health benefits. Iron is involved in many biological processes. (Valium) It is also well known to be a source of reactive oxygen species (ROS) trough the Fenton reaction, and the result can be oxidative stress with cellular, DNA, vascular and organ damage. The currently used laboratory reference range for normal serum ferritin typically covers from the 5th to the 80th or 90th population percentile and is gender dependent. However, there is considerable evidence that within this range adverse effects of iron are implicated, which impact the development and progression of a number of common disorders. There is also considerable data indicating that lowering ferritin levels within the normal range to values corresponding to near iron depletion produces beneficial results for a number of diseases. In addition, oxidative DNA damage is strongly and significantly correlated with ferritin levels within the normal reference range with no apparent threshold. There appears to be no consensus as to what constitutes an optimum level of iron body stores. However, there is a large range of these stores between anemia and abnormal levels that suggest intervention. It is suggested that optimum ferritin levels may be at the low end of the normal reference range near the threshold for anemia. The simplest and quickest way to dramatically lower ferritin levels is blood donation.
Introduction
In the September 2013 issue of Integrated Healthcare Practitioners, the potential toxicity of iron and the connection between iron and diabetes was explored (Ware 2013). While not emphasized, elevated levels of serum ferritin, a measure of iron body stores and active iron, are associated with enhanced risk for a number of disorders, and the levels from which lowering produced clinical benefits were well below the upper limit of the normal reference ranges used internationally. In what follows, this observation will be generalized to a number of disorders and the question, “what is the optimum serum ferritin level?” will be addressed.
Ferritin reference ranges and levels regarded as normal
There is some variation in the upper reference limits for serum ferritin that constitute a threshold for concern. In North America, typical values for the upper limit of normal are 300-320 ng/mL for men, and 150-307ng/ mL for women. Similar values are common in other regions. In a recent discussion of the diagnosis of hyperferritinemia, the authors indicate elevated ferritin levels are >300 ng/ mL for men and >200 ng/mL for women.
Hemochromatosis is not generally treated until ferritin levels are considerably above these upper limits of normal (Adams 2011).
The variation of serum ferritin levels with age and gender in the US can be obtained from the Third National Health and Nutrition Examination Survey (NHANES III) (Zacharski 2000). For Caucasian men, the mean serum ferritin at age 17-19 is about 60 ng/mL and by age 30-39 has a plateau at about 150 ng/mL where it remains until about age 60 when a steady decline to about 90 ng/mL by age 90 is observed. For women, the mean value is quite constant at around 30 ng/mL until after menopause and then increases to about 80 ng/mL by age 60 and then gradually increases to about 100 ng/mL at age 80-90.
The multiplicity of physiologic processes involving ferritin, and in particular its role as an acute phase reactant, has caused some to question the use of serum ferritin as a marker for the risk of various disorders (Adams 2012, Lee 2004). However, as will be discussed, risk of incidence of various disorders correlates with ferritin levels in a large number of studies with large sample size and a diversity of disorders. More importantly, there are also a number of disorders where lowering “normal” ferritin by blood removal and driving ferritin levels from above, near or even below the population mean to near iron depletion (threshold for anemia) produces significant improvements in clinical manifestations or markers.
Thresholds of ferritin levels and risk of disease
In addition to the associations between ferritin levels and diabetes discussed in the cited IHP article, the following are of interest.
• In a study of men and postmenopausal women, a ferritin threshold of >200 ng/mL was associated with an increase in risk of a first heart attack (Holay 2012).
• A study classified CHD-positive patients as having one or more coronary arteries with ≥ 50% blockage. Comparison of ferritin levels revealed that those CHD-positive had on average ferritin levels of 121 vs. 73 ng/mL for those CHD-negative (Haidari 2001).
• At a ferritin threshold of >137 ng/mL, increased risk of ischemic stroke was found in a study of postmenopausal women (van der A 2005).
• A ferritin threshold of 145 to 164 ng/mL was found for increased risk of acute ischemic stroke transforming to a hemorrhagic stroke in older men and women (Choi 2012).
• A large study examined the association between ferritin levels and laboratory measured cardiovascular fitness (CVF) in young men. The likelihood of the absence of CVF, adjusted for numerous potential confounders, became significantly apparent at a ferritin threshold of >150 (Mainous 2009).
• Middle-aged men had significant risk of developing hypertension, defined as BP ≥ 140/90 mm Hg, at a ferritin threshold of > 146 ng/mL (Kim 2012).
• A study of the relationship between increased ferritin, oxidative stress and insulin resistance in 151 healthy men revealed no threshold, only continuous increases in markers with ferritin levels from the first tertile (≤97 ng/mL) to the third (≥180 ng/mL). Notably, the correlations remained strong and significant after adjustment for inflammation (Syrovatka 2009).
• In a 5-year study the association between ferritin levels and the incidence and progression of carotid atherosclerosis was examined among a cohort of 826 men and women age 40 to 79 years. Serum ferritin was found to be the strongest risk factor of overall progression, and changes in ferritin levels during follow-up modified the risk with lower levels beneficial. The risk of incident carotid atherosclerosis increased linearly and doubled from quintile I (0-36 ng/mL) to quintile IV (130-222 ng/mL). When subjects with < 50 ng/mL of ferritin were compared with those ≥ 50 ng/mL, the increase in incidence of carotid atherosclerosis for elevated ferritin per 100 person years was approximately 0.3 vs. 4.8, 0.2 vs. 4.5 and 1.6 vs. 4.4 for men, premenopausal women and naturally menopausal women, respectively (Kiechl 1997).
• In a study of over 12,000 men, the ferritin threshold for increased risk of any coronary artery calcium was >257 ng/mL (Sung 2012).
• In a study of men from either northern Europe (Zutphen) or the Mediterranean south (Crete), mean ferritin levels were 134 ng/mL and 70 ng/mL, respectively. Those from Crete (70 ng/mL) had consistently lower levels of indicators of oxidative stress, higher antioxidant capacity and higher concentrations of major antioxidants than men from Zutphen (Buijsse 2007).
Ferritin thresholds and benefit in iron-lowering studies
The following studies are particularly important since they appear less subject to confounding than those cited above.
• In a randomized prospective trial, iron reduction in male smokers with peripheral arterial disease (PAD) reduced the risk of death or nonfatal heart attack, such that the number needed to treat with phlebotomy over five years to prevent one acute event was only eight, a very low number and uncommon in clinical studies. Significant benefits were also seen for all cause mortality, non-fatal MI and stroke. The initial and final mean ferritin levels were 125 ng/mL and 84 ng/ mL. In a larger study in which the above was imbedded, unequivocal benefits for all cause mortality, non-fatal MI and stroke were found for iron reduction in younger individuals 43- 61 years of age including non-smokers, when phlebotomy reduced ferritin levels to < 70 ng/ mL (Depalma 2012, Depalma 2013).
• A controlled trial involving phlebotomy which decreased ferritin levels from a mean of 188 to 105 ng/mL for a group of men and women with the metabolic syndrome, found a decrease in systolic blood pressure from 149 to 131 mm Hg, with no change in the control group. Fasting blood glucose, HbA1c and heart rate were also significantly decreased (Houschyar 2012).
• Use of the oral prescription chelator deferiprone over nine months in patients with non-diabetic kidney disease reduced ferritin from 144 to 59 ng/mL and resulted in significant clinical improvements (Rajapurkar 2013).
• In a trial using phlebotomy in patients with peripheral artery disease, a reduction in mean ferritin levels from 122 to 74 ng/mL resulted in a significant reduction in the incidence of visceral malignancy (Zacharski 2008).
Comparison of both the above thresholds for risk and the baseline ferritin levels from which lowering produced benefit reveals an incompatibility with the commonly used reference range values regarded as normal and of no concern.
Iron stores reductions and non-alcoholic fatty liver disease
Non-alcoholic fatty liver disease (NAFLD) starts with simple hepatic steatosis and can progress to non-alcoholic steatohepatitis (NASH). One hypothesis for the pathogenesis of this disorder is the involvement of iron in increased oxidative stress and liver inflammation (Nelson 2012). High ferritin levels have been found independently associated with advanced hepatic fibrosis (Kowdley 2012). The following iron depletion studies are thus of interest.
• A study of phlebotomy on an insulin resistant group of patients with NAFLD and markedly
elevated ferritin levels showed a significant reduction in insulin resistance (HOMA-IR decreased from 4.81 to 3.12) when ferritin levels were reduced from 438 to 52 ng/mL. More importantly, alanine transaminase (ALT) decreased from a mean of 58.1 to near normal 34.3 IU/L (Valenti 2007).
• A study examined the effect of phlebotomy in either carbohydrate intolerant subjects or type 2 diabetics, including some also diagnosed with NAFLD based on elevated ALT and ultrasound evidence of steatosis (Facchini 2002). The NAFLD and non- NAFLD groups had baseline mean ferritin levels of 299 and 220 ng/mL, respectively. Phlebotomy produced near iron depletion (ferritin 31-15 ng/mL) and ALT fell from 61 to 32 IU/L in the NAFLD group. There were insignificant ALT changes observed in the NAFLD-free group.
• In a study where ferritin levels were manipulated with diet, 12 patients with NASH were placed on a calorie, fat and iron restricted diet. Baseline mean ferritin levels were 280 ng/mL initially and 128 ng/mL at 6 months of intervention. ALT levels decreased from 104 to 42 IU/L over the same period. Large changes were also seen in aspartate aminotransferase (AST) levels (Yamamoto 2007).
Iron and oxidative damage
Urinary 8-hydroxydeoxyguanosine (8-OHdG) is a reliable and frequently used biomarker of systemic oxidative DNA damage (Nakano 2003, Valko 2006). The following studies are of particular interest because of the apparent absence of a threshold for adverse effects of iron.
For over 500 healthy Japanese aged 21-67, linear correlations between 8-OHdG and ferritin were found, with Spearmen rank correlation coefficients of 0.47, 0.76 and 0.73 for men overall, women aged less than 50, and women 50 years or older, respectively. These strong correlations were essentially unchanged after adjustment for potential confounders. Subjects exhibited a large range of ferritin levels from near iron depletion to around 300 ng/mL for men and 100 ng/mL for women (Hori 2010).
For a cohort of 2500 healthy individuals aged 22 to 89 years, a smooth, almost linear 2.5 fold increase in 8-OHdG was found for men as ferritin ranged from 10 ng/mL to about 300 ng/mL. For women, 8-OHdG was increased by a factor of 3 for ferritin levels ranging from below 9 to 160 ng/mL (Nakano 2003). These results suggest no threshold and this observation is consistent with a study of vascular function and ferritin levels (Zheng 2005), and a study of oxidative stress and insulin resistance (Syrovatka 2009).
In addition, a study of ferritin lowering with phlebotomy was found to reduce 8-OHdG in patients with chronic hepatitis C (Kato, 2001). Another study found a beneficial impact of reducing iron stores to near depletion on the development of hepatocellular carcinoma (HCC) from chronic hepatitis C (Kato 2007). In both studies, final ferritin levels were about 10 ng/mL.
Influence of blood donation on ferritin levels
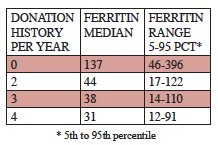
Blood donation typically removes 450-500 mL per visit. The following results from a large sample of Danish men 30 to 66 years of age illustrate the dramatic effect of blood donation (Milman 1996).
Danish study of the influence of blood donation of serum ferritin levels (ng/mL) in men, 30-66 years of age (Milman 1996).
These data are consistent with a recent American study (Cable 2011). Thus on average three to four donations per year will result in a ferritin level mostly below 100 ng/mL with a median representing near iron depletion.
Risks associated with blood donation
Blood donation is not totally free of adverse events. These include syncope, bruises and haematomas and sensory changes. Syncope is rare even among those over 40 (typically 0.05% incidence). Others, while frequently of higher incidence, are almost always transient and rarely of significance. The incidence of permanent injury has been cited as about 0.002% (Amrein 2012, Gavillet 2013, Newman 2012).
