The organization SafeMinds has recently demanded a long-overdue independent review of vaccine/autism research for data manipulation, scientific misconduct, and conflicts of interest. The demand follows upon the recent indictment on April 13th of Poul Thorsen, the principal coordinator of multiple studies funded by the Centers for Disease Control and Prevention (CDC) that have been used to deny a vaccine/autism link on 13 counts of fraud and nine counts of money- laundering. The charges relate to funding for work he conducted for the CDC, which claimed to disprove associations between the mercury-based vaccine preservative, thimerosal, and increased rates of autism. SafeMinds first voiced concerns in 2003 regarding a series of epidemiology based data from Denmark’s registry for psychiatric research and under the jurisdiction of Thorsen that provided the basis for the Institute of Medicine’s claim that there was no association between thimerosal and autism. That claim has been responsible for the continued questionable use of mercury in influenza vaccines in the United States and infant vaccines around the world. SafeMinds accessed the data registry and reported that a large percentage of diagnosed autism cases are lost from the Danish registry each year and that most of those lost cases were older children. Since the studies were based on finding fewer older thimerosal-exposed children than younger unexposed children, the validity of their conclusion exonerating thimerosal in autism was questionable and likely a result of missing records rather than true lower incidence rates among the exposed group. “The quality of this epidemiological research has always been questionable,” states Sallie Bernard, SafeMinds president. “Many biological studies support a link between mercury and autism, but these Danish studies have been used to suppress further research into thimerosal. With clear evidence of Dr. Thorsen’s lack of ethics, it is imperative to reopen this investigation.” Further background information on these studies, the charges against Dr. Thorsen, and documents obtained through the Freedom of Information Act that support SafeMinds’ concerns are available at, www.safeminds.org.
PASCOE Marks 50 Years of Research
Approximately 200 invited guests from science and industry, associations and government attended PASCOE’s 50th anniversary celebration on March 12th at the Justus-Liebig-University Giessen, Germany. The guest list included Professor Barbara Sickmueller of the Federal Association of the German Pharmaceutical Industry, Dr. Rose Schraitle of the Federation of German Pharmaceutical Manufacturers, Prof. Karin Kraft, Head of Chair of Natural Medicine at the University of Rostock, and assistant professor Dr. Werner Knöss of the Federal Institute for Drugs and Medical Devices (BfArM). The event was hosted by PASCOE Natural Medicine, who started their anniversary of 50 years of documented research with this event. Since the founding of PASCOE Natural Medicine in 1896, the family- owned manufacturer has been passionate for homeopathy and herbal medicine. Conscious of tradition and with an eye on innovation, the approximately 200 medicines have been investigated for efficacy, tolerability and safety. 50 years have now gone by since the foundation of the research department of the company PASCOE pharmaceutical preparations GmbH in 1961. The result of tireless efforts are impressive: 19 clinical studies, 86 medical data collection reports, 193 progress reports and 56 observational studies provided data and insights of over three million patients.
Infl ammatory Bowel Disease
Infl ammatory Bowel Disease
A sampling of targeted integrative therapies
Infl ammatory Bowel Diseases (IBD) such as Crohn’s disease (CD) and ulcerative colitis (UC) are chronic, relapsing-remitting infl ammatory diseases with several hallmarks of dysfunction, including a breakdown in intestinal barrier function and intestinal permeability, unchecked chronic infl ammation, and an exaggerated immune response characterized by imbalanced anti-infl ammatory and proinfl ammatory cytokines. Proinfl ammatory cytokines such as IL-1, IL-6, IL-12, IFN-gamma and TNF-alpha are increased with lowered anti-infl ammatory and regulatory cytokines like IL-10 and TGF-beta. When diagnosed with IBD later in life, aside from regular check-ups with gastroenterologists between procedures and to monitor effi cacy of pharmaceutical interventions, many adults have questions about their specifi c needs and are looking for additional support. However, many patients are reluctant to seek out integrative healthcare as they are concerned they will be encouraged not to take their medications which are helping to reduce their symptoms and provide quality of life. Naturopathic doctors have a unique opportunity to support patients with IBD by using the most appropriate treatments during diff erent stages of the disease process.
Supporting Intestinal Barrier Function and Intestinal Permeability
1.Prebiotic and Probiotic support Prebiotic supports (i.e. oligofructosaccharides and/or inulin found in chicory, wheat, onions, bananas) encourage growth of benefi cial bacteria and have the capability of skewing directional growth of probiotic strains which confer specifi c host benefi ts (Bouhnik 2004, Langlands 2004, Looijer-van Langen 2009). Some of these host benefi ts include providing energy sources for intestinal bacteria to ferment into short chain fatty acids (SCFA) (Looijer-van Langen 2009). Short chain fatty acids (SCFA’s) such as butyrate are a source of energy for colonocytes and can regenerate mucosa, as well as having the capacity to reduce infl ammation through enhancement of anti-infl ammatory cytokines such as IFN-gamma and NF-kB which are down-regulated in both Crohn’s disease and ulcerative colitis (Looijer-van Langen 2009). In a group of 19 patients with active UC (mild-moderate disease severity) administered inulin for 2 weeks in addition to the 3g/daily of mesalamine, a signifi cant reduction in calprotectin (a fecal infl ammatory marker increased in IBD) compared with the placebo control group was observed (Casellas 2007, Konikoff 2006). UC in general shows more signifi cant benefi t from prebiotic support compared with CD.
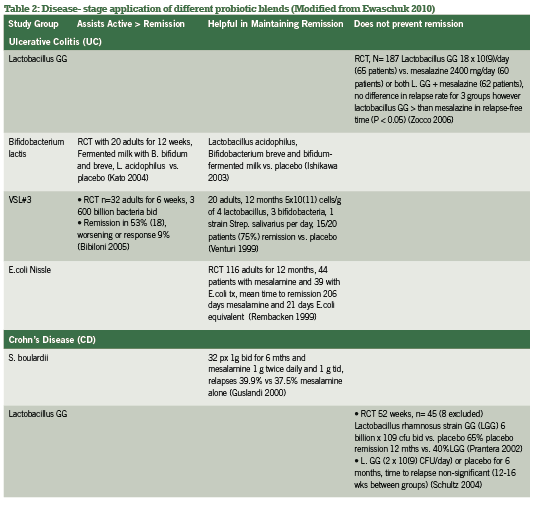
Probiotic support is widely considered a gold-standard intervention in both conventional and integrative medicine for both active and remission phases of IBD, with benefi ts of the most popular investigated strains summarized in Table 1. Current research has explored specifi c strains and their ability to infl uence active vs. remission phases of IBD, as summarized in Table 2. Of research produced to date, administering the proper probiotic strain(s) is crucial to seeing rapid improvements in bowel mucosa, stool quality and frequency, as well as reducing systemic infl ammation (Looijer-van Langen 2009). e probiotic VSL#3, which provides a high- dose milieu of bacteria, is more appropriate in IBD disease remission than is S. boulardii, which shows fantastic response for diarrhea-control and initiating remission (Ewaschuk 2010).
A recent meta-analysis comparing multiple studies and strains contrasted these conclusions for CD specifically, finding significance for E.coli and S. boulardii but not for lactobacillus strains (Rahimi 2008). Hence, screening for the proper strains by looking at disease activity and subjective symptomatology is the most useful strategy in selection appropriate probiotic blends for IBD management.
2. Amino Acid Supplementation Increased intestinal permeability can be predictive of how soon an individual relapses to active disease. IFN-gamma and TNF-a, central mediators of intestinal inflammatory diseases, induce intestinal epithelial barrier dysfunction (MacDonald 1990, Wyatt 2004). L-glutamine, the major feeding source for enterocytes, has been suggested to have antioxidant potential by reducing nitrous oxide (NO) as well as restoring loose connections between tight junctions of colonocytes (Coeffier 2010, Grozswitz 2009). Animal-models of IBD induced via dextran sulfate sodium or acetic acid show promise with reduction of proinflammatory cytokines (TNF-alpha and IL-8) and intestinal damage in the presence of L-glutamine consumption (Coeffier 2010). However, when used in human studies, glutamine supplementation does not show the same benefits in symptomatic reduction of disease activity through subjective scoring of the disease activity indices (CDAI) (Akobeng 2007, Den Hond 1999, Ockenga 2005). Study designs could use some improvements both in consistency of dosages administered as well as methodology of testing. Biopsies of inflamed gastrointestinal mucosa and measurements of cytokine levels are for the most part lacking in human trials post-glutamine consumption; perhaps it is in the microscopic changes of the intestinal barrier where the impact of L- glutamine supplementation will be observed (Coeffier 2005, Coeffier 2010)
Reducing Chronic Inflammation
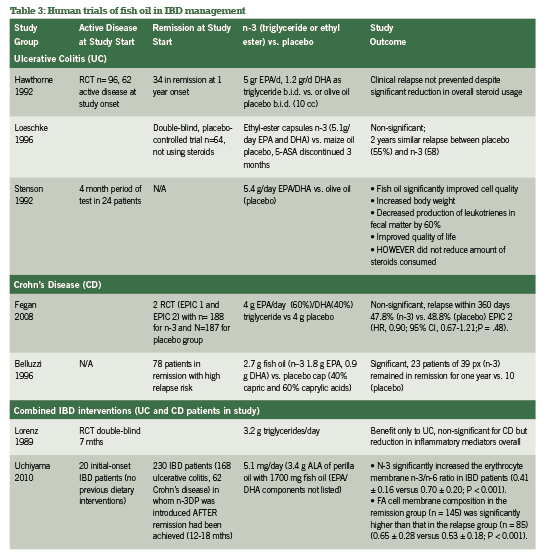
A. N-3 Fish oil The inflammation-reducing capacity of high-potency n-3 fish oils via the arachidonic acid pathways are well- publicized, acting as do current pharmaceutical supports for IBD including 5-ASA (Belluzzi 2000). While patients with IBD have steatorrhea and fat-soluble vitamin malabsorption, fish oil supplementation should be a top priority in integrative support of IBD (Hartman 2009). A selection of research groups in comparison of N-3, N-6 and N-9 oil usage for IBD across 10 years has been quickly summarized in Table 3 to further confirm the absolute necessity of n-3 fish oil for IBD support.
B. Curcuma longa or Curcumin Curcumin shows incredible promise from both preclinical models and recent human trials in IBD. It has demonstrated the ability to influence the arachidonic acid pathway and downregulate chemokine production (Arafa 2009, Goel 2007, Jagetia 2007). Neutrophil motility in IBD is correlated with disease severity with higher rates of neutrophil migration leading to reduced epithelial barrier function (Larmonier 2011); curcumin administration in murine models of IBD has demonstrated reduced neutrophil motility as well as reduced NF-kappaB as has led to further exploration in human IBD (Salh 2003).
Ex vivo¸biopsies from colonic mucosa and myofibroblasts from children and adults with active IBD exposed to curcumin in culture has demonstrated IL-10 production, reduction of IL-1B activity, and further reduction of cell signaling molecules in inflammatory pathways (Epstein 2009).
An RCT involving 89 patients with UC in remission were exposed to either 2 g/day of curcumin with sulfasalazine or placebo with sulfasalazine, with significant findings for delay of relapse in patients given curcumin (4.65%) as treatment compared with placebo (20.51%; p=0.040) (Hanai 2006). Further studies need to be completed, in addition to answering questions surrounding curcumins oral bioavailability (Marczylo 2007).
Oxidative stress
Reactive oxygen species and radical nitrogen metabolites accumulate rapidly during intestinal inflammation in patients with IBD, of which antioxidant support can be invaluable (Aghdassi 2003, Najafzadeh 2009). In addition to vitamin C, quercetin shows promise in animal models of IBD and has been used in combination with fish oil to restore glutathione concentration and to reduce COX-2 more significantly than with just fish oil alone (Camuesco 2006).
Dietary Support
As integrative healthcare providers, we concern ourselves with elimination of irritating proinflammatory foods such as dairy products (primarily cow’s milk), wheat gluten, peanuts, citrus fruits, fish and shellfish, synthetic and excessive sugar, and soy products. The “Western diet” high in animal meats, dairy, and sugars has been implicated in the increase in prevalence of both UC and CD in a comparison between 1990 and 2007 of reduced microbes in the intestines and food consumption (Asakura 2008).
Along the same vein, lactose intolerance and lactose malabsorption have a strong amount of research support with correlations to worsening IBD, especially CD compared with controls (Szilagyi 1998). Lactulose breath tests are often used to confirm this, however as most patients with IBD have imbalanced or higher than normal quantities of bowel flora, results prove to be inconsistent across many studies (Szilagyi 1998). Milk allergy and/or IgE antibodies to cow’s milk proteins are not often positive, which adds to the confusion where patients feel well avoiding dairy products but blood titres do not confirm the subjective improvements (Knoflach 1987, Mishkin 1997). Even if and when allergy results are positive and patients avoid offending foods for a period of time through elimination, intestinal permeability may still be increased as was demonstrated in a group of patients with IBD who avoided allergen exposure for six months yet still had high lactulose/mannitol ratios (Wyatt 1993). Intestinal permability can be an independent issue that is not easily solved with food avoidance.
In addition, replacement or substitution for these caloric losses in food avoidance is an issue as there is a need to nourish patients with IBD. Prednisone as an anti-inflammatory support in some patients with active IBD leaches calcium from the bones, and vitamin D is not only essential in directing calcium to the bones but to support proinflammatory cytokines such as IL-1 and TNF-alpha suppression in IBD and in colorectal cancer prevention (Raman 2011). Among patients in remission from CD, 50% were shown to have low plasma concentrations of vitamin C (84%), copper (84%), niacin (77%), and zinc (65%) in addition to malabsorption of fat and fat-soluble vitamins (Filippi 2006). Exploring all options including screening for celiac disease, lactose intolerance, and multiple food allergies would be prudent rather than strictly eliminating foods without testing for such intolerances, thus taking each patient’s care as a unique situation despite general IBD trends.
Conclusion
Integrative healthcare providers play a unique supportive role in IBD. Recognizing the current disease state (active vs remission), the extent of inflammation, bowel flora status, and supporting processes of oxidative stress can improve quality of life for patients. Patients need the reassurance that they will not be in a competition or battle between their integrative healthcare provider and conventional supports (GI specialist, MD) but that we can provide improved quality of life by working as part of an integrative health care team.
References:
Aghdassi E, Wendland BE, Steinhart AH, Wolman SL, Jeejeebhoy K, Allard JP. Antioxidant vitamin supplementation in Crohn’s disease decreases oxidative stress: a randomized controlled trial. Am J Gastroenterol 2003; 98(2): 348-53.
Akobeng AK, Thomas AG. Enteral nutrition for maintenance of remission in Crohn’s disease. Cochrane Database Syst Rev. 2007: CD005984.
Arafa HM, Hemeida RA, El-Bahrawy AL, Hamada FM (2009). Prophylactic role of curcumin in dextran sulfate sodium (DSS)-induced ulcerative colitis murine model. Food Chem Toxicol ; 47(6):1311-7.
Asakura et al. Is there a link between food and intestinal microbes and the occurrence of Crohn’s disease and ulcerative colitis? Journal of Gastroenterology and Hepatology. 2008; 23: 1794–1801.
Belluzzi A, Boschi S, Brignola C, Munarini A, Cariani G, and Miglio F. Polyunsaturated fatty acids and inflammatory bowel disease. Am J Clin Nutr 2000;71(suppl):339S–42S.
Belluzzi A, Brignola C, Campieri M, et al. Effect of an enteric-coated fish oil preparation on relapses in Crohn’s disease. N Engl J Med 1996; 334: 1557-616.
Bibiloni R, Fedorak RN, Tannock GW, Madsen KL, Gionchetti P, Campieri M, De SC, Sartor RB. VSL#3 probiotic-mixture induces remission in patients with active ulcerative colitis. Am J Gastroenterol. 2005;100:1539–46.
Bouhnik Y, Raskine L, Simoneau G, et al. The capacity of nondigestible carbohydrates to simulate fecal bifidobacteria in healthy humans: a double-blind, randomized, placebo-controlled, parallel-group, dose-response relation study. Am J Clin Nutr 2004, 80: 1658-1664.
