Sodium and risk of death: Villain or biomarker?
Introduction A large body of evidence has linked higher intake of sodium with increased risk of chronic disease, most notably stroke and cardiovascular disease, and all cause death (Yang 2011). In recognition of this association, many initiatives have been launched globally to reduce dietary sodium intake most notably though the reduction of sodium content of processed foods (Henney 2010, van Vliet 2011, Webster 2011). This article examines the evidence around sodium as a risk factor for cardiovascular disease and death, drawing on lessons learned from the beta carotene experience.
In Europe, the current leader in sodium reduction initiatives, national programs have been launched in 19 countries, with reductions of up to 25% in sodium content of key foods, yielding reductions of approximately 1-3 g per day in per capita sodium intake (from a baseline of between 8-12 g per day) (Webster 2011). This has been paralleled by documented reductions of up to 60-80% in coronary artery disease and stroke mortality in Finland and Japan respectively: Finland launched a comprehensive cardiovascular disease prevention program in the 1970s and experienced a 65% decrease in cardiovascular mortality by 1995 (He 2009, Laatikainen 2006, Puska 1998, Webster 2011). Likewise, according to the World Action on Salt and Health organization, Japan documented an 80% reduction in mortality in the wake of its salt reduction program (Iso 1999, WASH).
Currently, sodium recommendations for healthy individuals (adequate intake) in Canada and the US are 1500 mg per day (van Vliet 2011), with a tolerable upper limit of 2300 mg (Henney 2010), based on reference values set by the Institute of Medicine in 2005 (Henney 2010). The American Heart Association further recommends a limit of no more than 1500 mg per day (AHA 2011). Actual average daily intake during 2003–2006, estimated by the Institute of Medicine, was 3,614 mg/d (Henney 2010).
Sodium and Risk of Death: Observational Evidence
NHANES III, the third National Health and Nutrition Examination Survey (1988-2006) was a prospective cohort study of a nationally representative sample of US adults; Yang et al studied data from a subset of 12,267 subjects with available data for associations between sodium intake and mortality from all-causes, cardiovascular, and ischemic heart (IHD) disease (2011). Higher sodium intake was associated with significantly increased risk of all-cause mortality, hazard ratio HR 1.20 (95% confidence interval CI 1.03-1.41 per every 1000 mg/d increase), whereas higher potassium intake was associated with lower mortality risk (HR 0.80, 95% CI 0.67-0.94 per 1000 mg/d).
Furthermore, those in the highest quartile sodium-potassium intake ratio compared to the lowest quartile also had significantly elevated risk: HR 1.46 (95% CI 1.27-1.67) for all-cause mortality; HR 1.46 (95% CI 1.11-1.92) for CVD mortality; and HR 2.15 (95% CI 1.48-3.12) for IHD mortality.
Similar findings were reported based on data from the first NHANES study (He 1999). A total of 14,407 subjects were included, and dietary sodium and energy intake were estimated at baseline using a single 24-hour dietary recall method. Among overweight persons with an average energy intake of 7452 kJ (~1781 calories), a 100 mmol higher sodium intake was associated with a 32% increase (relative risk RR 1.32, 95% CI 1.07-1.64, P = .01) in stroke incidence; an 89% increase (RR 1.89, 95% CI 1.31-2.74, P<.001) in stroke mortality; a 61% increase (RR 1.61, 95% CI 1.32-1.96, P<.001) in cardiovascular disease mortality; and a 39% increase (RR 1.39, 95% CI, 1.23-1.58, P<.001) in mortality from all causes. However, in this study dietary sodium intake was not significantly associated with cardiovascular disease risk in non-overweight persons.
Such studies have also been conducted in Japan. Nagata et al (2004) investigated sodium intake and risk of death from stroke among 13,355 men and 15 724 women in Takayama City, Japan. In men, the highest compared with the lowest tertile of sodium intake was significantly positively associated with death from stroke (total from ischemic and hemorrhagic) after controlling for covariates, HR 2.33 ( 95% CI 1.23 -4.45). Significant positive associations were also observed between sodium intake and death from ischemic stroke (HR 3.22, 95% CI 1.22-8.53) as well as death from intracerebral hemorrhage (HR 3.85, 95% CI 1.16 -12.7). In women, a non significant association between sodium intake and death from stroke was found : HR 1.70, 95% CI, 0.96 -3.02 and HR 2.10, 95% CI 0.96 – 4.62, respectively.