Of greater concern is the impact of frequent donations on iron status. Deferral at blood donor clinics is generally based on a finger stick haemoglobin assay with a commonly used threshold for deferral of < 12.5 g/dL, a threshold that has been questioned (Cable 2011). The common lower limit for normal is 13.5 g/dL, and served as the older cut-off for deferral. A 12- month multicenter donor clinic follow-up study examined the iron status in blood donors screened by finger stick. The threshold for diagnosing unacceptable iron stores was a ferritin level < 12 ng/mL. This was found in 15% of those passing the haemoglobin screening. The finger stick haemoglobin test is evidently not excluding some individuals with low iron status and thereby increasing the risk of an adverse impact of donation. For frequent blood donors, an annual ferritin test and if appropriate, a follow-up test for iron deficient erythropoiesis are indicated (Cable 2011).
Conclusions
It is clear from the above studies that the serum ferritin thresholds for the appearance of risk and the baseline values from which lowering produces benefit are predominantly well below the upper reference range values for normal, and in fact more closely correspond to population 50th percentiles. Indeed, lowering ferritin to levels far below the 50th population percentile produces significant benefit associated with severity of a variety of disorders. In some studies, this is observed even when the baseline level for lowering is already quite low. The DNA oxidative stress studies strongly support the view that the optimum ferritin level is one representing near iron depletion, which can only be easily achieved by blood donation or phlebotomy. This conclusion challenges a widely held belief that ferritin anywhere in the normal reference range should not cause concern.
References
Adams,P.C. Diabetes: Serum ferritin levels and T2DM–are body iron stores elevated? Nat Rev Endocrinol 2012; 8(10): 573-575.
Adams,P.C. and Barton,J.C. A diagnostic approach to hyperferritinemia with a non-elevated transferrin saturation. J Hepatol. 2011; 55(2): 453-458.
Amrein,K., Valentin,A., Lanzer,G. and Drexler,C. Adverse events and safety issues in blood donation–a comprehensive review. Blood Rev 2012; 26(1): 33-42.
Buijsse,B., Feskens,E.J., Moschandreas,J., Jansen,E.H., Jacobs,D.R., Jr., Kafatos,A., Kok,F.J. and Kromhout,D. Oxidative stress, and iron and antioxidant status in elderly men: differences between the Mediterranean south (Crete) and northern Europe (Zutphen). Eur J Cardiovasc Prev Rehabil. 2007; 14(4): 495-500.
Cable,R.G., Glynn,S.A., Kiss,J.E., Mast,A.E., Steele,W.R., Murphy,E.L., Wright,D.J., Sacher,R.A., Gottschall,J.L., Vij,V. and Simon,T.L. Iron deficiency in blood donors: analysis of enrollment data from the REDS-II Donor Iron Status Evaluation (RISE) study. Transfusion 2011; 51(3): 511-522.
Choi,K.H., Park,M.S., Kim,J.T., Nam,T.S., Choi,S.M., Kim,B.C., Kim,M.K. and Cho,K.H. The serum ferritin level is an important predictor of hemorrhagic transformation in acute ischaemic stroke. Eur J Neurol 2012; 19(4): 570-577.
Depalma,R.G. and Zacharski,L.R. Iron reduction benefits: positive results from a “negative” prospective randomized controlled trial. Vasc Endovascular. Surg 2012; 46(7): 596-597.
Depalma,R.G., Zacharski,L.R., Chow,B.K., Shamayeva,G. and Hayes,V.W. Reduction of iron stores and clinical outcomes in peripheral arterial disease: outcome comparisons in smokers and non-smokers. Vascular 2013.
Facchini,F.S., Hua,N.W. and Stoohs,R.A. Effect of iron depletion in carbohydrate-intolerant patients with clinical evidence of nonalcoholic fatty liver disease. Gastroenterology 2002; 122(4): 931-939.
Gavillet,M., Tissot,J.D. and Canellini,G. Blood donation associated risks: data from a Swiss regional haemovigilance program. Transfus. Med 2013; 23(4): 269-271.
Haidari,M., Javadi,E., Sanati,A., Hajilooi,M. and Ghanbili,J. Association of increased ferritin with premature coronary stenosis in men. Clin Chem 2001; 47(9): 1666-1672.
Holay,M.P., Choudhary,A.A. and Suryawanshi,S.D. Serum ferritin-a novel risk factor in acute myocardial infarction. Indian Heart J 2012; 64(2): 173-177.
Hori,A., Mizoue,T., Kasai,H., Kawai,K., Matsushita,Y., Nanri,A., Sato,M. and Ohta,M. Body iron store as a predictor of oxidative DNA damage in healthy men and women. Cancer Sci 2010; 101(2): 517-522.
Houschyar,K.S., Ludtke,R., Dobos,G.J., Kalus,U., Broecker-Preuss,M., Rampp,T., Brinkhaus,B. and Michalsen,A. Effects of phlebotomy-induced reduction of body iron stores on metabolic syndrome: results from a randomized clinical trial. BMC Med 2012; 10: 54.
Kato,J., Kobune,M., Nakamura,T., Kuroiwa,G., Takada,K., Takimoto,R., Sato,Y., Fujikawa,K., Takahashi,M., Takayama,T., Ikeda,T. and Niitsu,Y. Normalization of elevated hepatic 8-hydroxy- 2’-deoxyguanosine levels in chronic hepatitis C patients by phlebotomy and low iron diet. Cancer Res 2001; 61(24): 8697-8702.
Kato,J., Miyanishi,K., Kobune,M., Nakamura,T., Takada,K., Takimoto,R., Kawano,Y., Takahashi,S., Takahashi,M., Sato,Y., Takayama,T. and Niitsu,Y. Long-term phlebotomy with low-iron diet therapy lowers risk of development of hepatocellular carcinoma from chronic hepatitis C. J Gastroenterol 2007; 42(10): 830-836.
Kiechl,S., Willeit,J., Egger,G., Poewe,W. and Oberhollenzer,F. Body iron stores and the risk of carotid atherosclerosis: prospective results from the Bruneck study. Circulation 1997; 96(10): 3300-3307.
Kim,M.K., Baek,K.H., Song,K.H., Kang,M.I., Choi,J.H., Bae,J.C., Park,C.Y., Lee,W.Y. and Oh,K.W. Increased serum ferritin predicts the development of hypertension among middle-aged men. Am J Hypertens. 2012; 25(4): 492-497.
Kowdley,K.V., Belt,P., Wilson,L.A., Yeh,M.M., Neuschwander-Tetri,B.A., Chalasani,N., Sanyal,A.J. and Nelson,J.E. Serum ferritin is an independent predictor of histologic severity and advanced fibrosis in patients with nonalcoholic fatty liver disease. Hepatology 2012; 55(1): 77-85.
Lee,D.H. and Jacobs,D.R., Jr. Serum markers of stored body iron are not appropriate markers of health effects of iron: a focus on serum ferritin. Med Hypotheses 2004; 62(3): 442-445.
Mainous,A.G. and Diaz,V.A. Relation of serum ferritin level to cardiovascular fitness among young men. Am J Cardiol 2009; 103(1): 115-118.
Milman,N. Serum ferritin in Danes: studies of iron status from infancy to old age, during blood donation and pregnancy. Int J Hematol 1996; 63(2): 103-135.
Nakano,M., Kawanishi,Y., Kamohara,S., Uchida,Y., Shiota,M., Inatomi,Y., Komori,T., Miyazawa,K., Gondo,K. and Yamasawa,I. Oxidative DNA damage (8-hydroxydeoxyguanosine) and body iron status: a study on 2507 healthy people. Free Radic Biol Med 2003; 35(7): 826-832.
Nelson,J.E., Klintworth,H. and Kowdley,K.V. Iron metabolism in Nonalcoholic Fatty Liver Disease. Curr Gastroenterol Rep 2012; 14(1): 8-16.
Newman,B. and Siegfried,B. Syncope after whole blood donation: factors associated with increased donor injury. Transfusion 2012; 52(1): 210-211.
Rajapurkar,M.M., Hegde,U., Bhattacharya,A., Alam,M.G. and Shah,S.V. Effect of deferiprone, an oral iron chelator, in diabetic and non-diabetic glomerular disease. Toxicol Mech Methods 2013; 23(1): 5-10.
Sung,K.C., Kang,S.M., Cho,E.J., Park,J.B., Wild,S.H. and Byrne,C.D. Ferritin is independently associated with the presence of coronary artery calcium in 12,033 men. Arterioscler Thromb Vasc Biol 2012; 32(10): 2525-2530.
Syrovatka,P., Kraml,P., Potockova,J., Fialova,L., Vejrazka,M., Crkovska,J. and Andel,M. Relationship between increased body iron stores, oxidative stress and insulin resistance in healthy men. Ann Nutr Metab 2009; 54(4): 268-274.
Valenti,L., Fracanzani,A.L., Dongiovanni,P., Bugianesi,E., Marchesini,G., Manzini,P., Vanni,E. and Fargion,S. Iron depletion by phlebotomy improves insulin resistance in patients with nonalcoholic fatty liver disease and hyperferritinemia: evidence from a case-control study. Am J Gastroenterol 2007; 102(6): 1251-1258.
Valko,M., Rhodes,C.J., Moncol,J., Izakovic,M. and Mazur,M. Free radicals, metals and antioxidants in oxidative stress-induced cancer. Chem Biol Interact. 2006; 160(1): 1-40.
van der A,D., Grobbee,D.E., Roest,M., Marx,J.J., Voorbij,H.A. and Van der Schouw,Y.T. Serum ferritin is a risk factor for stroke in postmenopausal women. Stroke 2005; 36(8): 1637-1641.
Ware,W.R. Iron and diabetes. Integrative Healthcare Practitioners 2013;(September): 76-81.
Yamamoto,M., Iwasa,M., Iwata,K., Kaito,M., Sugimoto,R., Urawa,N., Mifuji,R., Konishi,M., Kobayashi,Y. and Adachi,Y. Restriction of dietary calories, fat and iron improves non-alcoholic fatty liver disease. J Gastroenterol Hepatol. 2007; 22(4): 498-503.
Zacharski,L.R., Chow,B.K., Howes,P.S., Shamayeva,G., Baron,J.A., Dalman,R.L., Malenka,D.J., Ozaki,C.K. and Lavori,P.W. Decreased cancer risk after iron reduction in patients with peripheral arterial disease: results from a randomized trial. J Natl Cancer Inst 2008; 100(14): 996-1002.
Zacharski,L.R., Ornstein,D.L., Woloshin,S. and Schwartz,L.M. Association of age, sex, and race with body iron stores in adults: analysis of NHANES III data. Am Heart J 2000; 140(1): 98-104.
Zheng,H., Cable,R., Spencer,B., Votto,N. and Katz,S.D. Iron stores and vascular function in voluntary blood donors. Arterioscler Thromb Vasc Biol 2005; 25(8): 1577-1583.
The Hypothalamus- Pituitary-Adrenal Axis
The Hypothalamus- Pituitary-Adrenal Axis
Role in Borderline Personality Disorder
Abstract
Borderline personality disorder (BPD) is an axis II mental health disorder characterized by a series of symptoms which consists of recurrent patterns of emotional instability, strained interpersonal relationships, poor self-image and compromised inhibitory/impulse control. It is thought to affect between 1.4 to 6.0% of the population. The role of disrupted function of the hypothalamus-pituitary-adrenal (HPA) axis is an area of growing interest with respect to the pathophysiology of BPD. Overall, there is conflicting data on whether patients with uncomplicated BPD show significantly different HPA axis responsiveness compared to healthy controls, however more consistent patterns seem to emerge when evaluating BPD patients with secondary psychiatric cormorbidities. BPD patients with comorbid MDD appear to have increased cortisol levels at baseline and in response to stress, perhaps due to decreased sensitivity of the negative feedback system. BPD patients with comorbid PTSD appear to have lower levels of cortisol, indicating either cortisol hyper-suppression, or adrenal hypofunction. Treatments with the aim of regulating HPA axis responsiveness may be of benefit among this patient population.