Camuesco D, Galvez J, Nieto A, Comalada M, Rodriguez-Cabezas ME, Concha A, Xaus J, Zarzuelo A. Dietary olive oil supplemented with fish oil rich in EPA and DHA (n-3) polyunsaturated fatty acids, attenuates colonic inflammation in rats with DSS-induced colitis. J Nutr 2005; 135(4): 687-94.
Casellas F, Borruel N, Torrrejon A, et al. Oral oligofructose-enriched inulin supplementation in acute ulcerative colitis is well tolerated and associated with lowered faecal calprotectin. Aliment Pharmacol Ther. 2007;25:1061-1067.
Coeffier M, Marion-Letellier R, Dechelotte P. Potential for Amino Acids Supplementation During Inflammatory Bowel Diseases. Inflamm Bowel Dis 2010; 16:518-524.
Coëffier M, Hecketsweiler B, Hecketsweiler P, Déchelotte P. Effect of glutamine on water and sodium absorption in human jejunum at baseline and during PGE-1 induced secretion. J Appl Physiol. 2005 Jun;98(6):2163-8.Epub 2005 Jan 20.
Den Hond E, Hiele M, Peeters M, et al. Effect of long-term oral glutamine supplements on small intestinal permeability in patients with Crohn’s disease. JPEN 1999;23:7-11.
Epstein J, Docena G, MacDonald TT, Sanderson IR. Curcumin suppresses p38 mitogenactivated protein kinase activation, reduces IL-1beta and matrix metalloproteinase-3 and enhances IL-10 in the mucosa of children and adults with inflammatory bowel disease. Br J Nutr. 2010 Mar;103(6):824-32.
Ewaschuk JB and Dieleman LA . Prebiotics and probiotics in chronic inflammatory bowel diseases. World J Gastroenterol 2006; 12(37): 5941-5950.
Feagan BG, Sandborn WJ, Mittmann U, Bar-Meir S, D’Haens G, Bradette M, Cohen A, Dallaire C, Ponich TP, McDonald JW, Hébuterne X, Paré P, Klvana P, Niv Y, Ardizzone S,Alexeeva O, Rostom A, Kiudelis G, Spleiss J, Gilgen D, Vandervoort MK, Wong CJ, Zou GY, Donner A, Rutgeerts P. Omega-3 free fatty acids for the maintenance of remission in Crohn disease: the EPIC Randomized Controlled Trials. JAMA. 2008 Apr 9;299(14):1690-7.
Filippi J, Al-Jaouni R, Wiroth JB, Hebuterne X, and Schneider S. Nutritional Deficiencies in Patients With Crohn’s Disease in Remission. Inflamm Bowel Dis 2006;12:185Y19.
Goel A, Kunnumakkara AB, Aggarwal BB. Curcumin as “curecumin”: from kitchen to clinic. Biochem Pharmacol 2008;75:787-809.
Groschwitz KR and Hogan SP. Intestinal Barrier Function: Molecular Function and Disease Pathogenesis. J Allergy Clin Immunol 2009;124:3-20.
Guslandi M, Giollo P, Testoni P. A pilot trial of Saccharomyces boulardii in ulcerative colitis. Eur J Gastroenterol Hepatol 2003; 15:697-8.
Hanai H, Lida T, Takeuchi K, Watanabe F, Maruyama Y, Andoh A et al. Curcumin maintenance therapy for ulcerative colitis: randomized, multicenter, double-blind, placebo-controlled trial. Clin Gastroenterol Hepatol 2006; 4(12): 1502-6.
Hartman C, Eliakim R, Shamir R. Nutritional status and nutritional therapy in inflammatory bowel diseases. World J Gastroenterol 2009; 15(21): 2570-8.
Hasunuma O, Kurihara R, Iwasaki A, Arakawa Y. Randomized placebo-controlled trial assessing the effect of bifidobacteria-fermented milk on active ulcerative colitis. Aliment Pharmacol Ther 2004; 20: 1133-1141.
Hawthorne AB, Daneshmend TK, Hawkey CJ et al. Treatment of ulcerative colitis with fish oil supplementation: a prospective 12 month randomized controlled trial. Gut 1992, 22;922-8.
Ishikawa H, Akedo I, Umesaki Y, Tanaka R, Imaoka A, Otani T. Randomized controlled trial of the effect of bifidobacteria-fermented milk on ulcerative colitis. J Am Coll Nutr 2003; 22(1): 56-63.
Jagetia GC and Aggarwal BB. “Spicing up” of the immune system by curcumin. J Clin Immunol. 2007;27: 19-35.
Kato K,Mizuno S,Umesaki Y ,Ishii Y ,SugitaniM,Imaoka A, Otsuka M, Knoflach P, Park BH, Cunningham R, et al. Serum antibodies to cows’ milk proteins in ulcerative colitis and Crohn’s disease. Gastroenterology 1987; 92:479-85.
Konikoff MR, Denson LA. Role of fecal calprotectin as a biomarker of intestinal inflammation in inflammatory bowel disease. Inflammatory Bowel Dis 2006; 12: 524-34.
Langlands SJ, Hopkins MJ, Coleman N, et al. Prebiotic carbohydrates modify the mucosa associated microflora of the human large bowel. Gut 2004;53:1610-1616.
Larmonier CB, Midura-Kiela MT, Ramalingam R, Laubitz D, Janikashvili N, Larmonier N, Ghishan FK, Kiela PR. Modulation of neutrophil motility by curcumin: implications for inflammatory bowel disease. Inflamm Bowel Dis 2011; 17(2) 503-15.
Loeschke K, Ueberschaer B, Pietsch A, Gruber E, Ewe K, Wiebecke B, Heldwein W, Lorenz R. n-3 fatty acids only delay early relapse of ulcerative colitis in remission. Dig Dis Sci 1996; 41: 2087-2094.
Looijer-van Langen MAC and Dieleman LA. Prebiotics in Chronic Intestinal Inflammation. Inflamm Bowel Dis 15(3) 2009; 15: 454-462.
Lorenz R, Weber PC, Szimnau P, Heldwein W, Strasser T, Loeschke K. Supplementation with n-3 fatty acids from fish oil in chronic inflammatory bowel disease – a randomized, placebocontrolled, double-blind cross-over trial. J Intern Med Suppl 1989; 225-32.
MacDonald TT, Hutchings P, Choy MY, Murch S, Cooke A. Tumour necrosis factor- alpha and interferon-gamma production measured at the single cell level in normal and inflamed human intestine. Clin Exp Immunol 1990;81:301-5.
Marczylo TH, Verschoyle RD, Cooke DN, et al. Comparison of systemic availability of curcumin with that of curcumin formulated with phosphatidylcholine. Cancer Chemother Pharmacol 2007;60:171-177.
Mishkin S. Dairy sensitivity, lactose malabsorption, and elimination diets in inflammatory bowel disease. Am J Clin Nutr 1997; 65:564-7.
Najafzadeh M, Reynolds PD, Baumgartner A, Anderson D. Flavonoids inhibit the genotoxicity of hydrogen peroxide (H2O2) and of the food mutagen 2-amino-3-methylimadazo[4,5- f]-quinoline (IQ) in lymphocytes from patients with inflammatory bowel disease (IBD). Mutagenesis vol. 24 no. 5 pp. 405–411, 2009.
Ockenga J, Borchert K, Stuber E, et al. Glutamine-enriched total parenteral nutrition in patients with inflammatory bowel disease. Eur J Clin Nutr. 2005;59:1302–1309.
Prantera C, Scribano ML, Falasco G, Andreoli A, Luzi C. Ineffectiveness of probiotics in preventing recurrence after curative resection for Crohn’s disease: a randomised controlled trial with Lactobacillus GG. Gut 2002; 51(3): 405-9.
Rahimi R, Nikfar S, Rahimi F, Elahi B, Derakshani S, Vafaie M, Abdollah M. A Meta-Analysis on the Efficacy of Probiotics for Maintenance of Remission and Prevention of Clinical and Endoscopic Relapse in Crohn’s Disease. Dig Dis Sci 2008; 53: 2524-253.
Raman M, Milestone AN, Walters JRF, Hart AL, and Ghosh S. Vitamin D and gastrointestinal disease: inflammatory bowel disease and colorectal cancer. Ther Adv Gastroenterol 200; 4(1): 49-62.
Rembacken BJ, Snelling AM, Hawkey PM, Chalmers DM, Axon AT. Non-pathogenic Escherichia coli versus mesalazine for the treatment of ulcerative colitis: a randomised trial. Lancet 1999; 354(9179): 635-9.
Salh B, Assi K, Templeman V, Parhar K, Owen D, Gomez-Munoz A, Jacobson K. Curcumin attenuates DNB-induced murine colitis. Am J Physiol Gastrointest Liver Physiol 2003; 285(1): G235-43
Schultz M, Timmer A, Herfarth HH, Sartor RB, Vanderhoof JA, Rath HC. Lactobacillus GG in inducing and maintaining remission of Crohn’s disease. BMC Gastroenterol. 2004 Mar 15;4:5.
Stenson WF, Cort D, Rodgers J et al. Dietary supplementation with fish oil in ulcerative colitis. Ann Intern Med 1992; 116: 609-14.
Szilagyi A. Altered colonic environment, a possible predisposition to colorectal cancer and colonic inflammatory bowel disease: Rationale of dietary manipulation with emphasis on disaccharides. Can J Gastroenterl 1998; 12(3): 133-146.
Uchiyama K, Nakamura M, Odahara S, Koido S, Katahira K, Shiraishi H, Ohkusa T, Fujise K, Tajiri H. N-3 polyunsaturated fatty acid diet therapy for patients with inflammatory bowel disease. Inflamm Bowel Dis. 2010 Oct;16(10):1696-707.
Venturi A, Gionchetti P, Rizzello F, Johansson R, Zucconi E, Brigidi P, Matteuzzi D, Campieri M. Impact on the composition of the faecal fl ora by a new probiotic preparation: preliminary data on maintenance treatment of patients with ulcerative colitis. Aliment Pharmacol Ther 1999; 13: 1103-1108
Wyatt J, Vogelsang H, Hubl W, Waldhoer T, Lochs H. Intestinal permeability and the prediction of relapse in Crohn’s disease. Lancet 1993;341:1437-9.
Zocco MA, dal Verme LZ, Cremonini F, Piscaglia AC, Nista EC, Candelli M, Novi M, Rigante D, Cazzato IA, Ojetti V, Armuzzi A, Gasbarrini G, Gasbarrini A. Efficacy of Lactobacillus GG in maintaining remission of ulcerative colitis. Aliment Pharmacol Ther. 2006 Jun 1;23(11):1567-74.
HPA Axis Dysregulation and Human Health
HPA Axis Dysregulation and Human Health
What is the evidence?
Regulation of hypothalamus-pituitary-adrenal (HPA) axis activity has far reaching implications for many areas of human health. Abnormalities in HPA axis reactivity and secretion of its primary effector, cortisol, have been observed in various autoimmune diseases, chronic fatigue syndrome (CFS) and fibromyalgia (FM), as well as in patients with major depression and cancer (Brivio 2010, Chrousos 1995, Nussey 2001, Romer 2009). Cortisol exerts widespread anti-inflammatory, immunosuppressive, catabolic, hyperglycemic, and anti-reproductive effects that may modulate these disease processes; strategies that modulate cortisol activity either through direct hormone replacement, or herbal/ nutritional/ lifestyle strategies, may be useful as therapy for such conditions (Chrousos 1995). This article reviews determinants of the HPA stress response, the concept of hypoadrenalism/ adrenal fatigue, and existing human trials of low-dose hydrocortisone replacement. HPA axis function in specific conditions, adrenal function testing, and nutritional/ herbal interventions strategies are to be discussed in future articles.
HPA Axis Physiology
Activation of the HPA axis in response to a perceived stressor begins with the secretion of corticotropin releasing hormone (CRH) and arginine-vasopressin (AVP) from the paraventricular nucleus (PVN) of the hypothalamus (Smith 2006, Tsigos 2002). This triggers release of adrenocorticotropic hormone (ACTH) from the anterior pituitary, which in turn stimulates the secretion of glucocorticoids, notably cortisol, by the adrenal cortex. Cortisol acts through the ubiquitous distribution of glucocorticoid receptor (GR) throughout the body, altering cellular gene expression through activation of glucocorticoid response elements (Chrousos 1995). Inhibition of the HPA axis is by negative feedback of cortisol on hypothalamic and pituitary centers, thus limiting the duration of tissue exposure to the deleterious effects of chronic elevated cortisol (Nussey 2001).
Stressors
Stressors that drive CRH/ cortisol secretion can be categorized as physical stressors such as exertion, trauma, and lack of sleep; psychological stressors; and biochemical stressors such as hypoglycemia and inflammation (Balbo 2010). Sleep deprivation or irregularity is an often overlooked contributor to a patient’s overall stress load. Sleep onset modestly inhibits cortisol secretion, while awakenings are accompanied by increased secretion, an effect that is likely mediated in part by melatonin, the diurnal counterpart to cortisol (Balbo 2010). Chapotot demonstrated that plasma cortisol level, secretory rate, and pulse amplitude were all significantly increased in healthy young men after a night of sleep deprivation compared to regular nocturnal sleep (p<0.05) (2001). Sleep phase shift like that which occurs during travel across time zones or with irregular hours of sleep also induces dramatic increases in cortisol (Balbo 2010).
Inflammatory mediators such as TNF-α, IL-1, and IL-6 have also been shown to activate the HPA axis (Tsigos 2002). These cytokines, particularly IL-6, signal the HPA axis at multiple levels (hypothalamus, pituitary, and adrenal glands) to secrete more glucocorticoids in order to control inflammation (Chrousos 1995, Mastorakos 1995). Tsigos showed that administration of IL-6, a key mediator of inflammation, resulted in acutely elevated plasma ACTH and elevation in cortisol lasting for over four hours in healthy subjects (1997). Mastorakos found that IL-6 administration increased ACTH and cortisol levels in cancer patients (1993). Interestingly, Vgontzas showed that sleep deprivation in otherwise healthy subjects induced significantly increased daytime secretion of IL-6, which may represent an indirect mechanism by which sleep loss induces cortisol (1999).
Rapidly fluctuating blood glucose levels represent another unsuspected source of biochemical stress. Hypoglycemia requires adrenal compensation, triggering cortisol mediated liver gluconeogenesis in order to restore normal glucose levels (Chen 2010). It is interesting to note that a feeding response is also commonly observed, such that cortisol tends to rise modestly after meals, particularly at noon (Lemmens 2011, Vicennati 2002). Although there is conflicting evidence, it seems that intake of carbohydrates and simple sugar results in a higher post meal cortisol response compared to water, fats, and protein (Martens 2010), while animal studies suggest that high protein and high fat foods may reduce post meal cortisol (Lacroix 2004). This makes sense in light of the fact that consumption of fats and protein results in a smoother, more even post-prandial blood glucose profile, requiring less compensation by the adrenal glands. A corollary of this is that functional hypoglycemia may therefore also be an indication of hypoadrenalism (Gaby 2010).
Obesity has been associated with greater 24 hour cortisol secretion (Vicennati 2002), which may be due in part to the inflammatory milieu created by abdominal adiposity, with elevated CRP etc, however obesity is also characterized by HPA axis hyperresponsiveness to neuroendocrine stimuli (Vicennati 2000, 2004). Vincennati conducted a study of HPA stimulation and suppression through the administration of arginine-vasopressin (AVP, an HPA stimulant produced by the hypothalamus) and alprazolam (a short acting benzodiazepine and centrally acting HPA inhibitor used to treat anxiety) in women (2004). Overweight/ obese women showed higher ACTH and cortisol responses to the AVP stimulation test, and significantly greater inhibition after alprazolam compared to non-overweight controls (Vicennati 2004). Alprazolam also decreased 24h-urinary free cortisol excretion by approximately 50% in the overweight/obese subjects, but not in controls.
Other endocrine axes also interact with cortisol and the HPA-axis. In women, an age-related decrease in estradiol is thought to decrease the responsiveness of the HPA axis to stimulatory cytokines, based on evidence in animal models of ovarian failure (Straub 2000, Xia-Zhang 1995), and estrogen replacement therapy in menopausal women has been shown to suppresses the HPA axis response to emotional stress (Carrasco 2003, Lindheim 1992). Tsigos reports that activation of the HPA axis is associated with decreased production of TSH and decreased peripheral conversion of T4 to the more biologically active T3 (as seen in ‘‘euthyroid sick’’ syndrome), which may serve to conserve energy during stress (2002). In animal models, induction of the stress response (ACTH secretion) has been shown to decrease circulating levels of T4 and T3 (Helmreich 2005).