Takachi et al (2010) investigated the relationship between consumption of sodium and cancer and cardiovascular disease. Between 1995-1998, a total of 77,500 men and women aged 45-74 years were enrolled, and followed until the end of 2004; 4476 cases of cancer and 2066 cases of cardiovascular disease (CVD) were identified. Results showed that higher consumption of sodium was associated with a higher risk of CVD but not with the risk of total cancer: multivariate HR for the highest compared with lowest quintiles of intake were 1.19 (95% CI 1.01-1.40; P trend 0.06) for CVD and 1.04 (95% CI 0.93-1.16; P trend: 0.63) for total cancer.
Umesawa et al (Umesawa 2008) report a Japanese population study conducted between 1988 -1990 among 58,730 Japanese subjects aged 40-79 y with no history of stroke, coronary heart disease, or cancer. After 745,161 person-years of follow-up, 986 deaths from stroke (153 subarachnoid hemorrhages, 227 intraparenchymal hemorrhages, and 510 ischemic strokes) and 424 deaths from coronary heart disease were documented. Sodium intake was associated with mortality from total stroke, ischemic stroke, and total cardiovascular disease. The multivariable HR for the highest versus the lowest quintiles of sodium intake after adjustment for age, sex, and cardiovascular disease risk factors was 1.55 (95% CI 1.21- 2.00; P trend < 0.001) for total stroke; 2.04 (95% CI 1.41- 2.94; P for trend < 0.001) for ischemic stroke; and 1.42 (95% CI 1.20- 1.69; P for trend < 0.001) for total cardiovascular disease. As with NHANES III, this study also found an inverse relationship between potassium intake and risk of death.
Strazzullo et al conducted a meta-analysis of 19 prospective cohorts examining the association between sodium intake and risk of stroke or cardiovascular disease (2009). The analysis included 177,025 participants with follow-up between 3.5-19 years, and over 11,000 vascular events. Higher salt intake was associated with increased risk of stroke (relative risk RR 1.23, 95% CI 1.06 -1.43; P=0.007) and cardiovascular disease (1.14, 0.99 – 1.32; P=0.07). For cardiovascular disease, sensitivity analysis showed that the exclusion of a single study led to a significant result: RR 1.17 (1.02 – 1.34; P=0.02). The associations observed were greater the larger the difference in sodium intake and the longer the follow-up. The authors concluded: “high salt intake is associated with significantly increased risk of stroke and total cardiovascular disease. Because of imprecision in measurement of salt intake, these effect sizes are likely to be underestimated. These results support the role of a substantial population reduction in salt intake for the prevention of cardiovascular disease.”
The Beta Carotene Experience: A Harsh Lesson Learned in the Concept of Biomarker With the emergence of well- constructed theories of the role of oxidant stress in the initiation of atherosclerosis (Ross 1993) and cancer (Cerutti 1991) came an explosion of research into the potential for a handful of antioxidant nutrients to prevent and treat “the big two” chronic degenerative diseases. The landmark Basel study for many confirmed suspicions of the powerful ability of antioxidants to prevent such diseases; in an observational cohort of 4858 men followed for 12 years, Basel researchers found individuals in the lowest quintile of plasma beta carotene were at a 60% increased risk of developing cancer relative to individuals in the highest quintile of plasma beta carotene (Stahelin 1991). The stage was set for large, multicentre, randomized control trials of intervention with antioxidant nutrients for the prevention and treatment of heart disease and cancer.