Borderline Personality Disorder
Borderline personality disorder (BPD) is an axis II mental health disorder that has been found to be over-represented in psychiatric outpatient settings (Dixon- Gordon 2013). Its prevalence in the United States is estimated at between 1.4 % and 6.0% (Dixon-Gordon 2013, Ishak 2013), and currently there are no accurate rates of prevalence for the Canadian population (CAMH 2009). BPD is a personality disorder characterized by a series of symptoms which consists of recurrent patterns of emotional instability, strained interpersonal relationships, poor self-image and compromised inhibitory/impulse control (Carvalho 2013, Dixon-Gordon 2013, Lyons-Ruth 2011, Zimmerman 2009). Self-harm and increased suicide risk are also common features of BPD (Fox 2014). Research suggests that early life stressors are a major risk factor in the development of BPD, lending to a growing interest in the role of hypothalamus-pituitary-adrenal axis (HPA) disruption in the etiology of BPD (Carvalho 2012, Carvalho 2013, Zanarini 1997, Zimmerman 2009). This article will review current evidence pertaining to the role of the HPA-axis in BPD.
Conventional treatment for BPD focuses primarily on psychological treatments, such as Dialectical Behavioural Therapy and Cognitive Behavioural Therapy, and pharmacological therapy including use of mood stabilizers, small dose atypical antispsychotics, and antidepressants, all of which have been shown to improve the quality of life in BPD patients (Fox 2014, Ishak 2013). Due to the nature of their symptoms, in fact, BPD patients may be over-dependent on the healthcare system. A study performed by Zanarini et. al outlines the overuse of treatments available to BPD patients, such as therapy, psychiatric hospitalizations, and standing medications by BPD patients, compared to other axis II controls. Researchers found that 50-60% of BPD patients had participated in selfhelp groups, had numerous psychiatric hospitalizations, and/or day or residential treatments. Approximately 30-35% had been in either group, individual, and/or couples/family therapy, or had received intensive polypharmacy treatment (Zanarini 2001). There is a need for new strategies to enhance symptom management in this area.
Due to the complex nature of BPD, research in this area is striving to uncover new mechanisms involved in BPD, in order to improve treatment strategies that could result in more effective symptom management and enhanced quality of life for BPD patients. An area of emerging interest is the role of HPA-axis function on the pathophysiology of BPD; in this paper, we will assess the current evidence in this regard.
Hypothalamus-Pituitary-Adrenal Axis
The hypothalamus-pituitary-adrenal (HPA) axis is comprised of the hypothalamus, anterior pituitary, and adrenal cortex. This system regulates and controls the body’s response to external and internal stress by activating the commonly known “fight or flight” response. In response to a “stressor”, the hypothalamus secretes corticotropinreleasing hormone (CRH), which activates the anterior pituitary to release adrenocorticotropic hormone (ACTH), which in turn stimulates the adrenal cortex to release cortisol. Once optimal cortisol levels have concentrated the blood, this in turn activates the negative feedback system to inhibit the release of CRH from the hypothalamus, shutting down further release of hormones at all levels (Jones 2011, Zimmerman 2009). Impaired negative feedback is one of the possible problems that can occur with HPA-axis disruption.
Cortisol itself has also been an important focus as a biological mediator of psychiatric illness; conditions of hypercortisolemia, (i.e. Cushing’s syndrome), and hypocortisolemia (i.e. Addison’s disease) have both been associated with mental health symptoms such as anxiety, depression, and irritability (Sonino 1998). CRH has been found to be the stress-initiating hormone in the HPA axis; in vivo studies have shown CRH administration induces anxiety-like behaviour, such as increased emotional reactivity, withdrawal, increased motor activity and decreased social interaction in mice (Dunn 1990). Therefore, chronically elevated or depressed cortisol production by the adrenal cortex is another possible source of HPA-axis dysfunction.
HPA axis Function in BPD
A number of studies have found altered patterns in the HPA axis in BPD patients. Lieb et. al performed a study that measured salivary cortisol collected from 23 un-medicated female patients with BPD, and 24 healthy female controls, during ambulatory conditions. On days one and two, cortisol levels in response to awakening were determined in four-15 minute intervals. A low dose dexamethasone suppression test (DST, 0.5mg of dexamethasone) was administered after the last salivary sample on day two in order to assess the level of cortisol suppression on day three. Results found that patients with BPD displayed higher levels of cortisol compared to healthy controls during days one and two as well and day three. This study suggest that BPD may have increased cortisol output compared to healthy controls, perhaps through lowered sensitivity of the HPA axis negative feedback system (Lieb 2004).
Other recent studies have shown similar results. A study performed by Lyons-Ruth et. al compared salivary cortisol levels in 17 BPD females and 15 non-BPD females in response to a conflict discussion with their mothers. Results found that not only were cortisol levels higher in BPD patients anticipating the conflict discussion, BPD patients also reported higher levels of distress, post discussion compared to non-BPD patients (Lyons-Ruth 2011). Scott et. al found that women with BPD had higher baseline cortisol levels as well as higher subjective negative affect at baseline and in response to stress, compared to two control groups consisting of non-BPD patients; 1) trait-match controls (specifically on impulsivity and negative affect) and non-trait matched controls. Interestingly, this study found attenuated cortisol levels in response to stress (Scott 2013). Increased baseline cortisol levels, but attenuated cortisol levels in response to stress have been demonstrated in earlier studies. A study by Nater et. al confirmed this pattern of cortisol, but also found that ACTH and the catecholaminergic response were no different in BPD patients compared to controls (Nater 2010). This suggests that patients with BPD may have higher baseline cortisol levels, but lower cortisol response to endogenous ACTH, or stress, possibility pointing to hypofunctioning of cortisol secretion by the adrenal glands, or perhaps hypersuppression of cortisol caused by a dysfunction of the negative feedback system.
The Influence of Comorbid Psychiatric Conditions
The pattern of HPA axis function in BPD is yet to be established, with the majority of studies producing rather inconsistent results. It is possible that factors other than simply a BPD diagnosis may contribute to HPA axis regulation in BPD. BPD is strongly associated with comorbidity with other psychiatric health conditions (Zimmerman 2009, Lange 2005). The most common clinical conditions that have been found to be associated with HPA dysregulation include major depressive disorder (MDD), post-traumatic stress disorder (PTSD), and eating disorders, which occur at high rates in those with BPD (Carvalho 2012, Morris 2014, Rosenberg 2013, Wingenfeld 2013, Zimmerman 2009). In 2009, Zimmerman et. al performed a review of 18 studies published between 1950-2007. The review found that dysfunction of the HPA axis is an apparent feature in BPD based on four assessment methods used: 1) assessment of baseline cortisol levels, 2) cortisol response to the dexamethasone-suppression test (DST), 3) cortisol and ACTH response to the combined dexamethasone (DEX)- CRH test, and 4) cortisol response to psychosocial stressors. Results typically found no difference in HPA axis function in BPD patients when compared to healthy controls, with only two studies showing non-suppression of cortisol in BPD patients in the using the DST method. However, HPA axis dysfunction was more commonly linked to BPD in the presence of comorbid conditions and trauma history.
Interestingly, Zimmerman et. al. found an apparent distinction in HPA response based on the type of comorbidity present (2009). There was a paradoxical response of the HPA axis among BPD patients with either comorbid MDD or PTSD. BPD patients with comorbid MDD were found to be have impaired negative feedback inhibition of cortisol (nonsuppression) at a rate of 16-73% when administered a 1.0 mg dose of DEX. Alternately, BPD patients with comorbid PTSD showed to have elements of enhanced negative feedback (hyper-suppression) of cortisol in response to 0.5 mg dose of DEX. Patients with high level of dissociative symptoms showed increased response to psychosocial stressors, and BPD patients with a strong history of childhood abuse demonstrated increased ACTH release levels on CRH stimulation (Zimmerman 2009). A study performed by Lange et. al also showed this paradoxical response: BPD patients with PTSD showed increased suppression of cortisol when administered 0.5 mg of DEX, whereas patients with comorbid MDD and healthy controls did not demonstrate hypersuppression (Lange 2005). Another study performed by Wingenfeld et. al found that patients with BPD or BPD+PTSD had improved (autobiographical) memory when administered cortisol, whereas this memory-enhancing response to cortisol did not occur in patients with BPD+MDD (Wingenfeld 2013).
HPA axis function in BPD + MDD
Increased cortisol secretion has been established as a hallmark feature of MDD (Carvalho 2012, Schatzberg 2013), a condition often preceded by periods of chronic, or traumatic, episodic stress (Hammen 2005, Lee 2002). A recent study by Morris et. al found that patients diagnosed with MDD showed increase cortisol levels during the depressive episode, as well as during recovery, when compared to controls, who showed habituation of cortisol levels in response to stress (Morris 2014). Non-suppression of cortisol, or hypersecretion, among in BPD patients with comorbid MDD was also a pattern identified by Zimmerman et. al in their review (2009). A more recent study by Carvalho et. al found that patients with BPD and patients with MDD showed increased basal cortisol levels as well as higher post-DEX (0.5 mg) cortisol levels compared to healthy controls. The degree of increase was positively correlated with symptom severity, level of dissociation, and past history of childhood abuse (Carvalho 2012). Interestingly, this study did not find that increased cortisol levels was a result of impaired negative feedback in response to low dose DEX administration, as hypothesized (Carvalho 2012). Instead, these findings suggest that a history of exposure to chronic stress, characteristic of MDD, may be the reason for increased cortisol levels in BPD patients.
HPA axis function in BPD + PTSD
PTSD is a condition triggered by exposure to acutely traumatic events. Characteristics of PTSD can be explained through symptom clusters comprised of avoidance, numbing, separating, dysphoric- and anxious- arousal (Horn 2014). The review by Zimmerman et. al., along with other studies, found that patients with BPD who display comorbid PTSD demonstrated elements of HPA hyper-suppression, or enhanced negative feedback on cortisol, ultimately resulting in lower cortisol levels (Lange 2005, Zimmerman 2009). A recent study performed by Horn et. al compared the cortisol response to trauma among that adults with PTSD or healthy adult controls (2014). Not surprisingly, the adults with PTSD had significantly lower cortisol levels compared to healthy controls, consistent with findings outlined in the Zimmerman review (Horn 2014). A crossover study by Carvalho et. al. showed similarly consistent results, finding that divergent responses in HPA axis function upon comparing patients with BPD and BPD+PTSD. A total of 32 female patients with BPD and 32 healthy females were both given 10mg of hydrocortisone. They were then required to perform an adapted emotional go/no-go paradigm to assess response times to emotional face stimuli. It was found that both BPD and healthy controls displayed quicker reaction times to target stimuli when administered hydrocortisone, while BPD patients with comorbid PTSD displayed longer reaction times (Carvalho 2013).
In a recent study, Dixon-Gordon et. al investigated the relationship between BPD and emotional reactivity to a stressor in subjects with comorbid PTSD through subjective assessment and cortisol measurement (2013). Results showed that BPD patients with low level PTSD symptoms had a rapid onset of cortisol secretion in response to a stressor. However, those with more severe PTSD showed results consistent with previous findings: no change in cortisol in response to stress (Dixon-Gordon 2013). This study suggests that the reason for hyper-suppression of cortisol found in BPD patients with comorbid PTSD could be explained through emotional “numbing”, or allostatic load, a compensatory method for patients exposed to chronic stress/arousal of the HPA axis, typical to patients with PTSD (Jones 2011). Rather than a normal level of cortisol released in response to stress, the chronic stress and trauma experienced by a PTSD patient may place an excessive demand in the HPA axis, thereby creating HPA axis hypo-functioning over time (Horn 2014, Daskalakis 2013, Jones 2011). This may be a result of decreased synthesis of hormones, down-regulation of receptors in the pituitary glands, decreased cortisol (glucocorticord) sensitivity, or hyposecretion of cortisol by the adrenal glands, as a protective mechanism to subvert negative effects of prolonged exposure to cortisol to the body (Jones 2011); however exact mechanisms are still unclear and require more research. These studies nonetheless provide strong evidence that PTSD comorbidity may be the cause of cortisol hyper-suppression in patients with BPD.