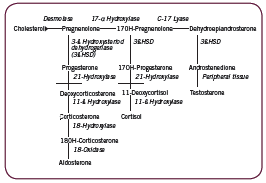
Cortisol synthesis
The adrenal cortex is responsible for three major pathways/ products: the mineralocorticoid pathway leading to production of aldosterone; the androgen/ estrogen pathway producing DHEA, testosterone, and estradiol; and the glucocorticoid pathway leading to production of cortisol. These are depicted in Figure 1. Two key enzymes convert intermediaries to cortisol: 21ß-hydroxylase converts 17-hydroxy-progesterone to 11- ßdeoxycortisol, which is then converted to cortisol by 11ß-hydroxylase. Measurement of serum 17-hydroxy-progesterone (17-OH-P) level is often used to assess the activity of these enzymes; in the event of an enzyme deficiency, as in non-classical congenitial adrenal hyperplasia (NCAH) for example, 17-OH-P will accumulate in the pathway (Kelestimur 2006). Negative (eg. catabolic) effects of cortisol are countered in part by the adrenal androgen DHEA (Straub 2000).
Cortisol rhythm
Cortisol secretion is pulsatile, diurnal, and stress-responsive (Balbo 2010). ACTH and cortisol levels peak in the morning (acrophase) just prior to waking, with gradual decline throughout the rest of the day, and an extended period of low levels around midnight (nadir), followed by a rapid rise during the second half of the night (Balbo 2010). Control of diurnal cortisol secretion is thought to be under direct neural control via a multisynaptic pathway from the pineal gland (site of melatonin synthesis), as well as through an intrinsic circadian oscillator in the adrenal gland itself that can be directly modulated by melatonin (Balbo 2010).
The Evolution of HPA and Disease
Seyle’s now-famous General Adaptation Syndrome (GAS) describes three phases of HPA activity in response to stress; the Alarm Reaction, Resistance, and Exhaustion. His 1950 paper describes the biology of each phase in surprising detail. In brief, during the first stage the adrenal glands release epinephrine and glucocorticoids to help restore homeostasis; when homeostasis is restored, adaptation is maintained through sustained secretion of glucocorticoids; when resistance to the stressor can no longer be maintained, exhaustion ensues, accompanied by the onset of disease. This has usually been thought of as the downregulation over time of adrenal gland responsiveness to ACTH/ stressors, or decreased adrenal reserve, resulting in inadequate cortisol secretion to meet physiological requirements and consequent loss of anti-inflammatory control (Gaby 2011, Jefferies 2004). Seyle wrote about “diseases of adaptation” (1950), providing a detailed list of chronic inflammatory diseases, including allergies, asthma, eczema, psoriasis, lupus, rheumatoid arthritis, ulcerative colitis, and chronic urticaria, which may be one of the earliest medical descriptions of what is now considered by naturopathic doctors as “adrenal fatigue” or mild hypoadrenalism.
Although glucocorticoid therapy has since evolved such that standard use involves large pharmacological doses for limited periods of time, William Jefferies pioneered the use of physiologic doses of cortisol as long term therapy of many conditions (2004). Jefferies distinguishes between 1) replacement dosage (~35-40 mg/d) such as that used in adrenalectomized patients or in Addison’s disease; 2) supra-replacement dosage (>40mg/d) or pharmacological doses; and 3) sub-replacement dosage, the dose he utilizes as physiologic cortisol therapy in patients with intact adrenals: hydrocortisone 20mg/d in four divided doses, with food to avoid gastrointestinal side effects such as hyperacidity (2004). In his book, Safe Uses of Cortisol, Jefferies reports cases of many patients with diverse clinical presentations from his own practice who have benefited from physiologic doses of cortisol: rheumatoid arthritis and autoimmune disease, allergies, infertility and recurrent miscarriage, viral infections, functional hypoglycemia, thyroid conditions, and chronic fatigue syndrome (Jefferies 2004).
Jefferies explains that although it is low dose, the physiologic use of cortisol has not historically been considered as a therapeutic option because of the assumption that it carries the same safety concerns as pharmacological doses (eg. diabetes, osteoporosis, etc). He emphasizes the importance of four divided doses (roughly q6h) rather than twice daily “because of evidence that normal blood levels and some metabolic effects … do not last longer than eight hours” (2004). This maintains a smoother cortisol curve without sharp peaks and troughs throughout the day, which more closely mimics physiological secretion. A retrospective chart analysis by Howlett lends support to this dosing schedule: using data from 130 patients and 174 3-point serum cortisol curves, Howlett found that TID dosing achieved “optimal replacement” more frequently than BID dosing, 60% versus 15% of cases (p<0.001) (1997). Optimal replacement was defined as the dose that achieved a urinary free cortisol and 0900h serum cortisol within the normal range to avoid over-replacement, while maintaining 1230h and 1730h cortisol above 50nmol/L, and ideally above 100 nmol/L to avoid under-replacement (1997).
In his text, Nutritional Medicine, Alan Gaby discusses mild hypoadrenalism or adrenocortical insufficiency, differentiating it from the severe, autoimmune hypoadrenalism of Addison’s disease (2011). He lists characteristic symptoms as including fatigue, weakness, anorexia, nausea, vomiting, weight loss, salt craving, hypotension or orthostatic hypotension, hypoglycemia, hyperpigmentation of the skin, decreased body hair in women, and poor tolerance to stress or exertion. Gaby notes that the inability of a clinically hypothyroid patient to tolerate even low doses of thyroid hormone may indicate hypoadrenalism. Gaby also describes low dose hydrocortisone therapy as a treatment option for hypoadrenalism and chronic fatigue syndrome, corroborating Jefferies’ experience (2011). He relates that he uses the lowest effective dose of hydrocortisone, sometimes lower than 5mg QID, and has noted improvement at doses as low as 1mg TID.
Hydrocortisone is the glucocorticoid of choice for treatment of hypoadrenalism because it is the biochemical equivalent to endogenous cortisol. Other corticosteroids are often more potent in their activity, and as discussed above are dosed very differently. A comparison of glucocorticoids adapted from the Compendium of Pharmaceuticals and Specialties (CPS 2011) is provided in Table 1. According to this table, Prednisone is approximately four times as potent as hydrocortisone.
Human Trials of Hydrocortisone Therapy: CFS and PTSD Chronic Fatigue Syndrome
It has been noted by many authors that CFS resembles Addison’s in its symptomotology (Cleare 2001, Gaby 2011). HPA abnormalities have been documented at many levels of the axis in CFS, but not all with equal consistency (Chrousos 1995, Cleare 2001, Gaab 2002, Jerjes 2007, Papadopoulos 2009, Scott 2000). Cleare reviews HPA axis activity in CFS, stating that “tests of negative feedback using dexamethasone support [the] hypersuppression of cortisol and negative feedback…with over half the studies [also] suggesting lowered basal cortisol and/ or blunted HPA axis responses” (2004). A detailed analysis is beyond the scope of this paper, however three controlled human trials of low dose hydrocortisone therapy conducted in patients with CFS are discussed below.
Cleare found benefit from hydrocortisone in a randomized placebo controlled crossover trial of 32 CFS patients without comorbid psychiatric disorder (1999, 2001). Two dosages investigated: either 5mg or 10mg, taken as a single daily dose with breakfast for 28 days. Both dosages had comparable effects on fatigue scores (p=0.90). In the collective treatment group fatigue score dropped 7.2 points, compared to 3.3 points in the placebo group, a mean difference of 4.5 points (p<0.009). Mean disability score also decreased with treatment but not placebo. Fatigue scores normalized in nine (28%) of the hydrocortisone group compared to two (9%) of the placebo group (p=0.05). Importantly, insulin stress testing showed that these doses of hydrocortisone did not induce adrenal suppression. Reported side effects in three patients on hydrocortisone included: worsening of acne, nervousness, and improvement of eczema.
In contrast, RCTs by McKenzie and Blockmans found no clinically significant benefits, albeit at doses and schedules that were shown to induce adrenal suppression: 25-35mg hydrocortisone BID and a combination of 5mg hydrocortisone + 20mg fludrocortisone daily (Blockmans 2003, McKenzie 1998). McKenzie did find that a greater percentage (53% vs 29%; P=.04) recorded an improvement of five or more points in Wellness score, and a higher average improvement in Wellness score on more days compared to placebo recipients (P<.001) (1998). A lack of correlation between the magnitude of impaired basal/ stimulated serum cortisol levels and disease severity in this study, yet small improvements with hydrocortisone therapy suggests the possibility of peripheral glucocorticoid resistance (Chrousos 1995, McKenzie 1998). Blockmans notes that CFS is a heterogeneous disorder, and treatment with low-dose steroids may only be effective for a certain subset of patients (2003).
Although hydrocortisone for treatment of fibromyalgia has not been investigated, patients with this condition also display HPA axis dysfunction, with lowered salivary cortisol, urinary cortisol excretion and more pronounced suppression of cortisol compared with controls on low dose dexamethasone testing (Izquierdo- Alvarez 2008, Riva 2010, Wingenfeld 2007).
Post Traumatic Stress Disorder (PTSD)
Aerni conducted a small randomized, double blind, placebo controlled cross over trial of three patients with PTSD (2004). Low dose hydrocortisone 10mg doses either as a single daily dose in one patient and as two divided doses in the other two patients or placebo was given for one month. Results showed that there was a significant treatment effect in all the patients, with “cortisolrelated reductions of at least 38% in one of the daily rated symptoms of traumatic memories, as assessed by self-administered rating scales” (Aerni 2004). Additionally, the clinician-Administered PTSD Scale ratings assessed after each month showed cortisolrelated improvements for reexperiencing symptoms, and in one patient, for avoidance symptoms also, but there was no impact on hyperarousal symptoms.
Conclusion
The HPA axis is activated in response to physical, psychological, and biochemical stresses. Mild hypoadrenalism is thought to contribute to the onset of a spectrum of chronic inflammatory conditions. Drs Jefferies and Gaby have pioneered the use of physiological doses of hydrocortisone as therapy for autoimmune disease and other conditions such as chronic fatigue syndrome. Although limited, human trials of low dose hydrocortisone suggest that this therapy may be of benefit to some patients with this condition, with one trial suggesting benefit in approximately 1/3 of patients. There is a need for better understanding of testing methods and natural strategies that can be used to treat conditions characterized by mild hypoadrenalism.
References:
Aerni A, Traber R, Hock C, Roozendaal B, Schelling G, Papassotiropoulos A, Nitsch RM, Schnyder U, de Quervain DJ. Low-dose cortisol for symptoms of posttraumatic stress disorder. Am J Psychiatry. 2004 Aug;161(8):1488-90.
Balbo M, Leproult R, Van Cauter E. Impact of sleep and its disturbances on hypothalamo-pituitary-adrenal axis activity. Int J Endocrinol. 2010;2010:759234.
Blockmans D, Persoons P, Van Houdenhove B, Lejeune M, Bobbaers H. Combination therapy with hydrocortisone and fludrocortisone does not improve symptoms in chronic fatigue syndrome: a randomized, placebo-controlled, double-blind, crossover study. Am J Med. 2003 Jun 15;114(9):736-41.
Brivio F, Fumagalli L, Fumagalli G, Pescia S, Brivio R, Di Fede G, Rovelli F, Lissoni P. Synchronization of cortisol circadian rhythm by the pineal hormone melatonin in untreatable metastatic solid tumor patients and its possible prognostic significance on tumor progression. In Vivo. 2010 Mar-Apr;24(2):239-41.
Carrasco GA, Van de Kar LD. Neuroendocrine pharmacology of stress. Eur J Pharmacol. 2003 Feb 28;463(1-3):235-72.
Chapotot F, Buguet A, Gronfier C, Brandenberger G. Hypothalamo-pituitary-adrenal axis activity is related to the level of central arousal: effect of sleep deprivation on the association of high-frequency waking electroencephalogram with cortisol release. Neuroendocrinology. 2001 May;73(5):312-21.
Chen CL, Willis BA, Mooney L, Ong GK, Lim CN, Lowe SL, Tauscher-Wisniewski S, Cutler GB Jr, Wiss SD. Cortisol response to individualised graded insulin infusions: a reproducible biomarker for CNS compounds inhibiting HPA activation. Br J Clin Pharmacol. 2010 Dec;70(6):886-94.
Chrousos GP. The hypothalamic-pituitary-adrenal axis and immune-mediated inflammation. N Engl J Med. 1995 May 18;332(20):1351-62.
Cleare AJ. The HPA axis and the genesis of chronic fatigue syndrome. Trends Endocrinol Metab. 2004 Mar;15(2):55-9.
Cleare AJ, Miell J, Heap E, Sookdeo S, Young L, Malhi GS, O’Keane V. Hypothalamopituitary- adrenal axis dysfunction in chronic fatigue syndrome, and the effects of lowdose hydrocortisone therapy. J Clin Endocrinol Metab. 2001 Aug;86(8):3545-54.
Cleare AJ, Heap E, Malhi GS, Wessely S, O’Keane V, Miell J. Low-dose hydrocortisone in chronic fatigue syndrome: a randomised crossover trial. Lancet. 1999 Feb 6;353(9151):455-8.
Compendium of Pharmaceuticals and Specialties. Corticosteroids: Systemic (CphA Monograph). Updated 2011. www-e-therapeutics-ca. Accessed 10 May 2011.
Gaab J, Hüster D, Peisen R, Engert V, Schad T, Schürmeyer TH, Ehlert U. Low-dose dexamethasone suppression test in chronic fatigue syndrome and health. Psychosom Med. 2002 Mar-Apr;64(2):311-8.
Gaby A. Nutritional Medicine. Concord: Fritz Perlberg Publishing, 2011.
Groves RW, Toms GC, Houghton BJ, Monson JP. Corticosteroid replacement therapy: twice or thrice daily? J R Soc Med. 1988 Sep;81(9):514-6.
Helmreich DL, Parfitt DB, Lu XY, Akil H, Watson SJ. Relation between the hypothalamic-pituitary-thyroid (HPT) axis and the hypothalamic-pituitary-adrenal (HPA) axis during repeated stress. Neuroendocrinology. 2005;81(3):183-92.
Hori H, Teraishi T, Sasayama D, Ozeki Y, Matsuo J, Kawamoto Y, Kinoshita Y, Hattori K, Higuchi T, Kunugi H. Poor sleep is associated with exaggerated cortisol response to the combined dexamethasone/CRH test in a non-clinical population. J Psychiatr Res. 2011 Apr 26. [Epub ahead of print]
Howlett TA. An assessment of optimal hydrocortisone replacement therapy. Clin Endocrinol (Oxf). 1997 Mar;46(3):263-8.
Izquierdo-Alvarez S, Bocos-Terraz JP, Bancalero-Flores JL, Pavón-Romero L, Serrano-Ostariz E, de Miquel CA. Is there an association between fibromyalgia and below-normal levels of urinary cortisol? BMC Res Notes. 2008 Dec 22;1:134.
Jefferies W. Safe Uses of Cortisol, Third Edition. Springfield: Charles C Thomas, 2004.
Jerjes WK, Taylor NF, Wood PJ, Cleare AJ. Enhanced feedback sensitivity to prednisolone in chronic fatigue syndrome. Psychoneuroendocrinology. 2007 Feb;32(2):192-8.
Kelestimur F. Non-classic congenital adrenal hyperplasia. Pediatr Endocrinol Rev. 2006 Aug;3 Suppl 3:451-4.
Lacroix M, Gaudichon C, Martin A, Morens C, Mathé V, Tomé D, Huneau JF. A longterm high-protein diet markedly reduces adipose tissue without major side effects in Wistar male rats. Am J Physiol Regul Integr Comp Physiol. 2004 Oct;287(4):R934-42.
Lemmens SG, Born JM, Martens EA, Martens MJ, Westerterp-Plantenga MS. Influence of consumption of a high-protein vs. high-carbohydrate meal on the physiological cortisol and psychological mood response in men and women. PLoS One. 2011 Feb 3;6(2):e16826.
Lindheim SR, Legro RS, Bernstein L, Stanczyk FZ, Vijod MA, Presser SC, Lobo RA. Behavioral stress responses in premenopausal and postmenopausal women and the effects of estrogen. Am J Obstet Gynecol. 1992 Dec;167(6):1831-6.
