The Case for Blood Donation
Why Blood Donation is a Larger Health Issue than Generally Appreciated
Abstract
Blood donation is probably more often motivated by altruism than by the notion that it carries health benefits. Iron is involved in many biological processes. (Valium) It is also well known to be a source of reactive oxygen species (ROS) trough the Fenton reaction, and the result can be oxidative stress with cellular, DNA, vascular and organ damage. The currently used laboratory reference range for normal serum ferritin typically covers from the 5th to the 80th or 90th population percentile and is gender dependent. However, there is considerable evidence that within this range adverse effects of iron are implicated, which impact the development and progression of a number of common disorders. There is also considerable data indicating that lowering ferritin levels within the normal range to values corresponding to near iron depletion produces beneficial results for a number of diseases. In addition, oxidative DNA damage is strongly and significantly correlated with ferritin levels within the normal reference range with no apparent threshold. There appears to be no consensus as to what constitutes an optimum level of iron body stores. However, there is a large range of these stores between anemia and abnormal levels that suggest intervention. It is suggested that optimum ferritin levels may be at the low end of the normal reference range near the threshold for anemia. The simplest and quickest way to dramatically lower ferritin levels is blood donation.
Introduction
In the September 2013 issue of Integrated Healthcare Practitioners, the potential toxicity of iron and the connection between iron and diabetes was explored (Ware 2013). While not emphasized, elevated levels of serum ferritin, a measure of iron body stores and active iron, are associated with enhanced risk for a number of disorders, and the levels from which lowering produced clinical benefits were well below the upper limit of the normal reference ranges used internationally. In what follows, this observation will be generalized to a number of disorders and the question, “what is the optimum serum ferritin level?” will be addressed.
Ferritin reference ranges and levels regarded as normal
There is some variation in the upper reference limits for serum ferritin that constitute a threshold for concern. In North America, typical values for the upper limit of normal are 300-320 ng/mL for men, and 150-307ng/ mL for women. Similar values are common in other regions. In a recent discussion of the diagnosis of hyperferritinemia, the authors indicate elevated ferritin levels are >300 ng/ mL for men and >200 ng/mL for women.
Hemochromatosis is not generally treated until ferritin levels are considerably above these upper limits of normal (Adams 2011).
The variation of serum ferritin levels with age and gender in the US can be obtained from the Third National Health and Nutrition Examination Survey (NHANES III) (Zacharski 2000). For Caucasian men, the mean serum ferritin at age 17-19 is about 60 ng/mL and by age 30-39 has a plateau at about 150 ng/mL where it remains until about age 60 when a steady decline to about 90 ng/mL by age 90 is observed. For women, the mean value is quite constant at around 30 ng/mL until after menopause and then increases to about 80 ng/mL by age 60 and then gradually increases to about 100 ng/mL at age 80-90.
The multiplicity of physiologic processes involving ferritin, and in particular its role as an acute phase reactant, has caused some to question the use of serum ferritin as a marker for the risk of various disorders (Adams 2012, Lee 2004). However, as will be discussed, risk of incidence of various disorders correlates with ferritin levels in a large number of studies with large sample size and a diversity of disorders. More importantly, there are also a number of disorders where lowering “normal” ferritin by blood removal and driving ferritin levels from above, near or even below the population mean to near iron depletion (threshold for anemia) produces significant improvements in clinical manifestations or markers.
Thresholds of ferritin levels and risk of disease
In addition to the associations between ferritin levels and diabetes discussed in the cited IHP article, the following are of interest.
• In a study of men and postmenopausal women, a ferritin threshold of >200 ng/mL was associated with an increase in risk of a first heart attack (Holay 2012).
• A study classified CHD-positive patients as having one or more coronary arteries with ≥ 50% blockage. Comparison of ferritin levels revealed that those CHD-positive had on average ferritin levels of 121 vs. 73 ng/mL for those CHD-negative (Haidari 2001).