Discussion
Investigations of HPA axis function in patients with BPD have produced variable results. While it appears that patients with BPD may generally exhibit altered HPA axis function, with the primary feature being increased baseline cortisol levels (Carvalho 2012, Lieb 2004), there is not yet sufficient evidence to definitively support altered HPA axis function in patients with BPD upon comparison with healthy controls (Carvalho 2013, Zimmerman 2009). Reasons for such varied results in HPA axis function in uncomplicated BPD could be due to failure to account for a number of variables. HPA axis function is easily influenced by age, gender, time of menstrual cycle, personal sleep/wake cycles, and nutritional status (Borsonelo 2011, Panagiotakopoulos 2014, Woods 2013). In addition, collection of cortisol samples can influence levels, as fear of needles can increase cortisol levels, and salivary cortisol collection can be altered by the presence of food particles in the saliva (Zimmerman 2009). Altered HPA function does appear to be more obvious in BPD patients in the presence of psychiatric comorbidities, most notably MDD and PTSD, (Carvalho 2012). BPD patients with comorbid MDD appear to have increased cortisol levels at baseline and in response to stress, perhaps due to decreased sensitivity of the negative feedback system. BPD patients with comorbid PTSD appear to have lower levels of cortisol, indicating either cortisol hyper-suppression, or adrenal hypofunction. These features are not unique to these comorbidities only in combination with BPD, but also have been demonstrated among patients with uncomplicated MDD and PTSD, independent of BPD. It is possible that while HPA axis function may not play a major role in the etiology of BPD, the presence of other psychiatric conditions contribute to altered HPA axis function. Nonetheless, current research does support possible low-grade alterations of HPA axis function in patients with BPD, suggesting that treatments focused on regulation of HPA axis may be of benefit to these patients, especially when BPD is associated with comorbid conditions, such as PTSD and MDD.
References
Borsonelo EC, Suchecki D, Galduróz JC. Effect of fish oil and coconut fat supplementation on depressive-type behavior and corticosterone levels of prenatally stressed male rats. Brain Res. 2011 Apr 18;1385:144-50.
Centre for Addiction and Mental Health. Borderline personality disorder: An information guide for families. URL: http://www.camh.ca/en /hospital / health_information /a_z_mental_health_and_addiction_ information /borderline_personality_disorder_an_ information_ guide_ for_ families /Pages /default.aspx Accessed January 2014
Carvalho Fernando S, Beblo T, Schlosser N, Terfehr K, Otte C, Löwe B, Wolf OT, Spitzer C, Driessen M, Wingenfeld K. Associations of childhood trauma with hypothalamic-pituitary-adrenal function in borderline personality disorder and major depression. Psychoneuroendocrinology. 2012 Oct;37(10):1659-68.
Carvalho Fernando S, Beblo T, Schlosser N, Terfehr K, Wolf OT, Otte C, Löwe B, Spitzer C, Driessen M, Wingenfeld K. Acute glucocorticoid effects on response inhibition in borderline personality disorder. Psychoneuroendocrinology. 2013 Nov;38(11):2780-8.
Daskalakis NP, Lehrner A, Yehuda R. Endocrine aspects of post-traumatic stress disorder and implications for diagnosis and treatment. Endocrinol Metab Clin North Am. 2013 Sep;42(3):503-13.
Dixon-Gordon KL, Gratz KL, Tull MT. Multimodal assessment of emotional reactivity in borderline personality pathology: the moderating role of posttraumatic stress disorder symptoms. Compr Psychiatry. 2013 Aug;54(6):639-48.
Dunn AJ, Berridge CW. Physiological and behavioral responses to corticotropin-releasing factor administration: is CRF a mediator of anxiety or stress responses? Brain Res Brain Res Rev. 1990 May- Aug;15(2):71-100.
Fox E, Krawczyk K, Staniford J, Dickens GL. A Service Evaluation of a 1-Year Dialectical Behaviour Therapy Programme for Women with Borderline Personality Disorder in a Low Secure Unit. Behav Cogn Psychother. 2014 Feb 13:1-16.
Hammen, C. (2005). Stress and depression. Annual Review of Clinical Psychology, 1,293–319.
Horn CA, Pietrzak RH, Corsi-Travali S, Neumeister A. Linking plasma cortisol levels to phenotypic heterogeneity of posttraumatic stress symptomatology. Psychoneuroendocrinology. 2014 Jan;39(11):88-93.
IsHak WW, Elbau I, Ismail A, Delaloye S, Ha K, Bolotaulo NI, Nashawati R, Cassmassi B, Wang C. Quality of life in borderline personality disorder. Harv Rev Psychiatry. 2013 May-Jun;21(3):138-50.
Jogems-Kosterman BJ, de Knijff DW, Kusters R, van Hoof JJ. Basal cortisol and DHEA levels in women with borderline personality disorder. J Psychiatr Res. 2007 Dec;41(12):1019-26.
Jones T, Moller MD. Implications of hypothalamicpituitary- adrenal axis functioning in posttraumatic stress disorder. J Am Psychiatr Nurses Assoc. 2011 Nov- Dec;17(6):393-403.
Lange W, Wulff H, Berea C, Beblo T, Saavedra AS, Mensebach C, Wingenfeld K, Driessen M. Dexamethasone suppression test in borderline personality disorder–effects of posttraumatic stress disorder. Psychoneuroendocrinology. 2005 Oct;30(9):919-23.
Lee AL, OgleWO, Sapolsky RM. 2002. Stress and depression: possible links to neuron death in the hippocampus. Bipolar Disord. 4:117–28.
Leonard BE. Serotonin receptors and their function in sleep, anxiety disorders and depression. Psychother Psychosom. 1996 Mar-Apr;65(2):66-75.
Lieb K, Rexhausen JE, Kahl KG, Schwieger U, Philipsen A, Hellhammer DH, et al. Increased diurnal salivary cortisol in women with borderline personality disorder. Journal of Psychiatric Research 2004;38:559–65.
Lyons-Ruth K, Choi-Kain L, Pechtel P, Bertha E, Gunderson J. Perceived parental protection and cortisol responses among young females with borderline personality disorder and controls. Psychiatry Res. 2011 Oct 30;189(3):426-32.
Morris MC, Rao U. Cortisol response to psychosocial stress during a depressive episode and remission. Stress. 2014 Jan;17(1):51-8.
Nater UM, Bohus M, Abbruzzese E, Ditzen B, Gaab J, Kleindienst N, Ebner-Priemer U, Mauchnik J, Ehlert U. Increased psychological and attenuated cortisol and alpha-amylase responses to acute psychosocial stress in female patients with borderline personality disorder. Psychoneuroendocrinology. 2010 Nov;35(10):1565-72.
Panagiotakopoulos L, Neigh GN. Development of the HPA axis: Where and when do sex differences manifest? Front Neuroendocrinol. 2014 Mar 13. pii: S0091-3022(14)00036-3.
Sonino N, Fava GA. Psychosomatic aspects of Cushing’s disease. Psychother Psychosom. 1998;67(3):140-6.
Rosenberg N, Bloch M, Ben Avi I, Rouach V, Schreiber S, Stern N, Greenman Y. Cortisol response and desire to binge following psychological stress: comparison between obese subjects with and without binge eating disorder. Psychiatry Res. 2013 Jul 30;208(2):156-61.
Scott LN, Levy KN, Granger DA. Biobehavioral reactivity to social evaluative stress in women with borderline personality disorder. Personal Disord. 2013 Apr;4(2):91-100.
Schatzberg AF, Keller J, Tennakoon L, Lembke A, Williams G, Kraemer FB, Sarginson JE, Lazzeroni LC, Murphy GM. HPA axis genetic variation, cortisol and psychosis in major depression. Mol Psychiatry. 2013 Oct 29.
Wingenfeld K, Driessen M, Terfehr K, Schlosser N, Fernando SC, Otte C, Beblo T, Spitzer C, Löwe B, Wolf OT. Effects of cortisol on memory in women with borderline personality disorder: role of co-morbid posttraumatic stress disorder and major depression. Psychol Med. 2013 Mar;43(3):495-505.
Woods DL, Kim H, Yefimova M. To nap or not to nap: excessive daytime napping is associated with elevated evening cortisol in nursing home residents with dementia. Biol Res Nurs. 2013 Apr;15(2):185-90.
Zanarini MC, Williams AA, Lewis RE, Reich RB, Vera SC, Marino MF, Levin A, Yong L, Frankenburg FR. Reported pathological childhood experiences associated with the development of borderline personality disorder. Am J Psychiatry. 1997 Aug;154(8):1101-6.
Zanarini MC, Frankenburg FR, Khera GS, Bleichmar J. Treatment histories of borderline inpatients. Compr Psychiatry. 2001 Mar-Apr;42(2):144-50.
Zimmerman DJ, Choi-Kain LW. The hypothalamicpituitary- adrenal axis in borderline personality disorder: a review. Harv Rev Psychiatry. 2009;17(3):167-83.
NEM Novel Joint Care Supplement
NEM Novel Joint Care Supplement
Abstract
Natural Eggshell Membrane (NEM®) is a relatively novel supplement that has been used to treat osteoarthritis as well as joint and connective tissue disorders. There is in vitro evidence that NEM® suppresses tumor necrosis factor-alpha production in peripheral blood mononuclear cell cultures, thus indicating a potentially promising anti-inflammatory action in humans. However, the amount of human clinical research conducted on NEM® is limited and the majority of the supportive data is derived from one group of researchers. The natural product industry has embraced NEM® with open arms and incorporated it into many supplements for joint health. This article reviews the four human studies conducted on NEM® to date in chronological order of publication. The first study reviewed was a combination of two pilot studies that demonstrated significant treatment responses. However, it suffered from limited enrollment and no placebo-control. The second study reviewed had a high drop-out rate, but calculated that one out of every five patients should experience a 50% pain reduction within 30 to 60 days. The third study reviewed had significant methodological flaws and demonstrated that NEM® only had an effect on post-exercise pain. The fourth study reviewed was pre-publication so the data are not described, but they showed positive results. Although more evidence is certainly warranted, the studies available lend support to the notion that NEM® is safe and effective. NEM® may serve as an alternative recommendation for patients suffering from joint pain.
Introduction
Natural Eggshell Membrane (NEM®) is a relatively novel supplement that has been used to treat osteoarthritis as well as joint and connective tissue disorders (Ruff 2009). NEM® contains naturally occurring glycosaminoglycans and proteins essential for maintaining healthy articular cartilage. The outer membrane is made of type I collagen and the inner membrane consists of type V collagen (Wong 1984). Type X collagen is also reported to occur in both these membrane structures (Arias 1991). There is in vitro evidence that NEM® suppresses tumor necrosis factor-alpha production in peripheral blood mononuclear cell cultures, thus indicating a potentially promising anti-inflammatory action in humans (Benson 2012). In addition, recent rat studies have demonstrated that oral supplementation with NEM® can also influence later-phase cytokines such as MCP-1, MIP-1α & β, RANTES and VEGF (Ruff 2014). Toxicological studies have shown that oral supplementation in rats up to 2000mg/kg body weight repeated daily for 90 days did not cause any signs of toxicity via multiple evaluation methods (Ruff 2012). However, the amount of human clinical research conducted on NEM® is limited and the majority of the supportive data is derived from one group of researchers. As such, it is imperative that the methodology and results of these trials be analyzed with scrutiny and put into context.
The natural product industry has embraced NEM® with open arms and incorporated it into many supplements for joint health. Although the trials that are available are promising, it is important for health practitioners to analyze the validity of the research available before implementing new potential therapies with patients. This article will review the four human studies conducted on NEM® to date in chronological order of publication. Although more evidence is certainly warranted, the studies available currently lend support to the notion that NEM® is both safe and effective.