Martens MJ, Rutters F, Lemmens SG, Born JM, Westerterp-Plantenga MS. Effects of single macronutrients on serum cortisol concentrations in normal weight men. Physiol Behav. 2010 Dec 2;101(5):563-7.
Mastorakos G, Magiakou MA, Chrousos GP. Effects of the immune/inflammatory reaction on the hypothalamic-pituitary-adrenal axis. Ann N Y Acad Sci. 1995 Dec 29;771:438-48.
Mastorakos G, Chrousos GP, Weber JS. Recombinant interleukin-6 activates the hypothalamic-pituitary-adrenal axis in humans. J Clin Endocrinol Metab. 1993 Dec;77(6):1690-4.
McKenzie R, O’Fallon A, Dale J, Demitrack M, Sharma G, Deloria M, Garcia- Borreguero D, Blackwelder W, Straus SE. Low-dose hydrocortisone for treatment of chronic fatigue syndrome: a randomized controlled trial. JAMA. 1998 Sep 23- 30;280(12):1061-6.
Nussey S, Whitehead S. Endocrinology: An Integrated Approach. Chapter 4: The Adrenal Gland. Feedback control of glucocorticoids. BIOS Scientific Publishers Limited, 2001. Available at NCBI: http://www.ncbi.nlm.nih.gov/books/NBK26/ Accessed 27 May 2011.
Riva R, Mork PJ, Westgaard RH, Rø M, Lundberg U. Fibromyalgia syndrome is associated with hypocortisolism. Int J Behav Med. 2010 Sep;17(3):223-33.
Römer B, Lewicka S, Kopf D, Lederbogen F, Hamann B, Gilles M, Schilling C, Onken V, Frankhauser P, Deuschle M. Cortisol metabolism in depressed patients and healthy controls. Neuroendocrinology. 2009;90(3):301-6.
Scott LV, Svec F, Dinan T. A preliminary study of dehydroepiandrosterone response to low-dose ACTH in chronic fatigue syndrome and in healthy subjects. Psychiatry Res. 2000 Dec 4;97(1):21-8.
Selye H. Stress and the general adaptation syndrome. Br Med J. 1950 Jun 17;1(4667):1383-92.
Smith SM, Vale WW. The role of the hypothalamic-pituitary-adrenal axis in neuroendocrine responses to stress. Dialogues Clin Neurosci. 2006;8(4):383-95.
Straub RH, Schölmerich J, Zietz B. Replacement therapy with DHEA plus corticosteroids in patients with chronic inflammatory diseases–substitutes of adrenal and sex hormones. Z Rheumatol. 2000;59 Suppl 2:II/108-18.
Tsigos C, Papanicolaou DA, Defensor R, Mitsiadis CS, Kyrou I, Chrousos GP. Dose effects of recombinant human interleukin-6 on pituitary hormone secretion and energy expenditure. Neuroendocrinology. 1997 Jul;66(1):54-62.
Tsigos C, Chrousos GP. Hypothalamic-pituitary-adrenal axis, neuroendocrine factors and stress. J Psychosom Res. 2002 Oct;53(4):865-71.
Vicennati V, Pasquali R. Abnormalities of the hypothalamic-pituitary-adrenal axis in nondepressed women with abdominal obesity and relations with insulin resistance: evidence for a central and a peripheral alteration. J Clin Endocrinol Metab. 2000 Nov;85(11):4093-8.
Vicennati V, Ceroni L, Gagliardi L, Gambineri A, Pasquali R. Comment: response of the hypothalamic-pituitary-adrenocortical axis to high-protein/fat and highcarbohydrate meals in women with different obesity phenotypes. J Clin Endocrinol Metab. 2002 Aug;87(8):3984-8.
Vicennati V, Ceroni L, Gagliardi L, Pagotto U, Gambineri A, Genghini S, Pasquali R. Response of the hypothalamic-pituitary-adrenal axis to small dose arginine-vasopressin and daily urinary free cortisol before and after alprazolam pre-treatment differs in obesity. J Endocrinol Invest. 2004 Jun;27(6):541-7.
Vgontzas AN, Mastorakos G, Bixler EO, Kales A, Gold PW, Chrousos GP. Sleep deprivation effects on the activity of the hypothalamic-pituitary-adrenal and growth axes: potential clinical implications. Clin Endocrinol (Oxf). 1999 Aug;51(2):205-15.
Wingenfeld K, Wagner D, Schmidt I, Meinlschmidt G, Hellhammer DH, Heim C. The low-dose dexamethasone suppression test in fibromyalgia. J Psychosom Res. 2007 Jan;62(1):85-91.
Xia-Zhang L, Xiao E, Ferin M. A 5-day estradiol therapy, in amounts reproducing concentrations of the early-mid follicular phase, prevents the activation of the hypothalamo-pituitary-adrenal axis by interleukin-1 alpha in the ovariectomized rhesus monkey. J Neuroendocrinol. 1995 May;7(5):387-92.
Homeopathy
Homeopathy
An introduction to application in pediatrics
The use of complementary and alternative medicine (CAM) in pediatric primary care is common, with homeopathy being one of the most widely used CAM modalities in the world. In a recent survey done in the UK, the rate of homeopathic use in children amongst GPs is 22%, with most prescribing taking place for infants less than one year old with minor self-limiting conditions at a relatively low rate of approximately one per month. A smaller number of GPs report prescribing much more frequently (Ekins-Daukes 2005). In Germany, homeopathy is the most frequently used CAM treatment for childhood ailments, where nearly 50% of the homeopathic medicines are prescribed by medical doctors and Heilpraktiker, and used to treat a wide range of conditions with a greater rate of use for self limiting conditions (Du 2009). The publications on the use of CAM in pediatric primary care and oncology indicate that homeopathy plays a complimentary but largely unofficial role in pediatric oncology in Israel (Ben Arush 2006), Canada (Fernandez 1998), Italy (Steinsbekk 2006), and Netherlands (Grootenhuis 1998). In the US, it is estimated that over 900 000 children are administered homeopathic medicines on a yearly basis (Barnes 2008). In India, homeopathy is widely used in pediatric care, as it is incorporated as a system of primary care within the established medical system (Ghosh 2010).
Homeopathic remedies are potentized substances at a dilution level often greatly surpassing the Avogadro number. This naturaly gives rise to questions about the mechanism of action of homeopathic remedies. Despite much experimentation, the exact mechanism through which homeopathic remedies effect change remains elusive. Recent work has focused on the memory of water hypothesis in which properties of water are held to be influenced by the history of substances water has been in contact with (Chaplin 2007). Other work on the subject has focused on quantum interactions and entanglement of the remedies with the substances from which they are made, as well as quantum entanglement between the patient and practitioner (Molski 2010, Neuhouser 2001). Although no firm consensus has yet emerged, research into the question is gaining solid ground (Enserink 2010).
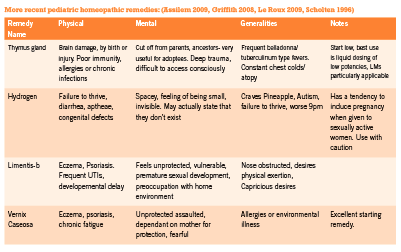
Most homeopathic remedies are made from natural ingredients (plants, minerals, or animals), yet numerous recent remedies have been created from a wider range of substances, such as colored light (Griffith 2008) and physiological tissues from the birth process (Assilem 2009). In addition, more recent work by Jan Scholten and the members of the Mumbai school in India have introduced classification of remedies into thematically oriented groups, and have offered deeper exploration into the unconscious levels of remedy pictures (Sankaran 2008). Their work has re-energized the field of homeopathy, widening the number of remedies that can be matched to childhood ailments.
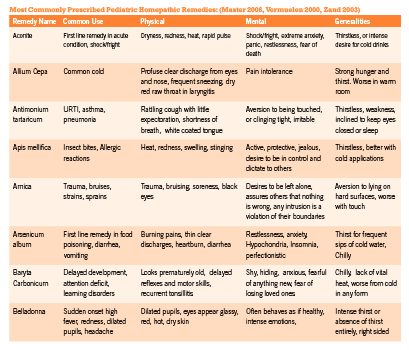
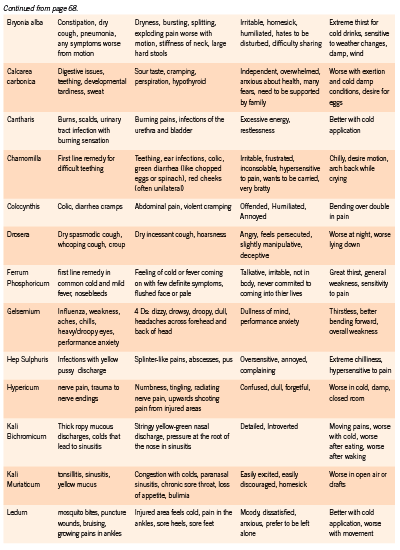
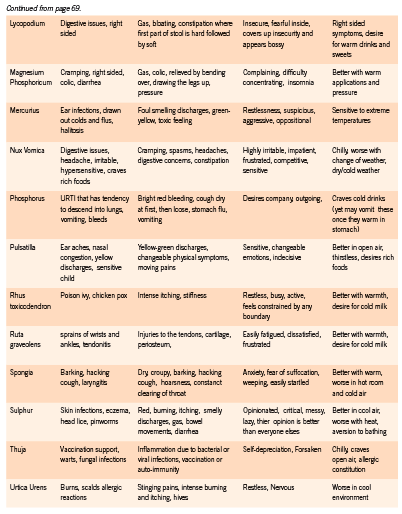
Homeopathic remedies are symptom specific. Prescribing in pediatric patients requires keen observational skills as well as a strong knowledge of materia medica. The choice of remedy must take into consideration general temperament of the child, the emotional responses to challenges that they are facing, as well as the unique details of the physical symptoms they are exhibiting or experiencing. This requires careful observation on the part of the parent and the physician since children are often unable to report the subtleties of their symptoms. In addition, observation of the parents and the social environment of the child may offer great clues to remedy selection (Master 2006).
Homeopathic remedies can be prescribed to children on an acute or constitutional basis. Constitutional prescribing is mostly indicated in chronic or recurrent conditions and is based on the child’s overall physical, emotional, and mental symptom picture. A constitutional remedy strengthens and stimulates the child’s vitality, improves physical and mental development and helps maintain their overall health over time. For acute presentations, symptom-specific prescribing based on the totality of the presentation at the time of intake is most commonly employed. A matching acute remedy will promptly releave the child’s symptoms and significantly increase rate of recovery.
Dosing strategies in children are similar to adult protocols. Although most books on pediatric dosing recommend low dose prescribing, for healthy children with strong vitality, dosing higher potencies of 200c, 1M, and 10M often show great results, as long as the remedy is a close similimum. It is generally prudent, however, to begin with lower potencies (such as 6c, 30c) and gradually move up to 200c, 1M and 10M if vitality of the child is vague, or if the remedy match is not entirely revealed. For children with chronic illneses and disabilities, higher potency remedies may provoke aggravations or worsening of the presenting symptoms. In such sensitive cases, liquid dosing and LM dosing are recommended.
The following protocol is designed to minimize aggravations in sensitive children, yet speed up progress of their condition considerably: Begin with 6c potency of the selected remedy diluted and succussed in alcohol and water. Administer 1-10 drops to the child and observe for a week. If no response, redose. If positive response results, redose only when you see child ceasing to improve. If progress comes to a plateau, dose more often until daily dosing. The typical progression of potencies in this protocol is 6c, 30c, LM-1, LM-2, and upwards (De Schepper 2004).
References:
Assilem, M. Matridonal Remedies of The Human Family: Gifts of the Mother. 2009. Idolatry. ink . USA. Pp. 60- 69.
Barnes P, Bloom B. Complementary and Alternative Medicine Use Among Adults and Chilren: United States 2007. National Health Statistics Reports. Number 12. Dec. 2008
Ben Arush MW, Geva H, Ofir R, Mashiach T, Uziel R, Dashkovsky Z. Prevalence and characteristics of complementary medicine used by pediatric cancer patients in a mixed western and middle-eastern population. Journal of Pediatric Hematology/Oncology. 2006;28(3):141–146.
Boon, HS et al. Practice patterns of naturopathic physicians: results from a random survey of licensed practitioners in two US States. “javascript:AL_get(this,%20’jour’,%20’BMC%20 Complement%20Altern%20Med.’);”BMC Complement Altern Med. 2004 Oct 20; 4:14.
Fernandez CV, Stutzer CA, MacWilliam L, Fryer C. Alternative and complementary therapy use in pediatric oncology patients in British Columbia: prevalence and reasons for use and nonuse. Journal of Clinical Oncology. 1998;16(4):1279–1286
“http://www.ncbi.nlm.nih.gov/pubmed?term=%22Chaplin%20 MF%22%5BAuthor%5D”Chaplin MF. The Memory of Water: an overview. “javascript:AL_ get(this,%20’jour’,%20’Homeopathy.’);”Homeopathy. 2007 Jul;96(3):143-50.
De Schepper, L. Achieving and maintaining the simillimum : strategic case management for successful homeopathic prescribing. 2004. Full of Life Publishing. Santa Fe, NM.
Du Y, Knopf H. Paediatric homoeopathy in Germany: results of the German Health Interview and Examination Survey for Children and Adolescents (KiGGS). Pharmacoepidemiol Drug Saf 2009 May;18(5):370-9
Ekins-Daukes S, Helms PJ, Taylor MW, Simpson CR, McLay JS. Paediatric homoeopathy in general practice: where, when and why? British Journal of Clinical Pharmacology. 2005;59(6):743–749.
Enserink M, Newsmaker Interview: Luc Montagnier, French Nobelist Escapes “Intellectual Terror” to Pursue Radical Ideas in China. Science 24 December 2010: Vol. 330 no. 6012 p. 1732.
Fernandez CV, Stutzer CA, MacWilliam L, Fryer C. Alternative and complementary therapy use in pediatric oncology patients in British Columbia: prevalence and reasons for use and nonuse. Journal of Clinical Oncology. 1998;16(4):1279–1286 “http://www.ncbi.
nlm.nih.gov/pubmed?term=%22Ghosh%20AK%22%5BAuthor%5D”Ghosh AK. A short history of the development of homeopathy in India. “javascript:AL_get(this,%20’jour’,%20 ’Homeopathy.’);”Homeopathy. 2010 Apr;99(2):130-6.
Griffith, C. The New Materia Medica: Key Remedies for the Future of Homeopathy. 2008. Sterling. New York.
Grootenhuis MA, Last BF, de Graaf-Nijkerk JH, van der Wel M. Use of alternative treatment in pediatric oncology. Cancer Nursing. 1998;21(4):282–288.
Le Roux, P. Butterflies. 2009. Narayana. Kandern, Germany. Pp. 42-43, 95-99.
Master F. Clinical Observations of Children’s Remedies. 2006. 3rd edition. Lutra Services BV. Eidhoven, Netherlands
Milgrom, L.R. Patient-practitioner-remedy (PPR) entanglement. Part 5. Can homeopathic remedy reactions be outcomes of PPR entanglement? Homeopathy 93, 94-98 (2004). “http:// www.ncbi.nlm.nih.gov/pubmed?term=%22Molski%20M%22%5BAuthor%5D”Molski M. Quasi-quantum phenomena: the key to understanding homeopathy. “javascript:AL_ get(this,%20’jour’,%20’Homeopathy.’);”Homeopathy. 2010 Apr;99(2):104-12.
Neuhouser ML, Patterson RE, Schwartz SM, Hedderson MM, Bowen DJ, Standish LJ. Use of alternative medicine by children with cancer in Washington State. Preventive Medicine. 2001;33(5):347–354.
Sankaran, R. Structure, Experiences with the Mineral Kingdom: Vol 1. 2008. Mumbai, India : Homeopathic Medical Publishers.
Scholten, J. Homeopathy and the Elements. 1996. Stichting Alonnissos. Utrecht, Netherlands. Simpson N, Roman K. Complementary medicine use in children: extent and reasons. A population-based study. Br J Gen Pract. 2001;51(472):914–16.
Steinsbekk A, Bentzen N, Brien S. Why do parents take their children to homeopaths? An exploratory qualitative study. Forschende Komplementärmedizin. 2006;13(2):88–93.