• At a ferritin threshold of >137 ng/mL, increased risk of ischemic stroke was found in a study of postmenopausal women (van der A 2005).
• A ferritin threshold of 145 to 164 ng/mL was found for increased risk of acute ischemic stroke transforming to a hemorrhagic stroke in older men and women (Choi 2012).
• A large study examined the association between ferritin levels and laboratory measured cardiovascular fitness (CVF) in young men. The likelihood of the absence of CVF, adjusted for numerous potential confounders, became significantly apparent at a ferritin threshold of >150 (Mainous 2009).
• Middle-aged men had significant risk of developing hypertension, defined as BP ≥ 140/90 mm Hg, at a ferritin threshold of > 146 ng/mL (Kim 2012).
• A study of the relationship between increased ferritin, oxidative stress and insulin resistance in 151 healthy men revealed no threshold, only continuous increases in markers with ferritin levels from the first tertile (≤97 ng/mL) to the third (≥180 ng/mL). Notably, the correlations remained strong and significant after adjustment for inflammation (Syrovatka 2009).
• In a 5-year study the association between ferritin levels and the incidence and progression of carotid atherosclerosis was examined among a cohort of 826 men and women age 40 to 79 years. Serum ferritin was found to be the strongest risk factor of overall progression, and changes in ferritin levels during follow-up modified the risk with lower levels beneficial. The risk of incident carotid atherosclerosis increased linearly and doubled from quintile I (0-36 ng/mL) to quintile IV (130-222 ng/mL). When subjects with < 50 ng/mL of ferritin were compared with those ≥ 50 ng/mL, the increase in incidence of carotid atherosclerosis for elevated ferritin per 100 person years was approximately 0.3 vs. 4.8, 0.2 vs. 4.5 and 1.6 vs. 4.4 for men, premenopausal women and naturally menopausal women, respectively (Kiechl 1997).
• In a study of over 12,000 men, the ferritin threshold for increased risk of any coronary artery calcium was >257 ng/mL (Sung 2012).
• In a study of men from either northern Europe (Zutphen) or the Mediterranean south (Crete), mean ferritin levels were 134 ng/mL and 70 ng/mL, respectively. Those from Crete (70 ng/mL) had consistently lower levels of indicators of oxidative stress, higher antioxidant capacity and higher concentrations of major antioxidants than men from Zutphen (Buijsse 2007).
Ferritin thresholds and benefit in iron-lowering studies
The following studies are particularly important since they appear less subject to confounding than those cited above.
• In a randomized prospective trial, iron reduction in male smokers with peripheral arterial disease (PAD) reduced the risk of death or nonfatal heart attack, such that the number needed to treat with phlebotomy over five years to prevent one acute event was only eight, a very low number and uncommon in clinical studies. Significant benefits were also seen for all cause mortality, non-fatal MI and stroke. The initial and final mean ferritin levels were 125 ng/mL and 84 ng/ mL. In a larger study in which the above was imbedded, unequivocal benefits for all cause mortality, non-fatal MI and stroke were found for iron reduction in younger individuals 43- 61 years of age including non-smokers, when phlebotomy reduced ferritin levels to < 70 ng/ mL (Depalma 2012, Depalma 2013).
• A controlled trial involving phlebotomy which decreased ferritin levels from a mean of 188 to 105 ng/mL for a group of men and women with the metabolic syndrome, found a decrease in systolic blood pressure from 149 to 131 mm Hg, with no change in the control group. Fasting blood glucose, HbA1c and heart rate were also significantly decreased (Houschyar 2012).
• Use of the oral prescription chelator deferiprone over nine months in patients with non-diabetic kidney disease reduced ferritin from 144 to 59 ng/mL and resulted in significant clinical improvements (Rajapurkar 2013).
• In a trial using phlebotomy in patients with peripheral artery disease, a reduction in mean ferritin levels from 122 to 74 ng/mL resulted in a significant reduction in the incidence of visceral malignancy (Zacharski 2008).
