A novel therapeutic approach
Abstract
Depressive disorders affect up to one third of adults, and encompass both psychological as well as physical symptoms, resulting in considerable disability with respect to daily functioning. Major depression is defined by the presence of five of the following symptoms: anhedonia (depressed mood), sleep disturbance, weight changes, psychomotor problems, fatigue, lack of concentration, worthlessness/guilt, and suicidal ideation. Selective serotonin/ norepinephrine reuptake inhibitors constitute the standard firstline conventional therapy. Key natural agents include eicosapentanoic acid, St. John’s wort, 5-hydroxy-tryptophan (5-HTP), and vitamin B12. Acetyl-L-carnitine (ALC) is a new, emerging agent with a growing body of evidence supporting its use in depression. ALC has been shown to improve cellular energy production in specific areas of the brain, increase brain levels of serotonin and norepinephrine, and may regulate expression of the glutamate receptor, mGlu2. Clinical studies indicate comparable efficacy for ALC compared to other active therapies including fluoxetine and amisulpride, and better tolerability. This article will review emerging research on the role of ALC in depressive disorders.
Introduction
Depression is a debilitating disorder that causes considerable disruption in an individual’s life, work, and health. Depression is most common during the second to third decade of life, with as many as up to 30% of patients confirming symptoms, and is twice as common among women compared to men (Merck 2010). Additionally, symptoms of mood disorders often overlap, making it difficult to distinguish depression from other mood disorders. Depressive disorders are associated with a host of emotional and physical symptoms that interfere with daily function and contribute to decreased interest in daily activities. The Diagnostic and Statistical Manual of Mental Disorders, 4th edition (DSM IV) classifies depressive disorders as follows: major depressive disorder, dysthymia and depressive disorder not otherwise specified and mixed depression/anxiety.
Major depressive disorder is defined by the DSM IV as having five of nine symptoms: depressed mood (anhedonia) for majority of the day every day, for a minimum of two consecutive weeks, and other symptoms including the following: sleep disturbance, weight changes, psychomotor problems, fatigue, lack of concentration, worthlessness/guilt and suicidal ideation (Zimmerman 2006 A,B).

Diagnostics methods and tools
The following questionnaires are often utilized to help assess depression:
a) The Beck Depression Inventory (BDI) measures the intensity, severity, and depth of depression by asking 21 questions.
b) The Hamilton Depression Rating Scale (HDRS) is a checklist given by a health care professional as a scale for evaluating therapeutic outcomes and impact on life.
c) The Major Depression Inventory (MDI) is a self-administered assessment for depression that incorporates both the ICD-10 symptoms of depression and the DSM-IV-TR symptoms of major depression.
d) The Center for Epidemiologic Studies Depression Screen (CES-D) measures depressive symptoms in the past week.
e) The Zung Self-Rating Depression Scale assesses four characteristics of depression: the pervasive effect, the physiologic equivalents, psychomotor activities and more.
f) The Inventory of Depressive Symptomatology (IDS) is designed for clinician use, as well as for patient self-administration.
g) The Primary Care Evaluation of Mental Disorders (PRIME-MD) is a practitioner administered screening tool for depression. A self-administered version is also available.
Etiology
The etiology of depression is explained by a combination of three predominant theories focusing on genetic, environmental, and chemical factors. The genetic theory proposes that depression is more common in those with a first degree relative with the same diagnosis. Interestingly, this theory also proposes that genetic factors may be responsible for coping responses to stressors. The environmental theory proposes that circadian rhythms, situational circumstances and poor coping strategies, among many other external factors can produce a depressive state. Finally, chemical theories of depression suggest that imbalances in neurotransmitters and metabolites result in depression; for instance, low serotonin levels promote low levels of norepinephrine, and other monoamine neurotransmitters, while norepinephrine may be involved in regulating serotonin activity, all impacting mood (Barlow 2005, Linner 2004). Yet other chemical theories propose mitochondrial dysfunction as an underlying factor in depression. This article will focus on the biochemical approach, with a particular view to the role of mitochondrial dysfunction and potential effects of acetyl-L-carnitine (ALC) in improving depression by modifying mitochondrial metabolism.
Conventional treatment
There are several categories of anti-depressant therapy, each targeting different mechanisms of action in depression. Classes of antidepressant therapy include:
a) Selective serotonin reuptake inhibitors (SSRIs): These are usually the initial treatment option as they are thought to have fewer side effects. Some examples are fluoxetine (Prozac), paroxetine (Paxil), sertraline (Zoloft) and citalopram (Celexa).
b) Serotonin and norepinephrine reuptake inhibitors (SNRIs): These medications include duloxetine (Cymbalta), venlafaxine (Effexor XR) and Desvenlafaxine (Pristiq).
c) Norepinephrine and dopamine reuptake inhibitors (NDRIs): An example is Bupropion (Wellbutrin).
d) Atypical antidepressants: Some examples of medications that do not fit into the above classifications but are still considered include trazodone (Oleptro), mirtazapine (Remeron) and vilazodone (Viibryd).