Vermuelen F. Concordant Materia Medica. 2000. 3rd Edition. Emryss bv Publishers. Haarlem, Netherlands
Zand J, Rountree R, Walton R. Smart Medicine for Healthier Child. 2003. 2nd Edition. Penguin Group. NY.
Dr John Gannage, MD
Dr John Gannage, MD
Markham Integrative Medicine
The pace with which awareness and interest in Integrative Medicine grows is staggering. Interacting with healthcare providers and patients over the course of a decade has clearly revealed this. The likelihood of a patient receiving words of caution from their family physician when informed of their decision to visit an integrative practitioner a decade ago was guaranteed to be greater than 90%. Seemingly with every passing year this number decreases. As a naturopathic doctor I have personally witnessed an encouraging increase in family physicians taking the time to contact me seeking information on the strategy adopted for a patient under both of our care.
There are invariably dozens of reasons contributing to this shift in attitude towards integrative systems of care. Improved regulatory standing and publicized broad scope of practice for integrative healthcare providers is a strong contributing force. Non- invasive and generally safe treatment strategies are also key. Most physicians interviewed by IHP who recently developed an interest in the area commonly site patient inquiry as a strong motivating force.
An understated contribution to the tremendous success of the integrative medicine movement is the pioneering efforts of conventionally trained physicians who made the decision to apply integrative medicine techniques decades ago, when many professionals in the field were still dealing with patients who requested arriving at the clinic through the back door located in the alley.
Dr John Gannage, MD, opened the Markham Integrative Medicine clinic in 1998, delivering the true essence of integrative, patient- centred healthcare since opening day. John represents a rare type of physician; conventionally trained yet devoted to philosophies of holistic care from the time his training in medicine began. He represents the present and future of integrative healthcare; world- class assessment and diagnostic skills used to construct an individualized holistic treatment plan. While conventionally trained physicians who adopt such an approach are increasing in number, finding one who has been in practice for more than a decade is truly rare. Each and every time I come across such a healer I am amazed at how simple the process of evolution to a “holistic” practitioner really is; simply expose an open- minded physician to quality evidence of application of natural therapeutics. The physician seems to do the rest on their own.
Dr Gannage went to medical school at the University of Toronto (1990 graduate) and followed this with residency training at the University of Ottawa. He describes his interest in integrative medicine coinciding with his arrival at the University of Toronto. John expressed frustration with the lack of patient centred care he observed. By 1995 he was working with chiropractors and osteopathic doctors in the field of sports medicine as a strategy to expose himself to different management systems. John felt his training in conventional medicine afforded powerful assessment tools but failed to provide sufficient options for treatment. He described a desire to expand his medicine bag, completing two diplomas in homeopathy as well as receiving training in chelation therapy, several integrative assessment procedures, and registering as a DAN! doctor, identifying a specialty in integrative management of autism.
As with many practitioners of integrative medicine, John describes a personal health crisis as an instrumental factor in the manner in which he delivers patient management today. Two separate visits over a two+ year period to a golf course produced severe, acute, potentially life- threatening illness. Following the first event Dr Gannage assumed he may have a severe sensitivity to pesticide exposure. Following a complete recovery from the first event, he confirmed his earlier suspicions with the onset of the second event. Environmental medicine has thus become a principle focus of the Markham Integrative Medicine clinic, with the clinic having an established reputation for offering of chelation services.
Chelation services vary widely in specific application. Most interestingly, the clinic played an instrumental role in the continued success of the ongoing TACT trial (trial to assess chelation therapy). This large, multicentre trial to assess a combination chelation protocol of EDTA, vitamin C, B vitamins, and magnesium for impact on progression of atherosclerosis was experiencing difficulties in patient recruitment in its early phases. There was concern that the trial was going to be discontinued.
Dr Gannage was able to quickly register the facility as a satellite clinic of TACT and enroll its first patient. At this critical time in the trials progress, investigators were able to announce that the trial was now a multinational trial and that the first international satellite clinic had successfully recruited its first patient into the trial. The protocol entails a three hour drip, a strong contributor to difficulties in patient recruitment. The results of TACT are eagerly awaited by many members of the integrative medical community.
In addition to offering EDTA chelation for patients with cardiovascular disease, Dr Gannage describes with passion the importance of heavy metal chelation (using DMPS, DMSA) for a wide array of common patient presentations. He feels the causal/ progressive role of heavy metals in many common pathologies is erroneously ignored, and reports tremendous success through applying heavy metal chelation in such cases. Heavy metal chelation is frequently applied to cases of autism and other autistic spectrum disorders, neurodegenerative diseases (dementia, Alzheimer’s, Parkinson’s, Huntington’s, MS, etc…), and several types of autoimmune disease (chronic fatigue syndrome, fibromyalgia, scleroderma, rheumatoid arthritis).
The essential basics of an integrative system of practice are evident throughout the clinic and in application of patient management. Food sensitivity testing and subsequent elimination diet strategies are a core treatment modality. A broad array of integrative diagnostic tools are used, including urinary organic acid testing, salivary hormone profiles, hair mineral analysis/ provocative urinary heavy metal assessment, assessment of whole- body oxidative load, live cell microscopy, nutrigenomic testing, and others. The strong interest in environmental medicine has seen the facility establish an important reputation as a centre for support of amalgam removal procedures.
Dr Gannage has expanded the healthcare team of the Markham Integrative Medicine clinic to include a naturopathic doctor (Jonathan Beatty) and a Registered Nutritional Consultant/ holistic whole foods teaching chef (Anna Midolo). While graetful for his dedication to “retrain” himself, Dr Gannage informed IHP that he advises colleagues to “bring a naturopath on board” rather than undergo the retraining themselves. He emphasizes the combined application of conventional medical practices with a team naturopath simply achieves better patient outcomes. Jonathan Beatty, ND, described to us an immense gratitude for the intensive training he has since received particularly in the realm of autism. This recent Canadian College of Naturopathic Medicine grad has had the privilege of exposure to a large number of cases of autism in a relatively short period of time, able to interact with patients at all stages of treatment, from those initiating an integrative approach to those who have been under Dr Gannage’s care for many years.
The Markham Integrative Medicine clinic calls a heritage home on historic Main Street Markham its home. The home has a unique history, serving as a hospital for patients with cancer in 1911. Dr Gannage converted the home into the clinic in 1998, with several efforts made to make the home tolerable by individuals with multiple chemical sensitivities. A state- of- the- art air recirculation system, avoidance of any furniture or building materials made from particle board, refinishing of the original hardwood floors present in the home, and low VOC finishes and paint are a handful of the efforts made to this effect.
Dr Gannage has recently undertaken the responsibility of overseeing a satellite clinic. NEX Health in Burlington, Ontario, was founded by the late Dr Johann Strobele, who, like Dr Gannage, was among the pioneers of conventionally- trained medical doctors to adopt integrative techniques of patient management. Through Dr Gannage’s willingness to work with the NEX Health team, Dr Strobele’s important work and the patients that have come to rely on his system of practice can continue to be served. We hope in time Dr Gannage and other conventionally- trained practitioners of integrative medicine will consider expanding their director role of satellite clinics, perhaps allowing for a cooperative system of practice between ND and integrative MD as currently exists between MD and nurse practitioner.
IHP is grateful to Dr Gannage and the entire team of the Markham Integrative Medicine clinic for allowing us to showcase their important work to you. The most direct route to better recognition, regulation, and implementation of integrative medicine is to increase the number of practitioners of the discipline. The more patients whose lives are benefited by integrative medicine, the more levels of government, academic institutions, and conventionally- trained physicians will feel compelled to work collaboratively. Pioneers like Dr Gannage have allowed the path of a newer generation of integrated healthcare provider to be much easier. Their tireless efforts have made integrative medicine a household name, a far cry from the state of practice for such practitioners a decade or two ago.
Arizona Medical and Natural Health Clinic
Arizona Medical and Natural Health Clinic
The power of scope of practice
Naturopathic medicine in Canada has undergone an exponential magnitude of progress in just a handful of years. The country has welcomed its second institution granting the ND diploma (Boucher Institute of Naturopathic Medicine in British Columbia), the province of British Columbia has secured legislative standing and subsequent scope of practice privileges unparalleled by any other jurisdiction in the country, Ontario has initiated the process of legislatively placing the profession into the Regulated Healthcare Practitioners Act likewise accompanied by revisions to scope of practice, and several other provinces are simultaneously making major regulatory strides. During this key time in the evolution of the profession in Canada, we at IHP felt it appropriate to showcase the work of a Canadian- trained ND practicing in Arizona. We hope the individuals charged with shaping the future of our profession in Canada take a moment to reflect on the Arizona Medical and Natural Health Clinic and recognize the power of scope of practice.
Dr Erika Sykes- McKeen graduated from the Canadian College of Naturopathic Medicine in 2004, and before long had successfully integrated herself in a facility of 20+ MD’s at the Westmount Community Health Centre in Hamilton, Ontario. Her passion, integrity, and mastery of naturopathic systems saw her practice build quickly. Her unparalleled mastery of pharmacology, laboratory diagnosis, differential diagnosis, and physical exam made the transition of practice alongside conventionally- trained physicians an easy one. Before long, the desire to begin a family saw her pack- up her Hamilton facility and move to Phoenix Arizona, never to see the snow again!
 Erika comes from a long line conventionally- trained physicians. When asked why she chose to pursue naturopathic medicine, she beams a huge smile and replies “well, MD’s are kind of limited in what they can do. As a naturopathic doctor, I can do everything an MD can, AND a whole bunch of other stuff that they never even learn about!” The state of Arizona has the most progressive legislative environment for ND’s of any jurisdiction in North America. They are recognized as primary healthcare providers, and provided a full formulary of prescription medications that is virtually unlimited. The title of a practicing naturopath in Arizona is not ND, but NMD, or Natural Medical Doctor.
Erika comes from a long line conventionally- trained physicians. When asked why she chose to pursue naturopathic medicine, she beams a huge smile and replies “well, MD’s are kind of limited in what they can do. As a naturopathic doctor, I can do everything an MD can, AND a whole bunch of other stuff that they never even learn about!” The state of Arizona has the most progressive legislative environment for ND’s of any jurisdiction in North America. They are recognized as primary healthcare providers, and provided a full formulary of prescription medications that is virtually unlimited. The title of a practicing naturopath in Arizona is not ND, but NMD, or Natural Medical Doctor.
The first impression of entering the Arizona Medical and Natural Health Clinic is that you have arrived at the facility of a neurosurgeon. 50” plasma televisions line almost any wall you encounter, running a series of health tips, recipe ideas, and the occasional profile of a product offered at the facilities adjacent natural health product dispensary. The floors are gleaming stone, the ambiance that of a modernized home of luxury. The 5000sqft facility houses 12 treatment rooms. Team members include four NMD’s, two chiropractors, a registered nurse, and two massage therapists.
The facility serves as a one- stop shop of wellness, offering services to the severely ill seeking solutions for chronic, debilitating illness, yet also providing a thriving selection of beauty and cosmetic services. Value- added offerings of the clinic include an infrared sauna, cold laser therapy, and an elegant IV lounge offering services of prescription medications, chelation therapy, vitamin/ mineral cocktails, and prolotherapy/ mesotherapy. Preservativefree immunizations are also made available.
A well- developed array of integrative diagnostic tests are routinely implemented by the team, including IgG food sensitivity testing, salivary cortisol profiles, salivary hormone testing, urinary metabolic profiles, and complete stool analysis. The facility has established a highly successful beauty and cosmetic platform offering services that include botox and filler (juvederm) injections, laser hair removal, accent laser, and microdermabrasion. The massage team offers a highly diversified selection of offerings, including sport massage, pregnancy massage, deep tissue massage, Swedish massage, hot stone massage, and reflexology.
Arizona represents a rare opportunity to observe the impact to community of ND’s afforded their full scope of practice. As Christoph Kind, president of the BCNA told IHP shortly after the province of British Columbia secured prescription rights for ND’s, “the right to prescribe legitimizes the decision not to prescribe”. When allowed to exercise the full skill set an ND is trained in, practitioners of naturopathic medicine quickly, effectively, and successfully integrate themselves into their respective communities as primary healthcare providers of unquestioned quality. It is not surprising to witness a preference of the general public to visit an ND in favour of an MD when there does not exist a fiscal incentive to visit the MD, or more appropriately the fiscal burden of visiting an ND.
The NMD’s of the Arizona Medical and Natural Health Clinic are quick to highlight their need for access to prescription medications. They are necessary and required tools of practice. But as the majority of ND’s would agree, systems of practice alter when tools other than pharmacotherapy are added to a practitioner’s medicine bag. If you think a prescription is the only possible thing that can help a patient, you are going to prescribe. When you are trained to recognize the impact diet, lifestyle, nutritional, nutraceutical, and botanical agents can have on health, you are likely to try them first, and defer to a prescription medication only if evidence- based natural treatments have failed.
IHP is grateful to Dr Erika Sykes- McKeen and the Arizona Medical and Natural Health Clinic for allowing us to showcase their facility to you. The clinic has surpassed expectations and set the bar of an outpatient primary care facility. During this period of rapid evolution of the profession, regulators and those charged with writing new legislation would be well served to take the time to investigate the success with which ND’s operate under more appropriate scope of practice regulation. In times of physician shortages reaching almost panic levels, awarding a thriving profession deserved ability to deliver safe, effective, and costeffective medical care must be made a top priority.
L-theanine
L-theanine
Neuropsychiatric applications
The amino acid L-theanine is considered to be unique because it is found almost exclusively in Camellia sinensis, a popular tea plant (Monograph 2005). Theanine is the dominant amino acid found in teas leaves and is primarily responsible for the flavor of teas (Juneja 1999). Teas are one of the most common beverages globally consumed. The popularization of tea is said to be due to its ‘relaxation’ type effects (Juneja 1999). Interest in theanine was sparked due to its abundance in traditional diets (particularly in the Japanese diet) and its positive effects such as the ‘relaxed but alert’ state associated with consuming tea. Current literature, including human and non-human studies, suggests that L-theanine is a psychoactive compound that improves cognitive function and reduces anxiety (Gomez-Remirez 2007, Kimura 2007, Kobayashi 1998, Lu 2004, Nobre 2008, Yokogoshi 1998). Studies indicate L-theanine is also neuroprotective, possesses anti-cancer and immune supporting properties, and may help control hypertension (Bryan 2008, Juneja 1999, Monograph 2005, Nathan 2006). Each one of these areas warrants consideration. However, this article will focus on evidence that links L-theanine with cognitive and mood related effects.
Physiology
L-theanine (N-ethyl-L-glutamine) is a derivative of glutamic acid which is also a neurotransmitter in humans (Yokogoshi 1998). It is a water-soluble compound and is absorbed in the small intestine by a Na+ -coupled transporter found in the brush boarder membrane (Monograph 2005, Unno 1999). In the central nervous system (CNS), L-theanine crosses the blood-brain barrier through a leucinepreferring transport system in a concentration-dependant manner (Nathan 2006, Terashima 1999, Yokogoshi 1998). L-theanine is eliminated through hydrolysis to glutamic acid and thylamine in the kidneys (Unno 1999). Peak serum and hepatic concentrations of L-theanine occur 1 hr after administration, while brain levels peak after 5 hrs (Terashima 1999). Concentrations then decline gradually over 24 hrs following administration (Terashima 1999).
Toxicology
Borzelleca (2006) conducted a study to evaluate the safety of L-theanine. Rats were administered L-theanine via the diet at various concentrations (adjusted weekly) to sustain target doses of 1500, 3000, and 4000 mg/kg bw/day for 13 weeks. The monitored parameters of adverse effects included; behavior, morbidity, mortality, body weight, food consumption, clinical chemistry, hematology, and unrinalysis. The results were as follows:
• No treatment-related deaths
• Lower body weights in both female and male animals were observed at 3000 and 4000mg/kg bw/day doses (p< 0.05) but was determined to be due to significantly lower food consumption (unpalatability of diet) • No significant behavioural changes
• Reduced locomotor activity in males at weeks 3 and 14 but deemed unrelated to treatment (biological significance is unknown)
• No treatment related adverse effects related to clinical pathology were reported.