Comparison of both the above thresholds for risk and the baseline ferritin levels from which lowering produced benefit reveals an incompatibility with the commonly used reference range values regarded as normal and of no concern.
Iron stores reductions and non-alcoholic fatty liver disease
Non-alcoholic fatty liver disease (NAFLD) starts with simple hepatic steatosis and can progress to non-alcoholic steatohepatitis (NASH). One hypothesis for the pathogenesis of this disorder is the involvement of iron in increased oxidative stress and liver inflammation (Nelson 2012). High ferritin levels have been found independently associated with advanced hepatic fibrosis (Kowdley 2012). The following iron depletion studies are thus of interest.
• A study of phlebotomy on an insulin resistant group of patients with NAFLD and markedly
elevated ferritin levels showed a significant reduction in insulin resistance (HOMA-IR decreased from 4.81 to 3.12) when ferritin levels were reduced from 438 to 52 ng/mL. More importantly, alanine transaminase (ALT) decreased from a mean of 58.1 to near normal 34.3 IU/L (Valenti 2007).
• A study examined the effect of phlebotomy in either carbohydrate intolerant subjects or type 2 diabetics, including some also diagnosed with NAFLD based on elevated ALT and ultrasound evidence of steatosis (Facchini 2002). The NAFLD and non- NAFLD groups had baseline mean ferritin levels of 299 and 220 ng/mL, respectively. Phlebotomy produced near iron depletion (ferritin 31-15 ng/mL) and ALT fell from 61 to 32 IU/L in the NAFLD group. There were insignificant ALT changes observed in the NAFLD-free group.
• In a study where ferritin levels were manipulated with diet, 12 patients with NASH were placed on a calorie, fat and iron restricted diet. Baseline mean ferritin levels were 280 ng/mL initially and 128 ng/mL at 6 months of intervention. ALT levels decreased from 104 to 42 IU/L over the same period. Large changes were also seen in aspartate aminotransferase (AST) levels (Yamamoto 2007).
Iron and oxidative damage
Urinary 8-hydroxydeoxyguanosine (8-OHdG) is a reliable and frequently used biomarker of systemic oxidative DNA damage (Nakano 2003, Valko 2006). The following studies are of particular interest because of the apparent absence of a threshold for adverse effects of iron.
For over 500 healthy Japanese aged 21-67, linear correlations between 8-OHdG and ferritin were found, with Spearmen rank correlation coefficients of 0.47, 0.76 and 0.73 for men overall, women aged less than 50, and women 50 years or older, respectively. These strong correlations were essentially unchanged after adjustment for potential confounders. Subjects exhibited a large range of ferritin levels from near iron depletion to around 300 ng/mL for men and 100 ng/mL for women (Hori 2010).
For a cohort of 2500 healthy individuals aged 22 to 89 years, a smooth, almost linear 2.5 fold increase in 8-OHdG was found for men as ferritin ranged from 10 ng/mL to about 300 ng/mL. For women, 8-OHdG was increased by a factor of 3 for ferritin levels ranging from below 9 to 160 ng/mL (Nakano 2003). These results suggest no threshold and this observation is consistent with a study of vascular function and ferritin levels (Zheng 2005), and a study of oxidative stress and insulin resistance (Syrovatka 2009).
In addition, a study of ferritin lowering with phlebotomy was found to reduce 8-OHdG in patients with chronic hepatitis C (Kato, 2001). Another study found a beneficial impact of reducing iron stores to near depletion on the development of hepatocellular carcinoma (HCC) from chronic hepatitis C (Kato 2007). In both studies, final ferritin levels were about 10 ng/mL.
Influence of blood donation on ferritin levels
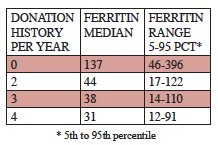
Blood donation typically removes 450-500 mL per visit. The following results from a large sample of Danish men 30 to 66 years of age illustrate the dramatic effect of blood donation (Milman 1996).