e) Tricyclic antidepressants: These are often prescribed if SSRI’s are not effective.
f) Monoamine oxidase inhibitors (MAOI’s) are often given as a last resort. Some examples are tranylcypromine (Parnate) and phenelzine (Nardil). These also have serious side effects if combined with tyramine-containing foods or SSRI’s.
g) Electroconvulsive therapy (ECT) is utilized for depression when other treatments do not work, and often brings faster symptomatic relief than medications. It is administered by passing electrical currents through the brain with the aim of altering levels of neurotransmitters that are thought to affect depression.
h) Counseling and psychotherapy: Psychotherapy such as cognitive behavioural therapy (CBT) attempts to help identify the causes of depression, target negative behaviour/thought patterns and change them.
Many patients wish to avoid medication due to their numerous side effects. These include sexual dysfunction, digestive abnormalities, weight gain, restlessness, headache, sweating, dry mouth, insomnia, tachycardia and constipation (Mayo Clinic 2012). Additionally, a lack of effective relief motivates other patients to seek other alternatives, including naturopathic therapies. Mainstays of naturopathic approaches to depression include but are not limited to eicosapentanoic acid, St. John’s wort, 5-hydroxy-tryptophan (5-HTP), passiflora, vitamin D, and vitamin B12. Some of these can be given alone or in conjunction with conventional treatments. Recently, novel treatment options are being examined which have been successful in other brain disorders, such as nucleotides, citicoline, and L-carnitine.
Potential role of L-carnitine
L-carnitine is an amino acid derivative that is synthesized from lysine and methionine. The carnitine pool in humans consists of L-carnitine (LC) (non-esterified) and acetyl L-carnitine (ALC) (esterified). L-carnitine is found in several supplemental forms: L-Carnitine (LC), acetyl-L-carnitine (ALC), L-carnitine L-tartrate (LCLT), and propionyl-L-carnitine bound to glycine (GPLC). L-carnitine and actyl-L-carnitine accumulate in the brain through active transport across the blood brain barrier (Nałecz 2004). One of the main roles of L-carnitine is facilitating fatty acid oxidation (beta oxidation) and mitochondrial production of ATP; L-carnitine enables the movement of fatty acids from the cytosol into the mitochondria, and the breakdown of fatty acid chains for energy. This biological function includes a process by which carnitine acyl-transferases catalyze the exchange of acyl groups between carnitine and coenzyme A (CoA) (Tong 2004).
Other biological functions of LC and ALC include reducing neurological damage by regulating mitochondrial permeability and protecting against excitotoxicity; increasing cellular insulin sensitivity; and improving smooth muscle function as in the cardiovascular system (DiNicolantonio 2013, Jones 2010, Vidal-Casariego 2013, Zanelli 2005). These functions render L-carnitine potentially useful in conditions including chronic fatigue syndrome, diabetes, hypertension, congestive heart failure, cancer and metabolic syndrome. ALC is already used to improve cognition in those with Alzheimer’s and dementia, with a large body of evidence on this subject (Montgomery 2003). Interestingly, carnitine is now being considered as a novel therapeutic option for other related neurological conditions, such as depression and other psychiatric diagnoses.
In the body, carnitine maintenance and turnover occurs via dietary intake, synthesis, and reabsorption in the kidneys. Carnitine is absorbed by the jejunum, through a sodium dependent transporter (Gross 1986). The levels of absorption are dependent on the dose and source of carnitine. Between 54 – 87% of carnitine is absorbed from food (mainly active transport and can use passive transport), and 14-18% from supplements (passive transport only-diffusion) (Rebouche 2004). Levels of carnitine are categorized as follows: a) large, slow turnover in muscle; and b) small, rapid turnover (in liver, kidney and other organs. At normal dietary intakes, whole-body turnover in humans is 38-119 hours (Rebouche 2004). Serum L-carnitine levels appears to be regulated in the range of 23–73μmol/L, while acetyl-L-Carnitine appears to be in the range of 3–14μmol/L (Minkler 2008).
The common dosage range of carnitine is between 500 to 2000 milligrams per day and varies depending on the intended use (Malaguarnera 2011).
Mechanisms of action in depression
Preclinical studies suggest therapeutic potential for L-carnitine in models of depression, delineating two possible mechanisms: 1) neurochemical and 2) epigenetic.
Smeland et al investigated the effect of ALC on cerebral ATP levels and neurotransmitter metabolism after supplementing mice with ALC for 25 days, providing a daily dose of about 0.5 g/kg, after which neurometabolites were measured in the hippocampus and cortex. ALC treated mice had higher amounts of adenosine nucleotides, phosphocreatine and ratio of phosphocreatine/creatine in the cerebral cortex. There was decreased glucose conversion to lactate, resulting in increased energy and altered monoamine metabolites. The study also showed increased concentrations of the neurotransmitters, norepinephrine in the hippocampus and serotonin in the cortex. These biochemical changes suggest that ALC may have an effect on neurochemical modulation in depression (Smeland 2012).