It was concluded that L-theanine is well tolerated and the LD50 is greater than 5000 mg/kg in rats, exponentially higher than the dose used for therapeutic administration in humans (Borzelleca 2006, Monograph 2005). Mutagenic or carcinogenic risks were not found in animals or bacteria (Borzelleca 2006, Monograph 2005).
Non-Human Studies
Although animal studies are not directly clinically applicable, the early in vivo evidence provides an understanding of the basic mechanism by which L-theanine exerts it neuropsychiatric effects, and the basis upon which further human investigations were conducted. A brief discussion of these findings is provided here.
Yokogoshi (1998) investigated (i) the transport system of theanine through the blood-brain barrier and (ii) the effects of theanine on brain monoamine (eg. dopamine) concentrations and the activity of dopaminergic neurons. It was determined that theanine competitively used the leucine- preferred transport system (LTS) to cross the blood brain barrier, since high concentrations of theanine appeared to affect levels of other amino acids that used the LTS. Non-LTS amino acid concentrations were unaffected. Catecholamines and 5-hydroxyindole levels were increased after theanine administration. This was especially noticed in the striatum where dopaminergic neurons are highly concentrated. Dopamine concentrations increased following theanine injection, but further investigation was suggested in order to more directly correlate theanine administration with dopaminergic effects. Collectively, these findings suggest that L-theanine may alter brain monoamine concentrations, thereby affecting cognitive function and mood. In this study serotonin concentration in the stratum and hippocampus were elevated, however, a subsequent study indicated a general decrease of serotonin release (Bryan 2008). It was suggested that theanine decreases serotonin levels only when caffeine is present (Bryan 2008, Nathan 2006, Yokogoshi 1998).
Lending support to such a caffeine-theanine interaction, another study demonstrated that theanine has antagonistic effects on caffeine’s stimulatory action. Kakuda (2000) monitored brain waves, using electroencephalography (EEG), after I.V. administration of caffeine and theanine in rats. The minimum dose of five micromol/kg body weight was determined to be the level at which caffeine was stimulating according to brain wave monitoring. Injection of theanine at a similar concentration inhibited caffeine’s stimulatory action; however, injection of theanine alone at levels of 1 -2 micromol/kg caused excitatory effects. These results suggest that theanine has inhibitory effects on caffeine’s stimulatory actions, but dose- dependant excitatory effects when administered alone. At the human level, this interaction has been confirmed by some studies while not in others, such as Rogers (2008) wherein theanine had no antagonistic effect on caffeine with respect to jitteriness, alertness or mood (please see Table 1 for summary).
Human Studies
Thirteen human studies were identified of L- theanine for impact on neuropsychiatric outcomes (see Table 1 and Table 2).
A novel trial administered 400mg/day of L- theanine as addon to antipsychotic treatment in randomized, double- blind, placebo controlled fashion to 60 patients with a DSM-IV diagnosis of schizophrenia or schizoaffective disorder (Ritsner 2011). Outcome measures included the Positive and Negative Syndrome Scale (PANSS), the Hamilton Anxiety Rating Scale (HARS), The Cambridge Neuropsychological Test Automated Battery (CANTAB), side effects and quality of life. Forty patients completed the eight week trial. Last observation carried forward (LOCF) was applied to the statistical analysis, and therefore 12 patients who dropped out of the trial between weeks four and six were included in the analysis.
L- theanine administration achieved significant benefit to the positive scores of the PANSS (p=0.009) compared to placebo, but failed to impact the PANSS negative subscales (p>0.05). On the five-Factor PANSS model the following was observed:
• Significant decrease of positive scores from week six onward
• Reduced activation (from week 2 onward)
• Reduced dysphoric mood (from week 2 onward)
•Reduced autistic preoccupations (from week 8 onward) On the HARS scores, improvement was seen on the second week compared to placebo. The results were as follows:
• Reduced anxious mood
• Reduced tension
No treatment- related adverse effects were observed. The authors conclude that co-treatment with antipsychotic therapy and Ltheanine improves anxiety and general psychopathology symptoms. Since the dopamine system has been implicated in schizophrenia, these findings would seem to support the preclinical indications that L-theanine acts upon this neurotransmitter pathway.
Lu and colleagues (2004) compared the anxiolytic effects of L-theanine to the benzodiazepine alprazolam. Sixteen patients were randomized to 200mg L- theanine, 1mg alprazolam, or placebo in double blind fashion. Participants were subjected to an experimentally induced anxiety related condition and also assessed under a relaxed condition. Acute effects of alprazolam and L-theanine were assessed by subjective self-reports of anxiety (Beck Anxiety Inventory (BAI), Visual Analogue Mood Scale (VAMS), and The State-Trait Anxiety Inventory (STAI)). The study determined that both L-theanine and alprazolam had no effect on subjective anxiety levels under the induced anxiety condition. On the VAMS tranquil-trouble subscale during resting conditions, L-theanine significantly reduced subjective anxiety when compared to alprazolam (p<0.05) and placebo (p<0.05). Therefore, L- theanine was able to produce a subjective anxiolytic outcome superior to alprazolam and placebo in a resting state.
Several human trials have investigated the impact of L- theanine on brain wave activity through application of EEG analysis. Reproducibly, L- theanine administration has been demonstrated to increase alpha wave activity (Gomez-Ramirez 2007, Kobayashi 1998, Nobre 2008) (see Table 1). Increased alpha wave activity is thought to indicate a state of wakeful relaxation. It has been associated with a general state of mental alertness or arousal; creativity; enhanced performance under stress; and improved cognitive function (learning and concentration), as well as decreased perceived anxiety (Bryan 2008, Gomez-Ramirez 2007, Mason 2001, Yokogoshi 1998).
Conclusion
L-theanine appears to be a safe, natural option for improving cognitive functioning such as attention, learning, and concentration. Evidence also suggests mood modulating effects (e.g. ‘relaxation’, sedation) which could be beneficial for improving anxiety related symptoms. Therapeutic dosage ranges from 50mg to 200mg and appears to complement conventional therapies well. Although the evidence to date is promising, the human level studies are minimal at present. Further human clinical trials are needed. In the mean time, dietary sources of L-theanine (green tea, black tea, white tea) appear to be beneficial to health and can be considered as a simple way to enhance or improve cognitive function and relaxation; administration of L-theanine can be considered as a complementary treatment for anxiety.
References
Bryan J. Psychological effects of dietary components of tea: caffeine and L-theanine. Nutr Rev. 2008; 66(2): 82-90.
Borzelleca JF, Peters D, Hall W. A 13-week dietary toxicity and toxicokinetic study with l-theanine in rats. Food Chem Toxicol. 2006 Jul;44(7):1158-66.
Dimpfel W, Kler A, Kriesl E, Lehnfeld R, Keplinger-Dimpfel IK. Source density of the human EEG after ingestion of a drink containing decaffeinated extract of green tea enriched with L-theanine and theogallin. Nutr Neurosci. 2007; 10(3-4): 169-180.
Einöther SJL, Martens VEG,Rycroft JA, De Bruin EA. L-Theanine and caffeine improve task switching but not intersensory attention or subjective alertness. Appetite. 2010; 54: 406-409.
Giesbrecht T, Rycroft JA, Rowson MJ, De Bruin EA. The combination of L-theanine and caffeine improves cognitive performance and increases subjective alertness. 2010: 13(6): 283-290.
Gomez-Ramirez M, Higgins BA, Rycroft JA, Owen GN, Mahoney J, Shpaner M, Foxe JJ. The deployment of intersensory selective attention: a high-density electrical mapping study of the effects of theanine. Clin Neuropharmacol. 2007; 30(1): 25-38.
Haskell CF, Kennedy DO, Milne AL, Wesnes KA, Scholey AB. The effects of L-theanine, caffeine and their combination on cognition and mood. Biol Psychol. 2008; 77: 113-122.
Juneja LR, Chu DC, Okubo T, Nagato Y, Yokogoshi H. L-theanine – a unique amino acid of green tea and its relaxation effect in humans. Trends Food Sci Tech. 1999; 10: 199-204.
Kakuda T, Nozawa A, Unno T, Okamura N, Okai O. Inhibiting effects of theanine on caffeine stimulation evaluated by EEG in the rat. Biosci Biotechnol Biochem. 2000; 64(2): 287-293.
Kelly SP, Gomez-Ramirez M, Montesi JL, Foxe JJ. L-theanine and caffeine in combination affect human cognition as evidenced by oscillatory alpha-band activity and attention task performance. J Nutr. 2008; 138: 1572S-1577S.
Kimura K, Ozeki M, Juneja LR, Ohira H. L-theanine reduces psychological and physiological stress responses. Biol Psychol. 2007; 74: 39-45.
Kobayashi K, Nagato Y, Aoi N, Juneja LR, Kim M, Yamamoto T, Sugimoto S. Effects of L-theanine on the release of α-brain waves in human volunteers. Nippon Nōgei Kagakukaishi. 1998; 72:153-157. (Abstract). Accessed on Google Scholar, March 2011.
Lu K, Gray MA, Oliver C, Liley DT, Harrison BJ, Bartholomeusz CF, Phan KL, Nathan PJ. The acute effects of L-theanine in comparison with alprazolam on anticipatory anxiety in humans. Hum Psychopharmacol Clin. 2004; 19: 457-465.
Mason R. 200 mg of zen: L-theanine boosts alpha waves, promotes alert relaxation. Alternative & Complimentary Therapies. 2001; 91-95.
Monograph. L-theanine. Altern Med Rev. 2005; 10(2): 136-138.
Nathan PJ, Lu K, Gray M, Oliver C. The neuropharmacology of L-theanine (N-Ethyl-L-Glutamine): a possible neuroprotective and cognitive enhancing agent. Journal of Herbal Pharmacology. 2006; 6(2): 21-30.
Nobre AC, Rao A, Owen GN. Ltheanine, a natural constituent in tea, and its effect on mental state. Asia Pac J Clin Nutr. 2008; 17(S1): 167-168.
Owen GN, Parnell H, De Bruin EA, Rycroft JA. The combined effects of L-theanine and caffeine on cognitive performance and mood. Nutr Neurosci. 2008; 11(4): 193- 198.
Ritsner MS, Miodownik C, Ratner Y, Shleifer T, Mar M, Pintov L, Lerner V. L-theanine relieves positive, activation, and anxiety symptoms in patients with schizophrenia and schizoaffective disorder: an 8-week, randomized, double-blind, placebo-controlled, 2-center study. J Clin Psychiat. 2011; 72(1): 34-42.
Rogers PJ, Smith JE, Heatherly SV, Pleydell-Pearce CW. Time for tea: mood, blood pressure and cognitive performance effects of caffeine and theanine administered alone and together. Psychopharmacology. 2008; 195: 569-577.
Terashima T, Takido J, Yokogoshi H. Time-dependent changes of amino acids in the serum, liver, brain, and urine of rats administered with theanine. Biosci. Biotechnol. Biochem: 1999: 63:615-618
Unno T, Suzuki Y, Kakuda T, Hayakawa T, Tsuge H. Metabolism of theanine, γ-glutamylethylamide, in rats. J Agric Food Chem; 1999: 1593-1596.
Yokogoshi H, Kobayashi M, Mochizuki M, Terashima T. Effect of theanine, γ-glutamylethylamide, on brain monoamines and striatal dopamine release in conscious rats. Neurochem Res. 1998; 23(5): 667-673.
Polycystic ovary syndrome
Polycystic ovary syndrome
Clinical considerations and therapeutic options
Polycystic ovary syndrome (PCOS) is a complex condition of metabolic and endocrine dysfunction. Estimates of its prevalence in women range from 6-18% worldwide (Azziz 2009, Kumarapeli 2008, March 2010), making it one of the most common human disorders (Goodarzi 2011).
The central features of PCOS include clinical hyperandrogenism or biochemical hyperandrogenemia, oligoanovulation and polycystic ovaries (Goodarzi 2011). Three diagnostic schemes appearing in the current literature identify a wide range of PCOS phenotypes involving diverse combinations of hyperandrogenism, hyperandrogenemia and ovarian dysfunction (March 2010). Diagnostic methods and criteria have evolved considerably since PCOS was first described in 1935 by Stein and Leventhal (Azziz 2009), but agreement has yet to be reached as to which symptoms truly define this syndrome.
Clinical presentation
Hyperandrogenism can appear clinically as hirsutism, acne, and androgenic alopecia. Of these three symptoms, hirsutism appears to be most predictive of PCOS, with 75-80% of hirsute women fitting diagnostic criteria for this syndrome (Azziz 2009). Ethnic variation does exist, and fewer East Asians with PCOS present with unwanted hair growth than white or black women (Goodarzi 2011). Clinical symptoms of hyperandrogenism may be supported by laboratory findings (hyperandrogenemia) which may include elevated testosterone and dehydroepiandrosterone sulfate (DHEAS). However, up to 40% of women with PCOS are estimated to have androgen levels within the normal range (Azziz 2009).
Menstrual dysfunction in the form of oligomenorrhea or amenorrhea occurs in 70-80% of women with PCOS (Teede 2010). Dysfunction may not be easily appreciated in women with regularly-occurring but anovulatory periods, and may only be revealed through comprehensive hormonal assessment.
Although polycystic ovaries are present in an estimated 80% of women with PCOS, their identification is not required for diagnosis (March 2010). As asymptomatic polycystic ovaries may be found in up to 30% of the population (Azziz 2009), changes in ovarian morphology, while common, are not an essential feature of PCOS, despite its name.
Diagnosis of exclusion
Alternative pathologies must be excluded before a diagnosis of PCOS may be made. Disturbances in thyroid function can cause changes to the menstrual cycle (Krassas 2000) while hyperprolactinemia is associated with infertility and amenorrhea (Patel 2007). Non-classic congenital adrenal hyperplasia may not appear until the reproductive years, and is associated with hyperandrogenism (Young 2010), while hirsutism may be a sign of Cushing’s syndrome (Shibli-Rahhal 2006). Clinicians must also be attuned to the possibility that these conditions may exist concomitantly.
Associated conditions
Other signs are often associated with PCOS and exacerbate the impact of this syndrome, although they are not required for diagnosis. The most important of these is insulin resistance, and associated hyperinsulinemia. Excess insulin drives endocrine derangement by stimulating androgen production (Bergh 1993), and decreasing hepatic production of sex hormone binding globulin (SHBG) (Goodarzi 2011). Insulin resistance is less commonly observed in lean women with PCOS (Stovall 2011) and is worsened by smoking (Cupisti 2010).
Obesity exacerbates the clinical features of PCOS by promoting insulin resistance (Teede 2010) and unopposed estrogen production (Azziz 2009). The link between PCOS and obesity is amplified further, as insulin-driven hyperandrogenemia, in turn, contributes to abdominal adiposity (Tolino 2005). While increased BMI only marginally increases the risk of developing PCOS (Yildiz 2008), and while as many as 50% of women with PCOS are not obese (Goodarzi 2011), obesity has a significant, negative impact on quality of life in this population (Jones 2008).
Fertility challenges are natural sequelae to any condition affecting ovarian function. PCOS is responsible for up to 70% of cases of anovulatory infertility (Brassard 2008), although as many as 60% of women with PCOS may be able to conceive (Teede 2010). Obesity independently contributes to decreased fertility in women with PCOS (Marquard 2010), and weight loss may improve the chances of conceiving (Crosignani 2003). During pregnancy, women with PCOS are at increased risk for pre-eclampsia, hypertension and gestational diabetes (Goodarzi 2011).
Associated risks
The long-term health effects of PCOS must also be of concern to the clinician:
• PCOS-associated insulin resistance increases the risk of developing Type 2 diabetes, especially in women who are also obese.
• Dyslipidemia, a common finding in US women with PCOS, increases the risk of cardiovascular disease.
• The metabolic syndrome, which incorporates abnormalities in both insulin and cholesterol metabolism, occurs more frequently in US women with PCOS than those without (Goodarzi 2011).
• Increased cardiovascular risk is a concern, even in lean women with PCOS (Costa 2010).
The significance of these risk factors is highlighted by reports of increased incidence of cardiovascular events and decreased event-free survival in this population (Shaw 2008).
Preliminary data show that rates of endometrial cancer may be slightly higher in women with PCOS (Chittenden 2009), and were significantly associated with high BMI, hirsutism and very irregular periods in an Australian case control study (Fearnley 2010). Investigations continue into associations between PCOS and other gynaecological cancers.