Danish study of the influence of blood donation of serum ferritin levels (ng/mL) in men, 30-66 years of age (Milman 1996).
These data are consistent with a recent American study (Cable 2011). Thus on average three to four donations per year will result in a ferritin level mostly below 100 ng/mL with a median representing near iron depletion.
Risks associated with blood donation
Blood donation is not totally free of adverse events. These include syncope, bruises and haematomas and sensory changes. Syncope is rare even among those over 40 (typically 0.05% incidence). Others, while frequently of higher incidence, are almost always transient and rarely of significance. The incidence of permanent injury has been cited as about 0.002% (Amrein 2012, Gavillet 2013, Newman 2012).
Of greater concern is the impact of frequent donations on iron status. Deferral at blood donor clinics is generally based on a finger stick haemoglobin assay with a commonly used threshold for deferral of < 12.5 g/dL, a threshold that has been questioned (Cable 2011). The common lower limit for normal is 13.5 g/dL, and served as the older cut-off for deferral. A 12- month multicenter donor clinic follow-up study examined the iron status in blood donors screened by finger stick. The threshold for diagnosing unacceptable iron stores was a ferritin level < 12 ng/mL. This was found in 15% of those passing the haemoglobin screening. The finger stick haemoglobin test is evidently not excluding some individuals with low iron status and thereby increasing the risk of an adverse impact of donation. For frequent blood donors, an annual ferritin test and if appropriate, a follow-up test for iron deficient erythropoiesis are indicated (Cable 2011).
Conclusions
It is clear from the above studies that the serum ferritin thresholds for the appearance of risk and the baseline values from which lowering produces benefit are predominantly well below the upper reference range values for normal, and in fact more closely correspond to population 50th percentiles. Indeed, lowering ferritin to levels far below the 50th population percentile produces significant benefit associated with severity of a variety of disorders. In some studies, this is observed even when the baseline level for lowering is already quite low. The DNA oxidative stress studies strongly support the view that the optimum ferritin level is one representing near iron depletion, which can only be easily achieved by blood donation or phlebotomy. This conclusion challenges a widely held belief that ferritin anywhere in the normal reference range should not cause concern.
References
Adams,P.C. Diabetes: Serum ferritin levels and T2DM–are body iron stores elevated? Nat Rev Endocrinol 2012; 8(10): 573-575.
Adams,P.C. and Barton,J.C. A diagnostic approach to hyperferritinemia with a non-elevated transferrin saturation. J Hepatol. 2011; 55(2): 453-458.
Amrein,K., Valentin,A., Lanzer,G. and Drexler,C. Adverse events and safety issues in blood donation–a comprehensive review. Blood Rev 2012; 26(1): 33-42.
Buijsse,B., Feskens,E.J., Moschandreas,J., Jansen,E.H., Jacobs,D.R., Jr., Kafatos,A., Kok,F.J. and Kromhout,D. Oxidative stress, and iron and antioxidant status in elderly men: differences between the Mediterranean south (Crete) and northern Europe (Zutphen). Eur J Cardiovasc Prev Rehabil. 2007; 14(4): 495-500.
Cable,R.G., Glynn,S.A., Kiss,J.E., Mast,A.E., Steele,W.R., Murphy,E.L., Wright,D.J., Sacher,R.A., Gottschall,J.L., Vij,V. and Simon,T.L. Iron deficiency in blood donors: analysis of enrollment data from the REDS-II Donor Iron Status Evaluation (RISE) study. Transfusion 2011; 51(3): 511-522.
Choi,K.H., Park,M.S., Kim,J.T., Nam,T.S., Choi,S.M., Kim,B.C., Kim,M.K. and Cho,K.H. The serum ferritin level is an important predictor of hemorrhagic transformation in acute ischaemic stroke. Eur J Neurol 2012; 19(4): 570-577.
Depalma,R.G. and Zacharski,L.R. Iron reduction benefits: positive results from a “negative” prospective randomized controlled trial. Vasc Endovascular. Surg 2012; 46(7): 596-597.