An understatement is to call the outcomes of these intervention trials “disappointing”. Cochrane recently summarized 67 trials of antioxidant nutrients with 232,550 participants collectively (Bjelakovic 2008). The reviewers found a 16% increased risk in all cause death from vitamin A supplementation (RR 1.16, 95% CI 1.10 to 1.24), a 7% increased risk in all cause death from beta carotene supplementation (RR 1.07, 95% CI 1.02-1.11), and a 4% increased risk in all cause death from vitamin E supplementation (RR 1.04, 95% CI 0.94-1.20).
The mechanistic basis upon which oxidant stress contributes to initiation and progression of heart disease and cancer remains sound. So what accounts for the lack of benefit, and marked detriment, from intervention with this selection of antioxidant nutrients?
The concept of biomarker was quickly identified. Plasma determination of any one antioxidant nutrient does a poor job of providing insight into the biological impact of that specific nutrient. Instead, plasma determination of an antioxidant serves as an accurate biomarker of exposure to fruit and vegetables. The collective conscious of nutritional scientists was quick to incorporate this important fact in interpretation of relevant evidence moving forward, and what has emerged is a new era of research into whole foods and intervention trials focused on diet modification as opposed to single nutrient intervention
Modern trials that intervene with diet counseling have come to rely on plasma determination of beta carotene for an important task; serving as an accurate marker of fruit and vegetable intake, assessment of plasma beta carotene objectively confirms compliance or lack thereof with diet instructions of the trial. A simple example is the use of beta carotene assessment by WHEL investigators, an intervention trial in 3088 breast cancer survivors with a mean follow- up of 7.3 years examining the impact of diet and lifestyle counseling on cancer- free survival, invasive breast cancer events, and all cause mortality (Pierce 2007).
Is Sodium a Biomarker?
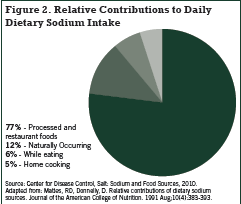
Very little sodium occurs naturally in foods, and it is widely recognized that processed foods are the major source of sodium in the Standard American Diet (SAD). In addition to sodium chloride, table salt, sodium may be added to food as the flavour enhancer monosodium glutamate, sodium benzoate, sodium bicarbonate, sodium citrate, sodium nitrite, and sodium acid pyrophosphate (Fischer 2009). The Canadian Stroke Network’s website www.sodium101.ca states that over 70% of dietary sodium comes from processed foods (Figure 1).
In view of the beta carotene experience, the undeniable link between sodium intake and risk of death, and the fact that upwards of 70% of dietary sodium originates from processed foods, we posit that sodium intake may in fact function as a biomarker of the effect of processed foods. Since processed foods represent by far the predominant source of dietary sodium, the link between sodium intake and risk of death in these population studies may actually represent the relationship between intake of processed foods and death/ disease. Besides sodium, processed foods contain several other harmful substances such as nitrates, saturated and trans fats, (sodium) benzoate, MSG, many other preservatives and flavoring agents, as well as possible byproducts of processing that are as yet unidentified. Processed foods are also typically quite calorie-dense, thus promoting obesity and its health consequences. Furthermore, high intake of processed foods is typically accompanied by low intake of fruit and vegetables, the primary source of dietary potassium, and this may help explain the additional predictive power of sodium: potassium ratio and mortality, as shown in the studies cited above.
A recent Cochrane review examining the effect of intervention with low-sodium diets lends further support to this hypothesis (Taylor 2011). After pooling seven RCTs including 6489 participants with follow up of between seven months to 12.7 years, Taylor et al found no “strong benefit” from sodium restriction on mortality and cardiovascular morbidity (2011). Among patients who were normotensive, relative risk for all cause mortality at the end of the trial was RR 0.67 (95% CI 0.40-1.12, 60 deaths), and after the longest follow up, risk was RR 0.90 (95% CI 0.58-1.40, 79 deaths).
Among hypertensive subjects, risk at the end of trial was RR 0.97 (95% CI 0.83-1.13, 513 deaths), and after the longest follow up, risk was RR 0.96 (95% CI 0.83-1.11, 565 deaths) showing no strong evidence of any effect of salt reduction. Cardiovascular morbidity in people with normal blood pressure or raised blood pressure at baseline showed no strong evidence of benefit from salt restriction. Conversely, salt restriction increased the risk of all-cause death in those with congestive heart failure (end of trial RR 2.59, 95% 1.04-6.44, 21 deaths).