Treatment of PCOS – Lifestyle interventions
A recent systematic review (Moran 2011) found that exercise, either alone or in combination with caloric restriction, addressed the exacerbating conditions associated with PCOS. Participants in the trials reviewed lost weight, decreased abdominal obesity, and saw improvements in insulin resistance. A meta-analysis was not performed, but the results appeared to be consistent throughout the trials. None of the included studies reported on reproductive outcomes, but observed improvements in both clinical and biochemical signs of hyperandrogenism suggest an additional impact upon endocrine parameters.
BMI, insulin resistance and serum hormone levels
Other studies confirm these results. Karimzadeh (2010) compared lifestyle changes (caloric deficit of 500kcal/day, combined strength/aerobic training for 20-60 minutes, 3-5 times per week) to pharmacological therapy (metformin and clomiphene citrate (CC)), and reported significant improvements in waist circumference, insulin, LDL and SHBG with the lifestyle intervention. Another small study (n=20) comparing regular aerobic exercise (30 minutes of exercise at 60-70% of the maximal oxygen consumption (VO2max), 3 times per week) to a high protein (35%), hypocaloric diet (deficit of 800kcal/day) reported improvement in both groups in BMI, insulin resistance and waist-hip ratio (WHR) (Palomba 2008).
Weight loss alone, achieved through strict caloric restriction, has been shown to improve insulin (Stamets 2004), SHBG and testosterone levels (Tolino 2005) in women with PCOS. Additionally, nutritional counselling and exercise therapy (90 minutes of combined aerobic/resistance training, 3 times per week) in the absence of weight loss was shown to benefit waist circumference and fasting insulin levels (Bruner 2006) through improved body composition. A combination of these therapies can reasonably be expected to have an additive and positive effect in PCOS patients.
Reproductive outcomes
Improvements in rates of ovulation and pregnancy may also be anticipated with lifestyle changes. A small, prospective trial of 33 overweight women with PCOS, treated with caloric restriction and exercise recommendations, saw improvements in ovulatory function and morphology, and 30% of participants (10/33) achieved pregnancy (Crosignani 2003). Karimzadeh (2010), found parallel rates of ovulation (55-66%) and pregnancy (12-20%) in all treatment arms (described above). Otta (2010) reports statistically significant amelioration of menstrual cycling with caloric restriction and exercise, both in combination with metformin and with placebo (between-group differences were not reported).
Treatment of PCOS – Inositol
Inositol phosphoglycan (IPG) is an insulin mediator influencing the uptake of glucose in the body (Baillargeon 2010). D-chiro inositol (DCI) is an important component of IPG, and defects in the DCI-IPG system have been linked with insulin resistance (Galazis 2011). In women with PCOS, DCI-IPG is less responsive to insulin challenge than in normal controls, and it is speculated that the uncoupling of DCI-IPG with insulin levels may contribute to insulin resistance in this population (Galazis 2011). Given the potential for hyperinsulinemia to exacerbate PCOS, improving insulin sensitivity is an important therapeutic goal.
Studies have investigated the impact of supplementation with inositol, both as DCI or its precursor, myo-inositol (MYO) (Papaleo 2009), in women with PCOS. Daily dosing in published research ranges from 200mg-1200mg of DCI and 2g-4g of MYO.
Although studies are small and heterogeneous, recent reviews (Fritz 2009, Galazis 2011) have described benefit from inositol therapy. The proposed effects of inositol supplementation are reviewed below.
Inositol and insulin resistance
Inositol does appear to benefit glucose metabolism in women with PCOS, likely through its interaction with IPG. Evidence of increased insulin sensitivity (Costantino 2009), decreased insulin resistance (Genazzani 2008) or improved glucose tolerance (Nestler 1999) has been seen in trials using either form of inositol. One researcher also attributed improvements in BMI to inositol, providing a second mechanism through which insulin resistance may be addressed (Gerli 2003, Gerli 2007).
Benefit to clinical features of PCOS
Clinical hyperandrogenism, evidenced by hirsutism and/or acne, improved with 4g MYO supplementation over a 6 month period (Zacche 2009). Hyperandrogenemia, assessed through serum testosterone and DHEAS, was significantly improved by the administration of both MYO (Costantino 2009, Genazzani 2008, Zacche 2009) and DCI (Iuorno 2002, Nestler 1999).
A series of small randomized, controlled trials (RCT) have found significant benefit of both forms of inositol in restoring ovulatory cycles in comparison to placebo, or to metformin (Costantino 2009, Gerli 2007, Nestler 1999 Raffone 2010). These improved rates of ovulation appear to translate into fewer oocytes of poor quality (Ciotta 2010, Papaleo 2009) and a trend towards increased frequency of pregnancies (Morgante 2011), although this latter outcome has yet to reach statistical significance in recent RCTs (Papaleo 2009, Raffone 2010).
Treatment of PCOS – N-acetylcysteine (NAC)
NAC has been shown to affect glucose metabolism (Ammon 1992) and may function as an insulin-sensitizing agent in women with PCOS, possibly through its influence upon intracellular levels of glutathione (GSH) (Fulghesu 2002). Two small trials of NAC as a monotherapy demonstrated improvement in insulin sensitivity following 4-6 weeks of treatment at a dose of 1.8g per day (Fulghesu 2002, Kilic-Okman 2004), although a third trial did not support these findings (Elnashar 2007). Further studies are needed to identify factors impacting the effect of this therapy.
NAC and ovulation induction
The benefit of NAC as an insulin-sensitizing agent may be most apparent in its use as an adjunct to clomiphene citrate (CC). Insulin-sensitizing agents are used to amplify response in resistant patients (Nestler 2000, Rizk 2005), and NAC has shown some promise in this application. Two recent trials found a significant increase in ovulation rates compared to controls (49.3% vs. 1.3% (Rizk 2005) and 52.1% vs. 17.9% (Badawy 2007) when 1.2g of NAC was administered with 100mg of CC.
Other insulin-sensitizing agents such as metformin have been shown to confer greater benefit in terms of ovulation, pregnancy and miscarriage incidence (Abu Hashim 2010), but NAC may still be a valuable option when these agents cannot be used. NAC may also exert its influence through insulin-independent pathways, having the potential for positive effects on vasculature, immune parameters and cervical mucous (Badawy 2007).
The hormonal effects of NAC may be significant in a PCOS-affected population as this agent has been shown to significantly decrease testosterone levels (Elnashar 2007) and increase SHBG (Fulghesu 2002, Kilic-Okman 2004). Fulghesu reported the greatest benefit to insulin levels and hormonal parameters in subjects that were hyperinsulinemic at baseline, indicating that effect of NAC on hormonal levels may be insulin-mediated.
Conclusion
PCOS, a complex, multifaceted condition, presents with varying degrees of endocrine and metabolic dysfunction and poses significant risk of short- and long-term morbidity. Caloric restriction and increases in aerobic and resistance exercise can powerfully impact BMI, WHR, insulin resistance and reproductive outcomes. Dietand lifestyle-based therapies are considered first-line, especially in overweight and obese patients (Franks 2011).
Pharmacological therapies may be used to prompt ovulation and menstruation, antagonize androgens, improve insulin sensitivity, and/or address cardiovascular risk factors. Recent research supports the use of inositol and NAC, both as monotherapies and as adjunctive agents to pharmacotherapy, with demonstrated benefits to insulin sensitivity and hormonal parameters. The wide range of clinical presentations and patient concerns in PCOS provide the clinician with numerous therapeutic targets requiring a truly comprehensive approach to care.
References
Abu Hashim H, Anwar K, El-Fatah RA. N-acetyl cysteine plus clomiphene citrate versus metformin and clomiphene citrate in treatment of clomiphene-resistant polycystic ovary syndrome: a randomized controlled trial. J Womens Health (Larchmt). 2010 Nov;19(11):2043-8.
Ammon HP, Müller PH, Eggstein M, Wintermantel C, Aigner B, Safayhi H, Stützle M, Renn W. Increase in glucose consumption by acetylcysteine during hyperglycemic clamp. A study with healthy volunteers. Arzneimittelforschung. 1992 May;42(5):642-5. (abstract)
Azziz R, Carmina E, Dewailly D, Diamanti-Kandarakis E, Escobar-Morreale HF, Futterweit W, Janssen OE, Legro RS, Norman RJ, Taylor AE, Witchel SF; Task Force on the Phenotype of the Polycystic Ovary Syndrome of The Androgen Excess and PCOS Society. The Androgen Excess and PCOS Society criteria for the polycystic ovary syndrome: the complete task force report. Fertil Steril. 2009 Feb;91(2):456-88.
Badawy A, State O, Abdelgawad S. N-Acetyl cysteine and clomiphene citrate for induction of ovulation in polycystic ovary syndrome: a cross-over trial. Acta Obstet Gynecol Scand. 2007;86(2):218-22.
Baillargeon JP, Iuorno MJ, Apridonidze T, Nestler JE. Uncoupling between insulin and release of a D-chiro-inositol-containing inositolphosphoglycan mediator of insulin action in obese women With polycystic ovary syndrome. Metab Syndr Relat Disord. 2010 Apr;8(2):127-36.
Bergh C, Carlsson B, Olsson JH, Selleskog U, Hillensjö T. Regulation of androgen production in cultured human thecal cells by insulin-like growth factor I and insulin. Fertil Steril. 1993 Feb;59(2):323-31.
Brassard M, AinMelk Y, Baillargeon JP. Basic infertility including polycystic ovary syndrome. Med Clin North Am. 2008 Sep;92(5):1163-92, xi.
Bruner B, Chad K, Chizen D. Effects of exercise and nutritional counseling in women with polycystic ovary syndrome. Appl Physiol Nutr Metab. 2006 Aug;31(4):384-91.
Chittenden BG, Fullerton G, Maheshwari A, Bhattacharya S. Polycystic ovary syndrome and the risk of gynaecological cancer: a systematic review. Reprod Biomed Online. 2009 Sep;19(3):398-405.
Ciotta L, Stracquadanio M, Pagano I, Carbonaro A, Palumbo M, Gulino F. [Effects of inositol on oocyte quality in patients affected with polycystic ovary syndrome.] Minerva Ginecol. 2010 Dec;62(6):525-531. [Article in Italian]
Costa EC, de Sá JC, Soares EM, Lemos TM, Maranhão TM, Azevedo GD. [Evaluation of cardiovascular risk by the LAP index in non-obese patients with polycystic ovary syndrome]. Arq Bras Endocrinol Metabol. 2010 Oct;54(7):630-5. [Article in Portuguese] (abstract)
Costantino D, Minozzi G, Minozzi E, Guaraldi C. Metabolic and hormonal effects of myo-inositol in women with polycystic ovary syndrome: a double-blind trial. Eur Rev Med Pharmacol Sci. 2009 Mar-Apr;13(2):105-10.
Crosignani PG, Colombo M, Vegetti W, Somigliana E, Gessati A, Ragni G. Overweight and obese anovulatory patients with polycystic ovaries: parallel improvements in anthropometric indices, ovarian physiology and fertility rate induced by diet. Hum Reprod. 2003 Sep;18(9):1928-32.
Cupisti S, Häberle L, Dittrich R, Oppelt PG, Reissmann C, Kronawitter D, Beckmann MW, Mueller A. Smoking is associated with increased free testosterone and fasting insulin levels in women with polycystic ovary syndrome, resulting in aggravated insulin resistance. Fertil Steril. 2010 Jul;94(2):673-7.
Elnashar A, Fahmy M, Mansour A, Ibrahim K. N-acetyl cysteine vs. metformin in treatment of clomiphene citrate-resistant polycystic ovary syndrome: a prospective randomized controlled study.Fertil Steril. 2007 Aug;88(2):406-9.
Fearnley EJ, Marquart L, Spurdle AB, Weinstein P, Webb PM; Australian Ovarian Cancer Study Group and Australian National Endometrial Cancer Study Group. Polycystic ovary syndrome increases the risk of endometrial cancer in women aged less than 50 years: an Australian case-control study. Cancer Causes Control. 2010 Dec;21(12):2303-8.
Franks S. When should an insulin sensitizing agent be used in the treatment of polycystic ovary syndrome? Clin Endocrinol (Oxf). 2011 Feb;74(2):148-51.
Fritz H. Polycystic ovary syndrome: role of inositol in PCOS management. Integrated Healthcare Practitioners 2009 Oct: 82-87.
Fulghesu AM, Ciampelli M, Muzj G, Belosi C, Selvaggi L, Ayala GF, Lanzone A. N-acetylcysteine treatment improves insulin sensitivity in women with polycystic ovary syndrome. Fertil Steril. 2002 Jun;77(6):1128-35.
Galazis N, Galazi M, Atiomo W. d-Chiro-inositol and its significance in polycystic ovary syndrome: a systematic review. Gynecol Endocrinol. 2011 Apr;27(4):256-62.
Genazzani AD, Lanzoni C, Ricchieri F, Jasonni VM. Myo-inositol administration positively affects hyperinsulinemia and hormonal parameters in overweight patients with polycystic ovary syndrome. Gynecol Endocrinol. 2008 Mar;24(3):139-44.
Gerli S, Mignosa M, Di Renzo GC. Effects of inositol on ovarian function and metabolic factors in women with PCOS: a randomized double blind placebo-controlled trial. Eur Rev Med Pharmacol Sci. 2003 Nov-Dec;7(6):151-9.
Gerli S, Papaleo E, Ferrari A, Di Renzo GC. Randomized, double blind placebo-controlled trial: effects of myo-inositol on ovarian function and metabolic factors in women with PCOS. Eur Rev Med Pharmacol Sci. 2007 Sep-Oct;11(5):347-54.
Goodarzi MO, Dumesic DA, Chazenbalk G, Azziz R. Polycystic ovary syndrome: etiology, pathogenesis and diagnosis. Nat Rev Endocrinol. 2011 Apr;7(4):219-31. Epub 2011 Jan 25.
Iuorno MJ, Jakubowicz DJ, Baillargeon JP, Dillon P, Gunn RD, Allan G, Nestler JE. Effects of d-chiro-inositol in lean women with the polycystic ovary syndrome. Endocr Pract. 2002 Nov-Dec;8(6):417-23.
Jones GL, Hall JM, Balen AH, Ledger WL. Health-related quality of life measurement in women with polycystic ovary syndrome: a systematic review. Hum Reprod Update. 2008 Jan-Feb;14(1):15-25.
Karimzadeh MA, Javedani M. An assessment of lifestyle modification versus medical treatment with clomiphene citrate, metformin, and clomiphene citrate-metformin in patients with polycystic ovary syndrome. Fertil Steril. 2010 Jun;94(1):216-20.
Kilic-Okman T, Kucuk M. N-acetyl-cysteine treatment for polycystic ovary syndrome. Int J Gynaecol Obstet. 2004 Jun;85(3):296-7.
Kumarapeli V, Seneviratne Rde A, Wijeyaratne CN, Yapa RM, Dodampahala SH. A simple screening approach for assessing community prevalence and phenotype of polycystic ovary syndrome in a semi-urban population in Sri Lanka. Am J Epidemiol. 2008 Aug 1;168(3):321-8.
Krassas GE. Thyroid disease and female reproduction. Fertil Steril. 2000 Dec;74(6):1063-70.
March WA, Moore VM, Willson KJ, Phillips DI, Norman RJ, Davies MJ.The prevalence of polycystic ovary syndrome in a community sample assessed under contrasting diagnostic criteria. Hum Reprod. 2010 Feb;25(2):544-51.
Marquard KL, Stephens SM, Jungheim ES, Ratts VS, Odem RR, Lanzendorf S, Moley KH. Polycystic ovary syndrome and maternal obesity affect oocyte size in in vitro fertilization/ intracytoplasmic sperm injection cycles. Fertil Steril. 2010 Nov 10.
Moran LJ, Hutchison SK, Norman RJ, Teede HJ. Lifestyle changes in women with polycystic ovary syndrome. Cochrane Database Syst Rev. 2011 Feb 16;2:CD007506.
Morgante G, Orvieto R, Di Sabatino A, Musacchio MC, De Leo V. The role of inositol supplementation in patients with polycystic ovary syndrome, with insulin resistance, undergoing the low-dose gonadotropin ovulation induction regimen. Fertil Steril. 2011 Feb 5.
Nestler JE. Obesity, insulin, sex steroids and ovulation. Int J Obes Relat Metab Disord. 2000 Jun;24 Suppl 2:S71-3.