STUDY ONE (2009)
Initially, two pilot clinical studies were conducted that evaluated the efficacy of NEM® for the relief of the pain and discomfort associated with joint and connective tissue disorders (Ruff 2009). To be eligible to participate, subjects must have had persistent pain lasting at least three months associated with a joint or connective tissue disorder, with a minimum baseline pain level of 2 on a 0 to 10 analog Likert scale (with 0 equating to no pain and 10 equating to the most severe pain). If currently taking medication, subjects had to undergo a washout period beforehand. Each study lasted for one month. The studies were single center, open-label, and controlled. One trial was single-arm and the other was doublearm. Subjects suspended all current pain relief medications to participate in the study. For the double-arm trial, eligible subjects were randomized to either NEM® formulation X or Y. These two formulations differed in the degree to which they were partially hydrolyzed prior to final ingredient blending (Y approximately 2.5 times more than X).
It is important to note that the investigators were not blinded to treatment (as both studies were open-label), or to randomization (in the double-arm trial). The subjects were blinded to randomization in the double-arm trial. The treatments consisted of a once daily supplementation of either NEM® in the singlearm trial, or the two different formulations of NEM® in the double-arm trial, provided in 500mg vegetarian capsules. Subjects visited the clinic at seven and 30 days following the onset of treatment. Compliance was checked by interview and by counting unused doses. Acetaminophen was allowed for pain relief rescue and subjects had to record the time and amount taken in diaries. In the single-arm and double-arm trials, the primary outcome measure was mean effectiveness of NEM® in relieving general pain, and secondary outcome measures were joint flexibility and range-of-motionassociated (ROM) pain. The Likert scale was used during the seven and 30 day clinical assessments.
In the single-arm trial, 11 subjects were enrolled and 10 of them completed the study protocol. For those that completed the study protocol, compliance was over 98%. The mean baseline pain value was 3.6 ± 1.8; the mean flexion ROM was 64.2° ± 36.5°; and the mean ROMassociated pain was 2.9 ± 2.6. Supplementation with NEM® produced a significant response at seven days for flexibility, as a 27.8% increase. At 30 days, there was a significant response for general pain (as a 72.5% reduction), an increase in flexibility (43.7% from baseline), and an improvement in ROM-associated pain (75.9% reduction). Of note, nearly half of the patients in the single-arm trial reported they were pain-free (reported score of 0) by 30 days of supplementation. Rescue pain medication was used very rarely, only once in ever 20-22 days.
In the double-arm trial, a total of 28 subjects were enrolled. 14 subjects were randomized to NEM® X and 14 were randomized to NEM® Y. 26 subjects completed the study, as one participant in each group dropped-out due to lack of efficacy. Compliance was over 96%. Supplementation with NEM® produced a significant response at seven days for both treatment arms (X: 18.4% reduction, Y: 31.3% reduction). The researchers determined that the 12.9% difference was not large and was not clinically meaningful. At 7 days, the Y arm crossed over to the X formulation for the remainder of the study. The significant treatment response continued to 30 days for pain, with a final reduction of 30.2%.
The bottom line from this trial is that there were no adverse events reported during either of the studies and the treatment was reported to be extremely well tolerated by the study participants. The safety profile for NEM® in this study was excellent, excluding the possible concern of egg allergy. In analyzing the limitations of this study, both trials suffered from limited enrollment. The double-arm trial had dropouts due to lack of efficacy. Both trials were open label, thus there is the possibility that the positive outcomes are largely due to a placebo response. The lack of investigator blinding and the fact that two of the main investigators are employed by the sponsor of the study could have further exaggerated any positive results. Overall, this study set the stage for a larger study with some modifications to help address some of these limitations.
STUDY TWO (2009)
The second and arguably more important study to examine is a randomized, double-blind, multicenter, placebo-controlled trial, known as OPTION, or Osteoarthritis Pain Treatment IncorpOrating NEM® (Ruff 2009). This study utilized subjects that suffered from symptomatic osteoarthritis of the knee. Similar to the pilot trials, subjects were required to suspend all current pain medication in order to participate. This time, subjects were randomized to receive either NEM® or placebo, using a permuted-block randomization table consisting of four subjects per block. All parties were blinded to the treatment. The treatment used was a once daily oral dose of NEM® or placebo. Clinic visits took place at 10, 30, and 60 days following the onset of treatment. Compliance was checked in a similar fashion as the previous study.
The primary endpoint was the measurement of the effectiveness of NEM® in relieving pain, stiffness, and discomfort as compared to placebo. The Western Ontario and McMasters Universities (WOMAC) Osteoarthritis Index- Visual Analog Scale was used. An absolute increase in the mean response rate of 35% was considered by the authors to be a clinically meaningful treatment effect. This trial initially had 67 subjects enrolled. However, only 20 in the treatment group and 18 in the placebo group completed the study protocol, due to various issues (stringent pain management requirements, small study sample size, some drop-outs due to lack of efficacy). Compliance was measured as over 97% for those that completed the study.
After analysis, the results showed that supplementation with NEM® produced a response rate ranging from 10.3% to 26.6% improvement better than placebo at all time points for both pain and stiffness (but not for function and overall WOMAC, these results were insignificant despite improvements).
The responses for pain and stiffness were rapid, occurring after only 10 days of supplementation. There were no adverse events associated with the treatment. The NNTs for at least a 50% reduction in pain at 10, 30, and 60 days were calculated. Clinically, the NNTs indicate that one out of every five patients should experience a 50% pain reduction within 30 to 60 days. For stiffness, the NNTs indicate that at 60 days, nearly one out of every two patients would experience a 50% reduction in stiffness. The limitations of this study included a limited initial enrollment, a high drop-out rate (43%), and a smaller mean treatment effect than anticipated.
STUDY THREE (2012)
One study was conducted and published more recently by the company Precision Nutrition Inc. on a product containing an encapsulated version of NEM® named Fast Joint Care+ (FJC+) (Berardi 2012). Although this study was not published in a peer-reviewed journal and was an independent research study, the information provided can still be examined. In this study, 60 adults experiencing chronic joint pain were supplement with FJC+ or placebo for four weeks. They completed a weekly exercise protocol that challenged their irritated joint. Subjects then rated their joint pain immediately and one day after the exercise challenge. The results showed that post-exercise joint pain in the FJC+ group decreased by four3. four%, while the placebo group experienced a 30.6% decrease in pain over the four weeks. It is important to note that there were large differences in Joint Pain Index scores at baseline for the treatment group and placebo group. The FJC+ group had a baseline score within the four0-50 range (indicating more pain), while the placebo group had Joint Pain Index scores in the 20-30 range (indicating less pain). The exact numbers were not reported directly in the study. This discrepancy would set the stage for a larger potential decrease in pain for the treatment group, which is exactly what happened. This challenges the internal validity of the study. In addition, there were no significant differences in joint pain scores across the four weeks of supplementation, indicating similar joint pain decreases in both the FJC+ group and the placebo group. Overall, this study appears to lend support to the safety of eggshell membrane, but not to its efficacy beyond immediately post-exercise.
STUDY FOUR (Pre-Publication)
A fourth human trial on NEM® has been completed by the company ESM Technologies, LLC and it is currently pre-publication (Danesch pre-publication). The trial is multicentered and open-label. It includes 44 subjects with knee and/or hip osteoarthritis. Currently, the data from this study cannot be shared publicly, but the results have been seen by this author, and permission has been given to report that the effects that were seen with NEM® were positive.
Conclusion
NEM® is a relatively novel supplement that has been used to treat osteoarthritis as well as joint and connective tissue disorders. The total quantity of human trials on NEM® is lacking. The first study reviewed was a combination of two pilot studies that demonstrated significant treatment responses. However, it suffered from limited enrollment and no placebo-control. The second study reviewed was the OPTION study, offering the most robust data on NEM® to date. Despite a high drop-out rate (43%), this study used NNTs to calculate that one out of every five patients should experience a 50% pain reduction within 30 to 60 days. With regards to stiffness, the NNTs indicated that at 60 days, nearly one out of every two patients would experience a 50% reduction. The third study reviewed had significant methodological flaws and demonstrated that NEM® only had an effect on post-exercise pain. The fourth study reviewed was pre-publication so the data were not examined closely, but still showed positive results. Overall, all trials to date have identified that NEM® is extremely safe when taken orally, with the possible exception in cases of egg allergy. The data presented suggests rapid improvements in pain and stiffness, particularly in osteoarthritis. Further well-conducted trials will provide clearer conclusions. For the time being, NEM® serves as an alternative recommendation for patients suffering from joint pain.
Author Disclosure
This author has witnessed patients reporting decreased pain after short periods of use of 500mg daily of NEM® in combination with 135mg of Boswellia serrata. Multiple patients with osteoarthritis and one case of ankylosing spondylitis reported decreased pain shortly after the initiation of treatment, while one case of rheumatoid arthritis reported no response. This author has no competing interests.
References Arias JL, Fernandez MS, Dennis JE, Caplan AI. The fabrication and collagenous substructure of the eggshell membrane in the isthmus of the hen oviduct. Matrix. 1991;11(5):313-20.
Benson KF, Ruff KJ, Jensen GS. Effects of natural eggshell membrane (NEM) on cytokine production in cultures of peripheral blood mononuclear cells: increased suppression of tumor necrosis factor-α levels after in vitro digestion. J Med Food. 2012;15(4):360-8.
Berardi, J. Eggshell Membrane Reduces Joint Pain. Precision Nutrition Inc. 2012. [Available Online: http://www.precisionnutrition.com/ wordpress/wp-content/uploads/2013/08/ Eggshell-Membrane_Precision-Nutrition.pdf]
Danesch U, Seybold M, Rittinghausen R, Treibel W, Bitterlich N. Efficacy & Safety Study of Natural Eggshell Membrane (NEM) for the Treatment of Joint & Connective Tissue Disorders [pre-publication]. Clinical Trial Identifier NCT00750854. [Clinical Trial Information Available Online: http:// clinicaltrials.gov/show/NCT00750854]
Ruff KJ, DeVore DP: Reduction of proinflammatory cytokines in rats following 7-day oral supplementation with a proprietary eggshell membrane-derived product. Modern Research in Inflammation. 2014;3:19-25.
Ruff KJ, DeVore DP, Leu MD, Robinson MA. Eggshell membrane: a possible new natural therapeutic for joint and connective tissue disorders. Results from two open-label human clinical studies. Clin Interv Aging. 2009;4:235- 40.
Ruff KJ, Endres JR, Clewell AE, Szabo JR, Schauss AG. Safety evaluation of natural eggshell membrane-derived product. Food Chem Toxicol. 2012;50(3-4):604-11.
Ruff KJ, Winkler A, Jackson RW, DeVore DP, Ritz BW. Eggshell membrane in the treatment of pain and stiffness from osteoarthritis of the knee: a randomized, multicenter, doubleblind, placebo-controlled clinical study. Clin Rheumatol. 2009;28(8):907-14.
Wong M, Hendrix MJ, von der Mark K, Little C, Stern R. Collagen in the egg shell membranes of the hen. Dev Biol. 1984;104(1):28-36.
Dr Meer Janjua, MD, CCFP
Clinic One
Dr Meer Janjua, MD, CCFP
Clinic One is an innovative health care center located in the heart of Winnipeg’s exchange district. The philosophy of the center is based on utilizing the combined expertise of various health professionals who are members of its interdisciplinary team. The clinic, founded by Dr. Meer Janjua MD, CCFP, boasts of a medical doctor working alongside a naturopathic doctor and a pharmacist. The clinic’s motto is “One team, one life,” describing the concept of several interdisciplinary practitioners coming together serving the same purpose: offering their patients the most complete and highest quality integrative medical care.
Clinic One is located on the main floor of a unique, historical building, the Maltese Cross Building. This seven-story heritage building was erected in 1909 for the Winnipeg Rubber Company, and served as one of the first distribution centers for goods traveling into Western Canada. The Company’s trademark was the modified Maltese Cross, which traces its roots to the Knights of St. John of Jerusalem during the time of the Crusades. This symbol can be found adorning the stone trim of the building. Today the building has been adapted as office space in the heart of Winnipeg.