Depalma,R.G., Zacharski,L.R., Chow,B.K., Shamayeva,G. and Hayes,V.W. Reduction of iron stores and clinical outcomes in peripheral arterial disease: outcome comparisons in smokers and non-smokers. Vascular 2013.
Facchini,F.S., Hua,N.W. and Stoohs,R.A. Effect of iron depletion in carbohydrate-intolerant patients with clinical evidence of nonalcoholic fatty liver disease. Gastroenterology 2002; 122(4): 931-939.
Gavillet,M., Tissot,J.D. and Canellini,G. Blood donation associated risks: data from a Swiss regional haemovigilance program. Transfus. Med 2013; 23(4): 269-271.
Haidari,M., Javadi,E., Sanati,A., Hajilooi,M. and Ghanbili,J. Association of increased ferritin with premature coronary stenosis in men. Clin Chem 2001; 47(9): 1666-1672.
Holay,M.P., Choudhary,A.A. and Suryawanshi,S.D. Serum ferritin-a novel risk factor in acute myocardial infarction. Indian Heart J 2012; 64(2): 173-177.
Hori,A., Mizoue,T., Kasai,H., Kawai,K., Matsushita,Y., Nanri,A., Sato,M. and Ohta,M. Body iron store as a predictor of oxidative DNA damage in healthy men and women. Cancer Sci 2010; 101(2): 517-522.
Houschyar,K.S., Ludtke,R., Dobos,G.J., Kalus,U., Broecker-Preuss,M., Rampp,T., Brinkhaus,B. and Michalsen,A. Effects of phlebotomy-induced reduction of body iron stores on metabolic syndrome: results from a randomized clinical trial. BMC Med 2012; 10: 54.
Kato,J., Kobune,M., Nakamura,T., Kuroiwa,G., Takada,K., Takimoto,R., Sato,Y., Fujikawa,K., Takahashi,M., Takayama,T., Ikeda,T. and Niitsu,Y. Normalization of elevated hepatic 8-hydroxy- 2’-deoxyguanosine levels in chronic hepatitis C patients by phlebotomy and low iron diet. Cancer Res 2001; 61(24): 8697-8702.
Kato,J., Miyanishi,K., Kobune,M., Nakamura,T., Takada,K., Takimoto,R., Kawano,Y., Takahashi,S., Takahashi,M., Sato,Y., Takayama,T. and Niitsu,Y. Long-term phlebotomy with low-iron diet therapy lowers risk of development of hepatocellular carcinoma from chronic hepatitis C. J Gastroenterol 2007; 42(10): 830-836.
Kiechl,S., Willeit,J., Egger,G., Poewe,W. and Oberhollenzer,F. Body iron stores and the risk of carotid atherosclerosis: prospective results from the Bruneck study. Circulation 1997; 96(10): 3300-3307.
Kim,M.K., Baek,K.H., Song,K.H., Kang,M.I., Choi,J.H., Bae,J.C., Park,C.Y., Lee,W.Y. and Oh,K.W. Increased serum ferritin predicts the development of hypertension among middle-aged men. Am J Hypertens. 2012; 25(4): 492-497.
Kowdley,K.V., Belt,P., Wilson,L.A., Yeh,M.M., Neuschwander-Tetri,B.A., Chalasani,N., Sanyal,A.J. and Nelson,J.E. Serum ferritin is an independent predictor of histologic severity and advanced fibrosis in patients with nonalcoholic fatty liver disease. Hepatology 2012; 55(1): 77-85.
Lee,D.H. and Jacobs,D.R., Jr. Serum markers of stored body iron are not appropriate markers of health effects of iron: a focus on serum ferritin. Med Hypotheses 2004; 62(3): 442-445.
Mainous,A.G. and Diaz,V.A. Relation of serum ferritin level to cardiovascular fitness among young men. Am J Cardiol 2009; 103(1): 115-118.