Taylor’s paper raised a great deal of controversy. He et al (2011) have argued that the pooled analysis had insufficient statistical power, and that this is the reason for null results. Nonetheless, it raises important questions as to the true role of sodium as an agent of cardiovascular disease.
Exceptions
Our hypothesis pertains to the population as a whole. We wish to point out that reduced sodium intake is a critical factor in a subset of hypertensive patients who are sodium-sensitive, as well as in patients with chronic renal disease.
Conclusion
There is a well- established link between dietary sodium intake and risk of death, in particular cardiovascular mortality. As illustrated by the beta carotene experience, however, sodium may function as a biomarker rather than as the causative agent. Since the major source of dietary sodium in the Standard American Diet is processed foods, measurement of sodium intake may actually reflect consumption of processed foods and the complex combination of food chemicals and/ byproducts contained therein. We suggest that sodium has been unfairly vilified as an agent of harm, and that in fact harm is due to the complex, disease-promoting constituents of processed foods.
References
AHA (American Heart Association). 2011 Dietary and Lifestyle Recommendations. Updated 2011. http://www.heart.org/HEARTORG/GettingHealthy/NutritionCenter/HealthyDietGoals/Dictionary-of-Nutrition_UCM_305855_Article.jsp. Accessed 8 October 2011.
Bjelakovic G, Nikolova D, Gluud LL, Simonetti RG, Gluud C. Antioxidant supplements for prevention of mortality in healthy participants and patients with various diseases. Cochrane Database Syst Rev. 2008 Apr 16;(2):CD007176.
Brown IJ, Tzoulaki I, Candeias V, Elliott P. Salt intakes around the world: implications for public health. Int J Epidemiol. 2009 Jun;38(3):791-813.
Cerutti PA, Trump BF. Inflammation and oxidative stress in carcinogenesis. Cancer Cells. 1991 Jan;3(1):1-7.
CDC (Center for Disease Control). Salt: Sodium and Food Sources. Updated 10 February 2010. http://www.cdc.gov/salt/food.htm Accessed 8 October 2011.
Fischer PW, Vigneault M, Huang R, Arvaniti K, Roach P. Sodium food sources in the Canadian diet. Appl Physiol Nutr Metab. 2009 Oct;34(5):884-92.
He J, Ogden LG, Vupputuri S, Bazzano LA, Loria C, Whelton PK. Dietary sodium intake and subsequent risk of cardiovascular disease in overweight adults. JAMA. 1999 Dec 1;282(21):2027-34.
He FJ, MacGregor GA. A comprehensive review on salt and health and current experience of worldwide salt reduction programmes. J Hum Hypertens 2009; 23:363 – 384.
He FJ, MacGregor GA. Salt reduction lowers cardiovascular risk: meta-analysis of outcome trials. Lancet. 2011 Jul 30;378(9789):380-2.
Institute of Medicine (US) Committee on Strategies to Reduce Sodium Intake; Henney JE, Taylor CL, Boon CS, editors. Strategies to Reduce Sodium Intake in the United States. Washington (DC): National Academies Press (US); 2010.
Iso H, Shimamoto T, Yokota K, Ohki M, Sankai T, Kudo M, Harada M, Wakabayashi Y, Inagawa M, Kitamura A, Sato S, Imano H, Iida M, Komachi Y. [Changes in 24-hour urinary excretion of sodium and potassium in a community-based heath education program on salt reduction]. Nihon Koshu Eisei Zasshi. 1999 Oct;46(10):894-903.
Laatikainen T, Pietinen P, Valsta L, Sundvall J, Reinivuo H, Tuomilehto J. Sodium in the Finnish diet: 20-year trends in urinary sodium excretion among the adult population. Eur J Clin Nutr 2006; 60:965 – 970.
Mattes, RD, Donnelly, D. Relative contributions of dietary sodium sources. Journal of the American College of Nutrition. 1991 Aug;10(4):383-393.