Dr. Meer Janjua MD, CCFP, is the clinic’s founder and a second-generation physician, whose father has practiced in the Toronto area for most of his life. Dr. Janjua completed his own studies in a number of countries, including premed at Hawaii Pacific University, kinesiology and health sciences at York University, and his medical degree at the University of the West Indies in Trinidad. Dr. Janjua has both Canadian and British citizenship and his medical degree is internationally recognized. He completed his residency in family medicine in 2011 at the University of Manitoba, where he fell in love with Winnipeg and decided to stay.
Dr. Janjua attributes his open mindedness in part to his travels. International travel allowed him to become better acquainted with “people different than myself,” and in addition to broadening his understanding of humanity in general, he says that this experience has made him a better physician and health care provider. It has taught him to respect customs, which in turn enhances patient compliance. Being of Indian descent and the fact that he speaks Hindi also helps him better reach out to and serve the immigrant community in Winnipeg.
Dr. Janjua’s practice focuses on primary care. He treats a range of commonly occurring conditions in the community, for instance hypertension, diabetes, and thyroid disorders, however he also has a developed interest in surgery. He has a surgical rotation at the nearby Grace Hospital, where he assists in surgeries, and also performs out-patient minor surgery at the clinic. When his patients ask for non-pharmaceutical strategies to help manage their conditions, he openly refers them to Dr. Kelly Brown, the naturopathic doctor within the clinic. In these situations, Dr. Janjua will then collaborate with Dr. Brown afterwards to monitor the patient’s ongoing status.
Dr. Janjua is noticeably enthused about having an interdisciplinary team at the clinic. He actively sought to bring a naturopathic doctor to the clinic. He sees conventional medicine as being deeply rooted in naturopathic medicine historically, and is convinced that “every modality has its place in healthcare.” He has been impressed with how NDs treat the patient as a whole, and sees naturopathic medicine as an important part of disease prevention. Of the all-conventional versus all-natural stances, he states that “neither extreme works well.” Instead, to truly help people, he says that the collaborative approach is key.
Dr. Kelly Brown, ND is a 2010 graduate from the Canadian College of Naturopathic Medicine. She offers natural therapies for a range of commonly presenting conditions including women’s health issues, endocrine disorders such as hypothyroidism and diabetes, digestive disorders, skin conditions, immune problems, as well as mental health issues from stress and anxiety to addiction. Dr. Brown also offers acupuncture services for various conditions, as well as objective testing in the form of bloodwork, food allergy testing, and hormone analysis.
We have had the privilege of interviewing many integrative MDs; Dr. Janjua conversely practices a very conventional form of medicine, while fully appreciating the value of natural medicine, and active collaboration with an ND. He fully utilizes the diagnostic and therapeutic tools in which he has been trained, yet encourages his patients to obtain specialized ND care where appropriate, to benefit from complementary testing, nutritional counseling, supplementation, and other natural healing strategies. We feel that this type of complementary relationship – where there is room for both conventional medicine and naturopathic medicine practiced at their best, with the element of collaborative patient management – is the future of integrative medicine.
In addition to Dr. Brown ND, the clinic also has an internist that does consults on a part time basis. He treats patients with advanced, uncontrolled diabetes, performs cardiac stress tests, and conducts cardiac assessments. The clinic has also very recently added a chiropractor, and a registered massage therapist to the team, and plans to add psychological services and conduct Restricted Environmental Stimulation Therapy and Floatation Research in the fall. This innovative and dynamic clinic is also reaching out to its patients using social media, including profiles on facebook and twitter. As part of this more holistic, convenient, and interactive style of medicine, the clinic accepts walk-in patients and patients without a referral.
Dr. Janjua has a love of historical buildings, and he sees the physical environment as a critical element of a patient’s therapeutic experience. In the clinic’s historical surroundings, high ceilings, and natural lighting, he watches patients’ stress levels immediately fall, and their sense of wellbeing begin to improve. The clinic’s décor is a combination of rustic and modern. The ceilings in the reception area are 4.6 meters high, with heating ducts and other mechanical systems fully exposed. Large windows run the length of the wall, filling the room with natural light. Each of the nine clinic rooms is fitted with a different, refurbished antique door. Dr. Janjua is also an avid student of history, and over time he has accumulated a collection of historical medical artifacts. He is in the process of preparing a display for them within the clinic.
Dr. Janjua says the feedback from patients has been outstanding. Patients love it because it is so different from any other clinic, and makes their visit an enjoyable experience rather than a chore. The clinic has received considerable attention in the community and has been featured by local media outlets. We think the clinic’s distinct appearance fits well with and draws attention to the clinic’s unique, progressive vision of healthcare. In this distinctive clinic, the collaborative relationship between practitioners and the clean, uplifting environment work together to create both an inspiring and a therapeutic milieu.
Clinic One boasts an affiliated pharmacy managed by pharmacist Jeff Froese. The Clinic One Pharmacy focuses on meeting the individual needs of patients, offering patient consultations, personalized home visits, diabetes support, compounding services and blister pack drug preparation, as well as free prescription delivery. The pharmacy also carries a selection of professional line natural health products to complement the emphasis on natural healing at the clinic.
IHP thanks Dr. Janjua and the Clinic One team for allowing our readers a brief look into the inner workings of their unique team and facility. We wish the clinic much success in bringing truly progressive, integrative medicine to the city of Winnipeg.
Living Well Integrative Health Center
Living Well Integrative Health Center
Collaboration On The East Coast
Doctors Theresa Jahn, ND and Rochelle Wilcox, ND were classmates and graduated from the Canadian College of Naturopathic Medicine in 2010. Returning to their roots, they relocated to the East Coast upon graduation, initially practicing in two different clinics within the city of Halifax. Both having an interest in creating more awareness and continuing education opportunities in Atlantic Canada, in 2011 and 2012 they co-chaired the Nova Scotia Naturopathic Conference. Working together, they were able to further develop the conference to attract a growing number of NDs from Atlantic Canada as well as Ontario, making this appealing for increased company sponsorship. This has helped to foster greater awareness, collaboration and communication within the Atlantic provinces for Naturopathic Doctors. During this time, Dr. Jahn and Dr. Wilcox developed a great working relationship and were exploring opportunities to be able to practice together.The Living Well Integrative Health Centre opened in September 2012 as a brand new initiative founded by Dr. Maria Patriquin MD, CCFP. The Centre is a two-story house that has been converted into commercial real estate. With a bright, peaceful and welcoming feel, the centre has hardwood floors, along with original artwork decorating it’s walls. It has 7 treatment rooms, one of which is large enough to host small group workshops and currently Dr. Patriquin MD is facilitating a Mindfulness Based Stress Reduction group.
Mattia Bianucci Trainer: TRAIL RUNNING Coaching Alessandria – PERSONAL Fitness TRAINER Novi Ligure masteron bodybuilding download free pdf user manual for hoist fitness plate loaded pull down rpl-5201-a exercise equipment for home
The opportunity to join this centre coincided with Drs. Jahn and Wilcox’s desire to work together within an exciting and collaborative environment. Alongside Drs. Jahn, Wilcox and Patriquin, the Centre also hosts a Registered Clinical Psychologist, a Clinical Social Worker, and a Physiotherapist. To the best of our knowledge, this is currently the only centre in Atlantic Canada that includes both Naturopathic and Medical Doctors. The practitioners at Living Well all maintain their own independent practices, while having an enhanced opportunity for collaboration and interdisciplinary learning.
Drs. Jahn and Wilcox make an effort to meet other practitioners within the community to create better working relationships and really feel further improvements in this domain would create a more optimal experience for patients and practitioners alike. When asked if they have any messages for other practitioners, they reply: “In our experience, reaching out to other practitioners to collaborate with them and to encourage patients to communicate with their healthcare providers has increased efficiency and improved care.”
Doctors Jahn and Wilcox have different clinical interests. Dr. Jahn’s primarily focuses on women’s health, weight loss, athletes, and mental health.
While Dr. Wilcox has an interest in chronic disease management. Addressing stress reduction is a large component of both their practices, and they have facilitated many workshops on this topic.
Drs. Jahn and Wilcox have their own dispensary, carrying products by reputable companies such as Ascenta, NFH, Thorne, Cytomatrix, Heel, Mediherb, Douglas Labs, Metagenics, Vibrant Health, and Genestra. They also carry tinctures and ointments by St. Francis Herb Farm. They use labs such as: Rocky Mountain Analytical, Labrix, and Genova and bioimpedance analysis for the appropriate patient populations.
A challenge of being an independent practitioner within a collaborative setting, has been determining an appropriate marketing strategy. They market themselves individually as well as collectively. The Centre maintains a website, and each practitioner also has their own. They have found, when working as a team, it is essential to assess the type of marketing vehicle, the target audience and how best to convey integrative healthcare. They have participated in Women’s Health Expos, have been featured in local newspapers, use print advertising and continue to give talks within the community. Drs Jahn and Wilcox are currently partnering with the Psychologist Harpreet Aulakh at Living Well, offering presentations to bring awareness to collaborative care at schools within the surrounding area.
Drs. Jahn and Wilcox feel there is an increasing public awareness and interest in the Naturopathic profession, noting significant growth within the membership of the Nova Scotia Association of Naturopathic .Doctors within the last few years. Both doctors find this exciting as this will help to create further awareness and increased opportunities for Naturopathic Doctors on the East Coast.
In terms of future directions, Dr. Wilcox is currently pursuing continuing education in Biological Medicine and envisions that it will have a greater role within her practice. As Dr. Jahn’s uses a Functional Medicine approach in her practice and her continuing education focuses on this she would like to offer lifestyle based wellness programs in the future. We wish these wonderful practitioners continued success.
Living Well Integrative Health Center
2176 Windsor Street, Halifax, NS
Phone: (902)-406-1500
Website: http://www.livingwellihc.ca
Maria Patriquin MD, CCFP Clinic Owner
Harpreet Aulakh Reg. Psych
Jennifer van Kessel MSW, RSW
Theresa Jahn BSc ND
Rochelle Wilcox BA ND
Robin Stamm MPT
Jane White, Administrative Staff
Susan MacLean, Administrative Staff
HealthSource Integrative Medical Centre
In this Clinic Profile, IHP is pleased to present the HealthSource Integrative Medical Centre. HealthSource is run by Dr. Michael Reid, ND and Dr. Som Thammasouk, ND. This husband and wife couple met in Naturopathic College. They started practicing together in a rehabilitation clinic, but their practices outgrew the space. They eventually purchased and moved their practices into a custom-built house that was designed and originally built specifically to be a medical office. They formed HealthSource Integrative Medical Centre at the end of 2011. Dr. Reid also works part-time with Dr. Anello, MD, who is also featured in this issue of IHP. HealthSource now runs the second largest IV practice (Dr. Anello having the largest) in the Waterloo region and service approximately 100 patient visits a week. This clinic positions itself in the market as an integrative medical centre, where they bridge the gap between conventional and naturopathic medicine.
The end result is that the community recognizes them as a location where both integrative care and naturopathic care can be obtained. They receive many referrals from other practitioners in the KitchenerWaterloo area, including complex and difficult cases.
The full clinic team consists of the two Naturopathic Doctors, a Registered Practical Nurse, a Registered Massage Therapist, a Registered Holistic Nutritionist, and an Office Manager.
The 2000 square foot clinic consists of six treatment rooms, one IV room, a preparatory IV/ lab room. The clinic
provides on-site parking for patients. As a highly advanced centre, HealthSource utilizes numerous assessment tools. They do all blood draws directly in the clinic and local labs pick-up the samples. They developed their own
panel of bloodwork with a laboratory and were able to offer it at a reduced rate that consists of nutrients, hormones, inflammatory markers and chemistry. Other common tests run at the centre include tumour markers,
food intolerances, candida yeast testing, plasma amino acids, saliva hormones, and heavy metal testing. They utilize
GDL, Doctor’s Data, Great Plains Lab, Rocky Mountain Analytical, Spectracell, Igenex, and Lifelabs. Their more advanced in-office testing includes arterial stiffness assessment to gage the risk of heart disease, live blood
cell analysis/dark field microscopy, and bio-impedence analysis to assess body composition. Dr. Reid believes many
patients see great results because of the clinical application of the functional markers. Their patients don’t simply
focus on a reference range. For this reason, almost every patient will get some degree of lab work immediately at
the start of their care.