Other studies in rat and mouse models add further confirmation to this concept at the molecular level. One study showed that ALC had an antidepressant function through epigenetic regulation of type 2 metabotropic glutamate (mGlu2) receptors. Both models showed that ALC increased transcription of the Grm2 gene (encoding for the mGlu2 receptor) in the hippocampus and prefrontal cortex; this was associated with a rapid-onset but long-lasting antidepressant effect observed in rats and mice exposed to chronic unpredictable stress (Nasca 2013). Cuccurazzu et al. showed similar findings (2013).
Clinical evidence
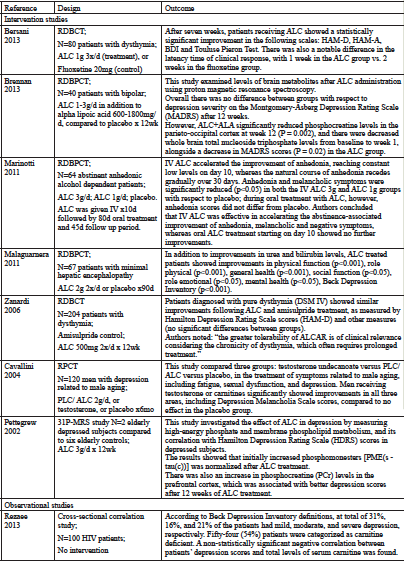
Intriguingly, there is an emerging pool of clinical evidence that supports carnitine as a novel therapeutic option in depression and associated mood disorders. It is thought that bipolar disorder, dysthymia and depression can be attributed in part to chronic mitochondrial dysfunction or imbalances of neurological metabolites (Anglin 2012, Tobe 2013, Torrell 2013). Several studies have attempted to examine this hypothesis by assessing whether patients treated with LC/ALC would experience an improvement in these conditions. Table 1 provides a summary of clinical studies that evaluate the potential benefit of carnitine.
Outcome
Intervention studies
After seven weeks, patients receiving ALC showed a statistically significant improvement in the following scales: HAM-D, HAM-A, BDI and Touluse Pieron Test. There was also a notable difference in the latency time of clinical response, with 1 week in the ALC group vs. 2 weeks in the fluoxetine group.
This study examined levels of brain metabolites after ALC administration using proton magnetic resonance spectroscopy.
Overall there was no difference between groups with respect to depression severity on the Montgomery-Asberg Depression Rating Scale (MADRS) after 12 weeks.
However, ALC+ALA significantly reduced phosphocreatine levels in the parieto-occipital cortex at week 12 (P = 0.002), and there were decreased whole brain total nucleoside triphosphate levels from baseline to week 1, alongside a decrease in MADRS scores (P = 0.02) in the ALC group.
IV ALC accelerated the improvement of anhedonia, reaching constant low levels on day 10, whereas the natural course of anhedonia recedes gradually over 30 days. Anhedonia and melancholic symptoms were significantly reduced (p<0.05) in both the IV ALC 3g and ALC 1g groups with respect to placebo; during oral treatment with ALC, however, anhedonia scores did not differ from placebo. Authors concluded that IV ALC was effective in accelerating the abstinence-associated improvement of anhedonia, melancholic and negative symptoms, whereas oral ALC treatment starting on day 10 showed no further improvements.
In addition to improvements in urea and bilirubin levels, ALC treated patients showed improvements in physical function (p<0.001), role physical (p<0.001), general health (p<0.001), social function (p<0.05), role emotional (p<0.05), mental health (p<0.05), Beck Depression Inventory (p<0.001).
Patients diagnosed with pure dysthymia (DSM IV) showed similar improvements following ALC and amisulpride treatment, as measured by Hamilton Depression Rating Scale scores (HAM-D) and other measures (no significant differences between groups).
Authors noted: “the greater tolerability of ALCAR is of clinical relevance considering the chronicity of dysthymia, which often requires prolonged treatment.”
This study compared three groups: testosterone undecanoate versus PLC/ALC versus placebo, in the treatment of symptoms related to male aging, including fatigue, sexual dysfunction, and depression. Men receiving testosterone or carnitines significantly showed improvements in all three areas, including Depression Melancholia Scale scores, compared to no effect in the placebo group.
This study investigated the effect of ALC in depression by measuring high-energy phosphate and membrane phospholipid metabolism, and its correlation with Hamilton Depression Rating Scale (HDRS) scores in depressed subjects.
The results showed that initially increased phosphomonesters [PME(s – tau(c))] was normalized after ALC treatment.
There was also an increase in phosphocreatine (PCr) levels in the prefrontal cortex, which was associated with better depression scores after 12 weeks of ALC treatment.
Observational studies
According to Beck Depression Inventory definitions, at total of 31%, 16%, and 21% of the patients had mild, moderate, and severe depression, respectively. Fifty-four (54%) patients were categorized as carnitine deficient. A non-statistically significant negative correlation between patients’ depression scores and total levels of serum carnitine was found.
____________________________