Milman,N. Serum ferritin in Danes: studies of iron status from infancy to old age, during blood donation and pregnancy. Int J Hematol 1996; 63(2): 103-135.
Nakano,M., Kawanishi,Y., Kamohara,S., Uchida,Y., Shiota,M., Inatomi,Y., Komori,T., Miyazawa,K., Gondo,K. and Yamasawa,I. Oxidative DNA damage (8-hydroxydeoxyguanosine) and body iron status: a study on 2507 healthy people. Free Radic Biol Med 2003; 35(7): 826-832.
Nelson,J.E., Klintworth,H. and Kowdley,K.V. Iron metabolism in Nonalcoholic Fatty Liver Disease. Curr Gastroenterol Rep 2012; 14(1): 8-16.
Newman,B. and Siegfried,B. Syncope after whole blood donation: factors associated with increased donor injury. Transfusion 2012; 52(1): 210-211.
Rajapurkar,M.M., Hegde,U., Bhattacharya,A., Alam,M.G. and Shah,S.V. Effect of deferiprone, an oral iron chelator, in diabetic and non-diabetic glomerular disease. Toxicol Mech Methods 2013; 23(1): 5-10.
Sung,K.C., Kang,S.M., Cho,E.J., Park,J.B., Wild,S.H. and Byrne,C.D. Ferritin is independently associated with the presence of coronary artery calcium in 12,033 men. Arterioscler Thromb Vasc Biol 2012; 32(10): 2525-2530.
Syrovatka,P., Kraml,P., Potockova,J., Fialova,L., Vejrazka,M., Crkovska,J. and Andel,M. Relationship between increased body iron stores, oxidative stress and insulin resistance in healthy men. Ann Nutr Metab 2009; 54(4): 268-274.
Valenti,L., Fracanzani,A.L., Dongiovanni,P., Bugianesi,E., Marchesini,G., Manzini,P., Vanni,E. and Fargion,S. Iron depletion by phlebotomy improves insulin resistance in patients with nonalcoholic fatty liver disease and hyperferritinemia: evidence from a case-control study. Am J Gastroenterol 2007; 102(6): 1251-1258.
Valko,M., Rhodes,C.J., Moncol,J., Izakovic,M. and Mazur,M. Free radicals, metals and antioxidants in oxidative stress-induced cancer. Chem Biol Interact. 2006; 160(1): 1-40.
van der A,D., Grobbee,D.E., Roest,M., Marx,J.J., Voorbij,H.A. and Van der Schouw,Y.T. Serum ferritin is a risk factor for stroke in postmenopausal women. Stroke 2005; 36(8): 1637-1641.
Ware,W.R. Iron and diabetes. Integrative Healthcare Practitioners 2013;(September): 76-81.
Yamamoto,M., Iwasa,M., Iwata,K., Kaito,M., Sugimoto,R., Urawa,N., Mifuji,R., Konishi,M., Kobayashi,Y. and Adachi,Y. Restriction of dietary calories, fat and iron improves non-alcoholic fatty liver disease. J Gastroenterol Hepatol. 2007; 22(4): 498-503.
Zacharski,L.R., Chow,B.K., Howes,P.S., Shamayeva,G., Baron,J.A., Dalman,R.L., Malenka,D.J., Ozaki,C.K. and Lavori,P.W. Decreased cancer risk after iron reduction in patients with peripheral arterial disease: results from a randomized trial. J Natl Cancer Inst 2008; 100(14): 996-1002.
Zacharski,L.R., Ornstein,D.L., Woloshin,S. and Schwartz,L.M. Association of age, sex, and race with body iron stores in adults: analysis of NHANES III data. Am Heart J 2000; 140(1): 98-104.
Zheng,H., Cable,R., Spencer,B., Votto,N. and Katz,S.D. Iron stores and vascular function in voluntary blood donors. Arterioscler Thromb Vasc Biol 2005; 25(8): 1577-1583.