Mistletoe Therapy
Improving Outcomes In Complementary Cancer Care[spacer height=”3px”]
ABSTRACT:[spacer height=”20px”]
Mistletoe therapy is an herbal medicine best known for its role in complementary cancer care. Pioneered by Rudolf Steiner, mistletoe, also known as Viscum album L or European mistletoe, has its roots in anthroposophic medicine; today there is a large body of evidence supporting use of mistletoe injection therapy as an adjunctive cancer treatment. Lectins and viscotoxins found in mistletoe appear to be responsible for its antitumor and immune stimulating effects. Mistletoe therapy has been investigated for its effects on survival, quality of life, and tolerability of chemotherapy/ side effects associated with chemotherapy. Meta analyses and systematic reviews have demonstrated a survival benefit associated with use of mistletoe injection therapy alongside chemotherapy or as a supportive therapy. There is also a large body of evidence indicating that mistletoe therapy improves quality of life, with improvements in global quality of life as well as symptoms such as fatigue, nausea and vomiting, pain, sleep disturbances, loss of appetite, and constipation found in a preliminary study. Finally, mistletoe therapy appears to reduce side effects of chemotherapy without decreasing its effectiveness. This article will summarize the history of mistletoe therapy and assess the current evidence surrounding its use in the area of oncology, including breast, lung, and pancreatic cancers to name a few.[spacer height=”20px”]
Introduction[spacer height=”20px”]
Mistletoe therapy is a leading complementary or adjunctive cancer therapy, widely used by naturopathic doctors and medical doctors working in integrative oncology, with known immune stimulatory and antitumor effects. Today an advanced therapy most often administered as a subcutaneous injection, mistletoe has a long history of use and has undergone considerable development over that period of time.
Historically, mistletoe (Viscum album L, European mistletoe) has its roots in German anthroposophic medicine, developed by Rudolf Steiner (1861-1925) (Bar-Sela 2011). This system of medicine sought to “acknowledge a spiritual-existential dimension in humanity” (Hamre 2009) and its interaction with physical health and disease (Hamre 2009, Horneber 2010). Initially, mistletoe therapy was used in the treatment of menstrual disorders, epilepsy, high blood pressure, artherosclerosis, diabetes, asthma, migraines, neuralgias, haemorrhages, endometriosis, eczema, foot ulcers, and labour pains (Bar-Sela 2011, Ostermann 2009). In 1920, Steiner introduced mistletoe extract as a treatment for cancer, recommending an extract produced through a complex preparation method that combined sap from mistletoe harvested in the summer and in the winter (Ostermann 2009). Based on this, mistletoe therapy has been used as a cancer therapy for almost a century. During this time, additional types of extracts have been developed, and are in use today. Presently, mistletoe therapy enjoys a prominent role in complementary cancer therapy, and is the subject of a wealth of research evaluating its impact on cancer treatment outcomes including survival, side effects of chemotherapy, and quality of life. This paper will examine the current use of mistletoe therapy in oncology and discuss the research relating to its effects on cancer related outcomes.
Sources and extracts
European mistletoe (Viscum album, L) is a partially woody, semi-parasitic plant that grows on deciduous and coniferous trees including pine, apple, oak and spruce (Kelter 2007, Melzer 2009). The medicinal parts are the stems, including the sap, and the leaves (Melzer 2009). Mistletoe extracts vary according to the type of plant material used, extraction method, as well as the type of tree that the mistletoe used grows on. The host tree that the product is derived from is denoted by a suffix at the end of the preparation name, for instance, Helixor-M is from mistletoe growing on the apple tree (Malus). Older, anthroposophic extracts are produced through a standardized manufacturing method but are not standardized to their constituents; while newer, “phytotherapeutic” extracts are standardized to their mistletoe lectin (ML-1) content (Melzer 2009). For instance, Eurixor is reported to contain between 50-70ng ML-1 per mL (NCI 2013). Helixor is standardized to its biological activity: degree of cytotoxic effects against a specific leukemia cell line. Iscador may contain between 0.0001–20 mg per mL, however this content refers to the amount of plant material rather than specific constituents such as lectins (Mistel-therapie 2014). The best-known extracts in North America are Helixor and Iscador. Helixor is a cold-water extract, while Iscador is a fermented extract of fresh leafy shoots and fruits harvested in the summer and winter (Ostermann 2009). Other commercially available extracts include Abnoba, Isorel, and Iscucin. Iscucin is produced in accordance with the German Homeopathic Pharmacopoeia. Phytotherapeutic extracts include Eurixor and Lektinol. Table 1 compares the characteristics of selected mistletoe extracts (adapted from Melzer 2009).
Pharmacology
European mistletoe contains many constituents, including lectins, viscotoxins, amino acids, flavonoids, phenylpropanoids, triterpenes, phytosterol, alkaloids, polyalcohols, and polysaccharides (Melzer 2009). The active components thought to be responsible for mistletoe’s antitumor effects are mistletoe lectins (ML), and less so, viscotoxins. Lectins are glycoproteins capable of binding to the outside of cells (NCI 2013). Three classes of lectins have been identified: ML-I, ML-II, and ML-III depending on what type of surface molecule they bind (Melzer 2009). Lectins possess direct cytotoxic effects, inhibiting protein synthesis and triggering apoptosis, as well as indirect antitumor effects through stimulation of cytokine release, and increasing NK cell and macrophages activity (Bar-Sela 2011, Kelter 2007). Viscotoxins are small polypeptides related to thionins and have cytotoxic effects (Bar-Sela 2011, Melzer 2009). Viscotoxins possess immunogenic effects, inducing production of anti-viscotoxin antibodies and causing rapid lysis of the cell membrane; and enhance cytotoxic T-cell activity, oxidative granulocyte bursting, and phagocytosis (Bar-Sela 2011, Klein 2002). Viscotoxins also inhibit cell division through inhibition of DNA and RNA synthesis (Bar-Sela 2011).
Tumour Cell Growth
Mistletoe therapy has been shown to inhibit tumour cell growth independently as a monotherapy as well as when used in conjunction with chemotherapy. This is effected both by direct cytotoxic effects mediated by the lectin- and viscotoxin- components as described above, as well as indirectly through immune stimulation (Bar-Sela 2011). Mistletoe therapy inhibits cell cycle progression, and induces tumour cell apoptosis and removal through phagocytosis (Friedel2009). We describe two case reports of mistletoe therapy as monotherapy, followed by stronger evidence for its use as an adjunctive treatment alongside chemotherapy. Two case reports describe decreases in tumour size in response to intratumoral mistletoe injections among patients not undergoing chemotherapy (Orange 2010). A patient diagnosed with Merkel cell cancer was treated with subcutaneous and intravenous mistletoe therapy over a period of 9.8 months, during which the patient received no other anti-cancer treatment (Orange 2010). A second patient diagnosed with bilateral breast cancer, who declined cancer staging and surgical treatment, received subcutaneous and intratumoral mistletoe injections over a period of 31 months. Both patients experienced complete and durable remissions following use of mistletoe as a monotherapy (Orange 2010). A much higher grade of evidence, a 2008 Cochrane review, analyzed twenty-one studies of mistletoe therapy in adult patients with cancer (Horneber 2008). Of these 21 studies, seven provided data on tumour response. Two of seven studies demonstrated additive benefit associated with combined mistletoe plus chemotherapy, compared to chemotherapy alone (Borrelli 2001, Lange 1993). The remaining five studies failed to demonstrate significant benefit on antitumor response compared to standard care alone, however there was no evidence of harm (Horneber 2008).

ML mistletoe lectins; SC subcutaneous
Survival
Data from several systematic reviews and meta-analysis indicate that mistletoe therapy has a survival benefit (Horneber 2008, Kienle 2009, Ostermann 2009, Ostermann 2012). These studies are summarized in Table 2. Ostermann et al performed two systematic reviews and meta analyses including over 50 studies and concluded that mistletoe therapy in addition to standard care resulted in approximately 40% improved survival, compared to standard care alone: HR 0.59 (95%CI 0.53- 0.66 and 0.50-0.70), where HR Preparation Method Host tree Harvest Administration Dosage Standardization Anthroposophic extracts Helixor aqueous (herb) apple (M:Malus) fir (A: Abies) pine (P: Pinus) summer and winter SC 0.01–50 mg/amp (1 ml); or 100 mg (2 ml) process Iscador aqueous lacto-fermented (herb) M,A,P, elm (U: Ulmus) oak (Q: Quercus) summer and winter SC 0.0001–20 mg per amp (1 ml) process Isorel aqueous (whole plant) M, A, P summer and winter SC, IM 1–60 mg process Phytotherapeutic extracts Eurixor aqueous (herb) poplar winter SC, IC, IV 1mg or 70ng per amp (1 ml) ML-I Lektinol aqueous (herb) poplar winter SC, IV 0.02–0.07 mg or 15 ng per amp (0.5 ml) ML-I Table 1. Characteristics of Select Mistletoe Extracts (adapted from Melzer 2009) Legend: amp ampoule; IC intracoelomic (in the abdominal cavity); IM intramuscular; IV intravenous; ML mistletoe lectins; SC subcutaneous Mistletoe RM.indd 75 14-02-18 9:35 AM 76 www.ihpmagazine.com l February / March 2014 <1.0 favours mistletoe (2009, 2012). There was evidence of publication bias, undermining the strength of these findings, however these are nonetheless notable findings. A third, 2009 systematic review assessing the effectiveness of mistletoe in 19 RCTs, 16 non- randomized studies, and 11 cohort studies of patients with breast or other gynaecological cancer found that 12 of 22 studies showed significant benefits on survival, and three of nine studies showed significant benefits for remission or time to relapse (Kienle 2009). Finally, the 2008 Cochrane review included twenty-one studies, of which thirteen evaluated survival as an endpoint (Horneber 2008). Six of these 13 studies showed a survival benefit associated with mistletoe therapy, although it was noted that they lacked methodological rigor. Collectively, the weight of data from these reviews suggests that despite certain limitations in the literature, mistletoe may have clinically important anticancer effects.
A 2013 randomized controlled trial recently assessed mistletoe therapy in 72 patients with advanced non-small-cell lung cancer (NSCLC) (Bar-Sela 2013). All patients received chemotherapy with carboplatin plus gemcitabine or pemetrexed, and were then randomized to receive either mistletoe or no additional treatment. Iscador was dosed three times weekly until progression. This study found a non-significant improvement in time to tumour progression and overall survival associated with mistletoe therapy. Time to tumour progression was 6.0 months in the mistletoe group compared to 4.8 months in the control group. Overall survival (OS) was 15.9 months in the mistletoe group compared to 13.3 months in the control group. Importantly, fewer patients in the mistletoe group required chemotherapy dose reductions (13% versus 44%, p=0.005), experienced grade 3-4 non-haematological toxicities (16% versus 41%, p=0.043), or had hospitalizations (24% versus 54%, p=0.016).
Another recently published randomized controlled trial assessed overall survival among 220 patients with advanced pancreatic cancer not receiving other cancer therapy (Troger 2013). Patients were randomized to treatment with mistletoe or no treatment, although both groups received the best available supportive care. Mistletoe was dosed subcutaneously three times per week, with the primary endpoint being overall survival (OS) at 12 months. At the time of this writing, interim results are available, showing median OS as 4.8 months for the mistletoe group, compared to 2.7 months for control patients (prognosis-adjusted hazard ratio, HR 0.49, p<0.0001). Among the subgroup of patients with a ‘good’ prognosis at baseline, median OS was 6.6 versus 3.2 months (HR 0.43, p<0.0001); while among the ‘poor’ prognosis subgroup, median OS was 3.4 versus 2.0 months, respectively (HR 0.55, p=0.0031). There were no side effects related to mistletoe. These interim results indicate a significant survival benefit associated with use of mistletoe, in this case even as a monotherapy in advanced cancer patients.