Nagata C, Takatsuka N, Shimizu N, Shimizu H. Sodium intake and risk of death from stroke in Japanese men and women. Stroke. 2004 Jul;35(7):1543-7.
Ni Mhurchu C, Capelin C, Dunford EK, Webster JL, Neal BC, Jebb SA. Sodium content of processed foods in the United Kingdom: analysis of 44,000 foods purchased by 21,000 households. Am J Clin Nutr. 2011 Mar;93(3):594-600.
Pierce JP, Natarajan L, Caan BJ, Parker BA, Greenberg ER, Flatt SW, Rock CL, Kealey S, Al-Delaimy WK, Bardwell WA, Carlson RW, Emond JA, Faerber S, Gold EB, Hajek RA, Hollenbach K, Jones LA, Karanja N, Madlensky L, Marshall J, Newman VA, Ritenbaugh C, Thomson CA, Wasserman L, Stefanick ML. Influence of a diet very high in vegetables, fruit, and fiber and low in fat on prognosis following treatment for breast cancer: the Women’s Healthy Eating and Living (WHEL) randomized trial. JAMA. 2007 Jul 18;298(3):289-98.
Puska P, Vartiainen E, Tuomilehto J, Salomaa V, Nissinen A. Changes in premature deaths in Finland: successful long-term prevention of cardiovascular diseases. Bull World Health Organization 1998; 76:419–425.
Ross R. The pathogenesis of atherosclerosis: a perspective for the 1990s. Nature. 1993 Apr 29;362(6423):801-9.
Stahelin HB, Gey KF, Eichholzer M, Ludin E, Bernasconi F, Thurneysen J, Brubacher G. Plasma antioxidant vitamins and subsequent cancer mortality in the 12-year follow-up of the prospective Basel Study. Am J Epidemiol. 1991 Apr 15;133(8):766-75.
Strazzullo P, D’Elia L, Kandala NB, Cappuccio FP. Salt intake, stroke, and cardiovascular disease: meta-analysis of prospective studies. BMJ. 2009 Nov 24;339:b4567.
Takachi R, Inoue M, Shimazu T, Sasazuki S, Ishihara J, Sawada N, Yamaji T, Iwasaki M, Iso H, Tsubono Y, Tsugane S; Japan Public Health Center-based Prospective Study Group. Consumption of sodium and salted foods in relation to cancer and cardiovascular disease: the Japan Public Health Center-based Prospective Study. Am J Clin Nutr. 2010 Feb;91(2):456-64.
Taylor RS, Ashton KE, Moxham T, Hooper L, Ebrahim S. Reduced dietary salt for the prevention of cardiovascular disease. Cochrane Database Syst Rev. 2011 Jul 6;(7):CD009217.
Umesawa M, Iso H, Date C, Yamamoto A, Toyoshima H, Watanabe Y, Kikuchi S, Koizumi A, Kondo T, Inaba Y, Tanabe N, Tamakoshi A; JACC Study Group. Relations between dietary sodium and potassium intakes and mortality from cardiovascular disease: the Japan Collaborative Cohort Study for Evaluation of Cancer Risks. Am J Clin Nutr. 2008 Jul;88(1):195-202.
Van Vliet BN, Campbell NR; Canadian Hypertension Education Program. Efforts to reduce sodium intake in Canada: why, what, and when? Can J Cardiol. 2011 Jul-Aug;27(4):437-45.
Webster JL, Dunford EK, Hawkes C, Neal BC. Salt reduction initiatives around the world. J Hypertens. 2011 Jun;29(6):1043-50.
WASH (World Action on Salt and Health). Evidence: Population Studies http:// www.worldactiononsalt.com. Accessed 8 October 2011.
Yang Q, Liu T, Kuklina EV, Flanders WD, Hong Y, Gillespie C, Chang MH, Gwinn M, Dowling N, Khoury MJ, Hu FB. Sodium and potassium intake and mortality among US adults: prospective data from the Third National Health and Nutrition Examination Survey. Arch Intern Med. 2011 Jul 11;171(13):1183-91. 078-