Quality of Life
Quality of life (QOL) has proved to be a very important component in terms of cancer treatment. At certain stages of disease, patients are no longer candidates for curative treatment. As a result, practitioners and members of the health care team focus on prolonging life without compromising quality of life, and on relieving symptoms (Bar-Sela 2012).
Clinical data derived from studies of mistletoe therapy indicates an improvement in quality of life and decrease in side effects when used alongside chemotherapy (Bar-Sela 2013, Bussing 2012, Kim 2012). A 2012 RCT found that mistletoe significantly improved QOL (global health) (p <0.01), and increased leukocyte- and eosinophil counts (p ≤0.01) in 32 resected gastric cancer patients starting chemotherapy with doxifluridine, a 5-fluorouracil prodrug, compared to chemotherapy alone (Kim 2012). Diarrhea was also less frequently reported (7% vs. 50%, p=0.014) in the mistletoe group. A cohort study of 25 patients with various cancers reported similar findings, with improvements in QOL as measured by the EORTC QLQ-C30 questionnaire following three months of mistletoe therapy (Brandenberger 2012).
In a 2012 meta analysis, Bussing found a moderate effect size for Iscador in improving quality of life when used alongside chemotherapy, standardized mean difference 0.56 (95%CI 0.41-0.71) (Bussing 2012). Similarly, a 2010 systematic review by Kienle found that of the 26 RCTs included, 22 reported a QOL benefit associated with use of mistletoe extracts, with the remaining four studies showing either no benefit or not reporting results (2010). Improvement was seen for coping, fatigue, sleep, exhaustion, energy, nausea, vomiting, appetite, depression, anxiety, ability to work, and emotional and functional well-being. The 2008 Cochrane review found that of 16 trials evaluating these outcomes, 14 showed significant benefits on QOL, performance score, or side effects of chemotherapy (Horneber 2008).
Side Effects of Chemotherapy
Mistletoe therapy may improve the tolerability of treatment and offset the need for dose reductions among patients undergoing chemotherapy. Most of the clinical research on mistletoe in oncology has beenconducted among patients also undergoing standard care. For instance, the study by Bar-Sela cited above showed that there were significantly fewer dose reductions as well as non-hematological toxicities among NSCLC patients receiving mistletoe (2013). The study by Kim showed a reduction in the incidence of diarrhea among patients on doxifluridine (2012). In addition, several systematic reviews indicate a reduction in side effects of chemotherapy, although in most reviews, this was an endpoint combined with quality of life (Kienle 2010, 2009).
Mistletoe therapy has also been shown to improve symptoms during cancer patients’ aftercare, defined as the five years following the completion of standard care (Beuth 2008). Data from a cohort of 681 breast cancer patients found that use of mistletoe was associated with decreased overall symptoms (56% versus 70%) as well as a reduction in specific symptoms including mucositis, fatigue, pain, and headache during aftercare, compared to patients not using mistletoe (Beuth 2008).
Safety
Mistletoe therapy has a good safety profile, with most side effects being mild to moderate soreness and inflammation at injection sites, headache, fever, and chills (NCI 2013). Although rare, allergic reactions and anaphylaxis are possible. Dose escalation strategies are used to prevent such reactions.
Interactions
Based on a large volume of data evaluating the use of mistletoe alongside standard care including
chemotherapy , and the existence of several meta analyses showing survival benefits (Table 1), mistletoe appears to have a good safety profile alongside chemotherapy. If anything, there appear to be beneficial interactions such that mistletoe reduced side effects of chemo, while
improving cancer treatment outcomes such as survival and time to tumour progression (Bar-Sela 2013,
Ostermann 2012).
In particular, human studies have evaluated mistletoe therapy alongside the following chemotherapy regimens, finding that there was no evidence of mistletoe reducing the anticancer effectiveness of chemotherapy:
gemcitabine (Mansky 2013), carboplatin plus gemcitabine or pemetrexed (Bar-Sela 2013), CMF (cyclophosphamide, methotrexate and fluorouracil) (Auerbach 2005,
Horneber 2008), 5-fluorouracil (Heiny 1997, Horneber 2008), and cisplatin and ifosfamide (Lange 1993,
Horneber 2008) (Lange 1993 is an unpublished study cited by Horneber 2008). Newly published in vitro experiments suggest that mistletoe is unlikely to reduce the effectiveness of the following chemotherapy drugs: doxorubicin, gemcitabine, docetaxel and mitoxantrone, and docetaxel and cisplatin (Weissenstein 2014). The study reported that “VAE [Viscum album extract] did not inhibit chemotherapy induced cytostasis and cytotoxicity in any of our experimental settings. At higher
concentrations VAE showed an additive inhibitory effect [with chemotherapy drugs]” (Weissenstein 2014).
Conclusion
Mistletoe therapy has a long history of use as a
complementary cancer therapy, dating from the 1920s. This therapy demonstrates a good safety profile as well as important benefit on several endpoints related to cancer, including up to 40% improvement in survival, as well as benefits tof quality of life and tolerability of
chemotherapy. There are several different mistletoe extracts commercially available, however the best-known and the most researched extracts are Iscador and Helixor. Mistletoe constituents, namely lectins and viscotoxins, exert direct and indirect cytotoxic effects and acts as an immune stimulant. There is a wealth of clinical data on European mistletoe, including several systematic reviews and meta-analyses; however, there is still a need for more rigorous study design and trial reporting in future research on this agent.
References
Auerbach L, Dostal V, Vaclavik-Fleck I, Kubista E, Rosenberger A, Rieger S, et al.[Signifikant höherer Anteil aktivierter NK–Zellen durch additive misteltherapie bei chemotherapierten Mamma–Ca–Patientinnen in einer prospektiv randomisierten doppelblinden Studie]. Fortschritte in der Misteltherapie – Aktueller Stand der Forschung und klinische Anwendung. Essen: KVC Verlag, 2005:1–11. [Cited by Horneber 2008] Bar-Sela G. White-Berry mistletoe (Viscum album L.) as complementary treatment in cancer: does it help? European Journal of Integrative Medicine. 2011;3(2):55-62. Bar-Sela G, Wollner M, Hammer L, Agbarya A, Dudnik E, Haim N. Mistletoe as complementary treatment in patients with advanced non-small-cell lung cancer treated with carboplatin-based combinations: a randomised phase II study. Eur J Cancer. 2013 Mar;49(5):1058-64.
Borrelli E. Evaluation of the quality of life in breast cancer patients undergoing lectin standardized mistletoe therapy. Minerva Medica 2001;92(Suppl 1 Nr 3):105–7.
Brandenberger M, Simões-Wüst AP, Rostock M, Rist L, Saller R. An exploratory study on the quality of life and individual coping of cancer patients during mistletoe therapy. Integr Cancer Ther. 2012 Jun;11(2):90-100.
Büssing A, Raak C, Ostermann T. Quality of life and related dimensions in cancer patients treated with mistletoe extract (iscador): a meta-analysis. Evid Based Complement Alternat Med. 2012;2012:219402.
Friedel WE, Matthes H, Bock PR, Zänker KS. Systematic evaluation of the clinical effects of supportive mistletoe treatment within chemo- and/or radiotherapy protocols and long-term mistletoe application in nonmetastatic colorectal carcinoma: multicenter, controlled, observational cohort study. J Soc Integr Oncol. 2009 Fall;7(4):137-45.
Hamre HJ, Kiene H, Kienle GS. Clinical research in anthroposophic medicine. Altern Ther Health Med. 2009 Nov-Dec;15(6):52-5.
Heiny BM, Albrecht V. [Komplementäre Therapie mit Mistellektin–1–normiertem Extrakt – Lebensqualitätsstabilisierung beim fortgeschrittenen kolorektalen Karzinom – Fakt oder Fiktion?]. Medizinische Welt 1997;48:419–423. [Cited by Horneber 2008].
Horneber MA, Bueschel G, Huber R, Linde K, Rostock M. Mistletoe therapy in oncology. Cochrane Database Syst Rev. 2008 Apr 16;(2):CD003297.
Kelter G, Schierholz JM, Fischer IU, Fiebig HH. Cytotoxic activity and absence of tumor growth stimulation of standardized mistletoe extracts in human tumor models in vitro. Anticancer Res. 2007 Jan-Feb;27(1A):223-33.
Kienle GS, Glockmann A, Schink M, Kiene H. Viscum album L. extracts in breast and gynaecological cancers: a systematic review of clinical and preclinical research. J Exp Clin Cancer Res. 2009 Jun 11;28:79.
Kienle GS, Kiene H. Review article: Influence of Viscum album L (European mistletoe) extracts on quality of life in cancer patients: a systematic review of controlled clinical studies. Integr Cancer Ther. 2010 Jun;9(2):142-57.
Kim KC, Yook JH, Eisenbraun J, Kim BS, Huber R. Quality of life, immunomodulation and safety of adjuvant mistletoe treatment in patients with gastric carcinoma – a randomized, controlled pilot study. BMC Complement Altern Med. 2012 Oct 3;12:172.
Klein R, Classen K, Berg PA, Lüdtke R, Werner M, Huber R. In vivo-induction of antibodies to mistletoe lectin-1 and viscotoxin by exposure to aqueous mistletoe extracts: a randomised double-blinded placebo controlled phase I study in healthy individuals. Eur J Med Res. 2002 Apr 30;7(4):155-63.
Lange O, Scholz G, Gutsch J. Modulation of the subjective and objective toxicity of an aggressive chemoradiotherapy with Helixor [Modulation der subjektiven und objektiven Toxizität einer aggressiven Chemo/Radiotherapie mit Helixor]. 1993: 17268. [Cited by Horneber 2008].
Mansky PJ, Wallerstedt DB, Sannes TS, Stagl J, Johnson LL, Blackman MR, Grem JL, Swain SM, Monahan BP. NCCAM/NCI Phase 1 Study of Mistletoe Extract and Gemcitabine in Patients with Advanced Solid Tumors. Evid Based Complement Alternat Med. 2013;2013:964592.
Melzer J, Iten F, Hostanska K, Saller R. Efficacy and safety of mistletoe preparations (Viscum album) for patients with cancer diseases. A systematic review. Forsch Komplementmed. 2009 Aug;16(4):217-26.
Mistel-therapie. Iscador. No date updated provided. http://www.mistel-therapie.de/index.php5?page=39&lang=1 Accessed 2 February 2014.
NCI, National Cancer Institute. Mistletoe Extracts PDQ. Updated 06/07/2013. URL: http://www.cancer.gov/cancertopics/pdq/cam/mistletoe/HealthProfessional/page1 Accessed 10 Jan 2014.
Orange, M., Fonseca, M., Lace, A., Von Laue, H. B., Geider, S. (2010). Durable tumour responses following primary high dose induction with mistletoe extracts: two case studies. European Journal of Integrative Medicine. 2010;2(2):63-69.
Ostermann T, Büssing A. Retrolective studies on the survival of cancer patients treated with mistletoe extracts: a meta-analysis. Explore (NY). 2012 Sep-Oct;8(5):277-81.
Ostermann T, Raak C, Bussing A. Survival of cancer patients treated with mistletoe extract (iscador): a systemic literature review. Biomed Central. 2009;9:451-460.
Tröger W, Galun D, Reif M, Schumann A, Stankovicec N, Milicevic M. Viscum album [L.] extract therapy in patients with locally advanced or metastatic pancreatic cancer: a randomised clinical trial on overall survival.Eur J Cancer. 2013Dec;49(18):3788-97.
Weissenstein U, Kunz M, Urech K, Baumgartner S. Interaction of standardized mistletoe (Viscum album) extracts with chemotherapeutic drugs regarding cytostatic and cytotoxic effects in vitro. BMC Complement Altern Med. 2014 Jan 8;14(1):6.