Nestler JE, Jakubowicz DJ, Reamer P, Gunn RD, Allan G. Ovulatory and metabolic effects of D-chiro-inositol in the polycystic ovary syndrome. N Engl J Med. 1999 Apr 29;340(17):1314-20.
Otta CF, Wior M, Iraci GS, Kaplan R, Torres D, Gaido MI, Wyse EP.Clinical, metabolic, and endocrine parameters in response to metformin and lifestyle intervention in women with polycystic ovary syndrome: a randomized, double-blind, and placebo control trial. Gynecol Endocrinol. 2010 Mar;26(3):173-8.
Papaleo E, Unfer V, Baillargeon JP, Fusi F, Occhi F, De Santis L. Myo-inositol may improve oocyte quality in intracytoplasmic sperm injection cycles. A prospective, controlled, randomized trial. Fertil Steril. 2009 May;91(5):1750-4.
Patel SS, Bamigboye V. Hyperprolactinaemia. J Obstet Gynaecol. 2007 Jul;27(5):455-9.
Palomba S, Giallauria F, Falbo A, Russo T, Oppedisano R, Tolino A, Colao A, Vigorito C, Zullo F, Orio F. Structured exercise training programme versus hypocaloric hyperproteic diet in obese polycystic ovary syndrome patients with anovulatory infertility: a 24-week pilot study. Hum Reprod. 2008 Mar;23(3):642-50.
Raffone E, Rizzo P, Benedetto V. Insulin sensitiser agents alone and in co-treatment with r-FSH for ovulation induction in PCOS women. Gynecol Endocrinol. 2010 Apr;26(4):275-80.
Rizk AY, Bedaiwy MA, Al-Inany HG. N-acetyl-cysteine is a novel adjuvant to clomiphene citrate in clomiphene citrate-resistant patients with polycystic ovary syndrome. Fertil Steril. 2005 Feb;83(2):367-70.
Shaw LJ, Bairey Merz CN, Azziz R, Stanczyk FZ, Sopko G, Braunstein GD, Kelsey SF, Kip KE, Cooper-Dehoff RM, Johnson BD, Vaccarino V, Reis SE, Bittner V, Hodgson TK, Rogers W, Pepine CJ. Postmenopausal women with a history of irregular menses and elevated androgen measurements at high risk for worsening cardiovascular event-free survival: results from the National Institutes of Health–National Heart, Lung, and Blood Institute sponsored Women’s Ischemia Syndrome Evaluation. J Clin Endocrinol Metab. 2008 Apr;93(4):1276-84.
Shibli-Rahhal A, Van Beek M, Schlechte JA. Cushing’s syndrome. Clin Dermatol. 2006 Jul-Aug;24(4):260-5.
Stamets K, Taylor DS, Kunselman A, Demers LM, Pelkman CL, Legro RS. A randomized trial of the effects of two types of short-term hypocaloric diets on weight loss in women with polycystic ovary syndrome. Fertil Steril. 2004 Mar;81(3):630-7.
Stovall DW, Bailey AP, Pastore LM. Assessment of insulin resistance and impaired glucose tolerance in lean women with polycystic ovary syndrome. J Womens Health (Larchmt). 2011 Jan;20(1):37-43.
Teede H, Deeks A, Moran L. Polycystic ovary syndrome: a complex condition with psychological, reproductive and metabolic manifestations that impacts on health across the lifespan. BMC Med. 2010 Jun 30;8:41.
Tolino A, Gambardella V, Caccavale C, D’Ettore A, Giannotti F, D’Antò V, De Falco CL. Evaluation of ovarian functionality after a dietary treatment in obese women with polycystic ovary syndrome. Eur J Obstet Gynecol Reprod Biol. 2005 Mar 1;119(1):87-93.
Yildiz BO, Knochenhauer ES, Azziz R. Impact of obesity on the risk for polycystic ovary syndrome. J Clin Endocrinol Metab. 2008 Jan;93(1):162-8.
Young J, Tardy V, de la Perrière AB, Bachelot A, Morel Y; French Society of Endocrinology. Detection and management of late-onset 21-hydroxylase deficiency in women with hyperandrogenism. Ann Endocrinol (Paris). 2010 Feb;71(1):14-8. (abstract)
Zacchè MM, Caputo L, Filippis S, Zacchè G, Dindelli M, Ferrari A. Efficacy of myo-inositol in the treatment of cutaneous disorders in young women with polycystic ovary syndrome. Gynecol Endocrinol. 2009 Aug;25(8):508-13.
Bacopa monnieri
Bacopa monnieri
Application for cognitive enhancement
Our population demographics are changing – Statistics Canada has projected that by 2056 between 25 – 30 % of the Canadian population will be over the age of 65 (up from 13.7% in 2006) (Statistics Canada 2010). This has both social and economic implications – from the stand point of cognitive health and performance; it is plausible that cognitive enhancing agents may become increasingly sought after by the otherwise healthy aging adult (Calabrese 2008). A recent (2004) community survey of 2551 adults between 60-64 years of age conducted in Australia showed that 2.8% of the sample reported using a non-prescription drug for memory aid or cognitive enhancement. Bacopa monnieri (B. monnieri) was amongst the top three agents used (Jorm 2004).
Bacopa monnieri, also commonly known as Water Hyssop or Brahmi in India is a member of the Scrophulariaceae plant family (Calabrese 2008). It has ancient historical use as a brain tonic for anxiety, memory, poor cognition and concentration in traditional Ayurvedic medicine; chronicles of its use date back to as early as 100 A.D. (Calabrese 2008, Chowdhuri 2002, Usha 2008). B. monnieri has many other purported actions including analgesic, anti-inflammatory, antipyretic, sedative and anti-epileptic properties, however its use as a memory enhancing agent has predominated (Nathan 2001, Stough 2001).
The following review examines available clinical evidence for the use of B. monnieri for cognitive enhancement, specifically for its use in memory augmentation.
Mechanism of action:
The exact mechanism of B. monnieri on psychoneurological parameters remains unknown (Stough 2001). In vitro as well as in vivo animal studies indicate that the effect of B. monnieri on memory appears to be multifocal and suggests that it includes modulation of the cholinergic system, altered antioxidant status as well as anti stress activity by modulation of HSP70, P450 and superoxide dismutase within the brain (Bhattacharya 2000, Roodnenrys 2002, Stough 2001). Most recently B. monnieri has demonstrated neuroprotective effects and cognitive enhancement in preclinical Alzheimer’s disease models (Limpeanchob 2008, Uabundit 2010).
The cognitive enhancing effects of B. monnieri extracts are attributed to the saponins within the plant, specifically bacosides A and B (Joshua- Allan 2008). Research suggests that the bacosides are the component of the plant that are involved in the reversal of amnesic effects of neurotoxins, scopolamine, phenytoin, electroschock, immobilization stress and diazepam as seen in animal studies (Calabrese 2008, Prabhakar 2008).
A rat study showed that B monnieri extract produced a dose-dependent effect on antioxidants superoxide dismutase, catalase and glutathione peroxidase activity in the frontal cortex, striatum and hippocampus of the brain (Bhattacharya 2000). It has been hypothesized that this effect may in part result in enhancement of the cognitive functions associated with those parts of the brain: memory, executive decision making and information processing (Bhattacharya 2000). It should be noted that the standardization of bacosides in this rat study was to bacoside A at 82% +/- 0.5 % which is significantly higher than that used in human trials (bacoside standardization of 40-55%).
Clinical Evidence:
Bacopa monnieri has been the subject of eight double blind randomized placebo controlled trials (n=498) with the primary objective including assessing the effect of B. monnieri on memory. All but two of the trials were conducted on healthy adults ranging from 18 – 65 years of age and lasting up to 16 weeks in duration. Several of the trials also assessed for safety, tolerability as well as anxiety and depression.
In all of the trials, assessments of cognitive enhancement was conducted through a battery of neuropsychological testing including the Rey Auditory Verbal Learning Test (AVLT), which objectively measures audioverbal and visual memory, and the Wechsler Adult Intelligence Scale, but the types and number of assessments employed varied significantly between studies (Morgan 2010).
Raghav (2006) (n=40) and Morgan (2010) (n=98) specifically examined the effect of B. monnieri on adults (> 55 yoa) with age related memory impairment in the absence of diagnosed dementia. Both trials showed statistically significant improvements in the trial groups. Morgan (2010) assessed for audio-verbal and visual memory performance using the AVLT, Rey-Osterrith Complex Figure Test (CFT) and Reitan Trail Making Test (TMT). Subjective memory performance was also assessed. The team found that B. monnieri was able to produce statistically significant improvements in memory acquisition and retention as per AVLT assessment. Raghav and colleagues used the Wechsler memory scale and its subsets and reported improvements in logical memory, mental control and paired associated learning (Raghav 2006).
All but one RCT concluded that B. monnieri produced cognitive enhancement in adults with or without adult age related memory impairment. The trials by Calabrese 2008, Stough 2001, Morgan 2010 and Roodenrys 2002 all used similar dosing of B. monnieri; 300 mg qd for a 12 week duration. All four trials concluded that B. monnieri enhanced cognition in adults; however the specific effects reported on cognition differed between trials (See Table 1).
The study by Stough and colleagues showed improved information processing, learning rate and memory consolidation (2001). Roodenrys 2002 which employed a slight variation in dosing depending on the subject weight (300 mg qd for subjects < 90kg and 405 mg qd for subjects >90 kg) reported improvements in retention of new information; however no improvements were noted in attention, verbal and visual short term memory, or retrieval of pre-existing knowledge or anxiety, which had previously been reported to be improved in the studies conducted by Calabrese 2008 and Stough 2002 (Roodenrys 2002). Morgan 2010 also reported significant improvements in memory acquisition whereas Roodenrys 2002 suggests it is the rate of forgetting that was decreased by B. monnieri (Morgan 2010).
From this is becomes apparent that there is a need for further clinical trials which use the same neuropsychological assessments in order to ascertain the specific effect B. monnieri has on memory – whether it increases fact retention or decreases the rate of forgetting.
The trial that did not show any statistically significant effect of B. monnieri on neuropsychological parameters was investigating the acute (2 hours) effects of the herb on cognitive parameters (Nathan 2001).
Supplementation with B. monnieri may have cognitive enhancing effects even after its intake is discontinued (Rhagav 2006). In one double blind RCT the trial groups were randomized to either B. monnieri or placebo for 12 weeks, after this period the B. monnieri group was also given placebo for a subsequent four weeks; where as the placebo group consumed placebo for the entire 16 week trial (Raghav 2006). Neuropsychological assessments were significantly improved in the B. monnieri group at the end of the trial and no decrease in memory performance scores were noted between week 12 and week 16 time points (Raghav 2006). This study demonstrates that outcomes delivered through B monnieri supplementation persist for at least four weeks following discontinuation of the intervention.
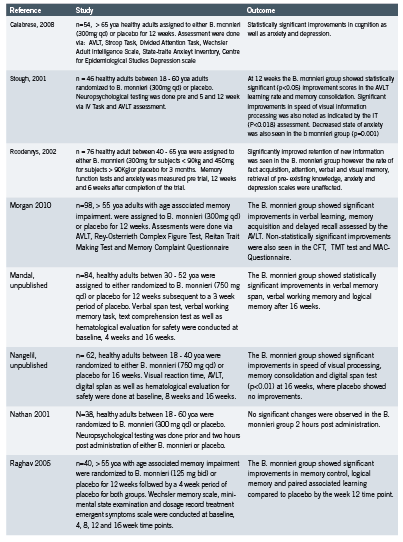
Safety & Dosing:
Dosing varied between the trials (from 250 mg qd to 750 mg qd), with the majority prescribing 300 mg qd of standardized B. monnieri extract either in one single dose or in divided doses. The extracts were standardized to bacoside content, which varied between 40 – 55% (Calabrese 2008, Morgan 2010, Nathan 2001, Raghav 2006, Stough 2001). Two studies used 750 mg qd of which 100 mg was a whole plant extract and the remaining 650 mg was powdered plant (Mandal, Nangelil). In Roodenrys (2002) dosing was done by weight in which persons under 90kg where given 300 mg and those over 90kg were given 450 mg of B. monnieri, which was equated to 6 and 9g of dried B. monnieri rhizome, respectively.
A 2007 toxicological in vivo study in Sprague-Dawley rats reported a median oral lethal dose of 2400mg/kg of B. monnieri extract (Joshua- Allan 2007). Repeated daily oral dosing for 14 days was well tolerated in rats at up to 500mg/kg; furthermore, a sub-chronic study for 90 days with doses up to 500 mg/kg did not result in toxicities in these animals (Joshua- Allan2007).
A phase I safety study on a B. monniere extract has been conducted in 23 healthy human subjects who were given 300 mg of the extract with a dose escalation up to 450 mg qd for a total of 30 days (Morgan 2010, Pravina 2007). Three of the 23 subjects reported gastrointestinal adverse events; the trial concluded that B. monnieri was safe and did not produce either clinical nor laboratory evidence indicating that it was unsafe at 300 mg qd (Pravina 2007).
B. monnieri has a long historical use which speaks to its safety profile; that being said, a small number of adverse events were noted in four of the studies, they included gastrointestinal effects (increased stool frequency, abdominal cramping and nausea) (Calabrese 2008, Morgan, 2010, Pravina 2007, Roodnenrys 2002) fatigue and dry mouth (Stough 2001). It has been hypothesized that the gastrointestinal adverse events may be due to the cholinergic effects of B. monnieri (Morgan 2010).
Conclusion:
At present, clinical as well as laboratory research has shown that B. monnieri appears to have cognitive enhancing effects; however the specific mechanisms of action are not well understood and from the clinical trials completed there appears to be a discrepancy as to how it may impact memory specifically.
There is a large number of limitations to the current research including: small trial sample size, large variation in assessment methods between trials, lack of long term data, inconsistency in dosing and standardization of B. monnieri extract. Thus far the research is encouraging and combined with the historical use of B. monnieri as a cognitive enhancement agent in Ayurvedic medicine, this should provide a basis for further research for this promising agent.
References:
Bhattacharya S.K., Bhattacharya A., Kumar A., Ghosal S. Antioxidant activity of Bacopa monniera in rat frontal cortex striatum and hippocampus. Phytother Res. 2000; 14(3): 174 – 179.
Calabrese C., Gregory W., Leo M., Kraemer D., Bone K., Oken B. Effect of standardized Bacopa monnieri extract on cognitive performance, anxiety, depression in the elderly: randomized, double-blind, placebo-controlled trial. The Journal of Alternative and Complementary Medicine. 2008; 14(6): 707-713.
Chowdhuri DK., Parmar D., Kakkar P., Shukla R., Seth P.K., Srimal R.C. Antistress effecs of Bacosides of Bacopa monnieri: Modulation of Hsp70 expression, superoxide dismutase and cytochrome p450 activity in rat brain. Phytother. Res. 2002; 16: 639-645.
Jorm AF, Rodgers B, Christensen H. Use of medications to enhance memory in a large community sample of 60-64 year olds. Int Psychogeriatr. 2004;16:209–217.
Joshua- Allan J., Damodaran A., Deshmukh N.S., Goudar K.S., Amit A., Safety evaluation of a standardized phytochemical composition extract from Bacopa monnieri in Sprague-Dawley rats. Food and Chemical Toxicology. 2007; 45: 1928-1937.
Limpeanchob N., Jaipan S., Rattanakaruna S., Phrompittayarat W., Inkaninan K., Neuroprotective effect of Bacopa monnieri on beta-amyloid-induced cell death cortical culture. Journal of Ethnopharmacology. 2008; 120: 112 – 117
Mandal A. A clinical study to evaluate the efficacy and safety of Bacopa caplets in memory and learning ability: A double blind placebo controlled study. The Himalayan Drug Company. [Received via e-mail on 29 January 2010]. Unpublished.
Morgan A., Stevens J. Does Bacopa monnieri improve memory performance in older persons? Results of a randomized, placebo controlled, double blind trial. The Journal of Alternative and Complementary Medicine. 2010; 16 (7): 753-759
Nangelil V. Effect of Bacopa caplet on human memory: a double blind placebo controlled study. The Hialayan Drug Company. [ Received via e-mail on 29 January 2010]. Unpublished.
Nathan P.J., Clarke J., Lloyde J., Hutchison C.W., Downey L., Stough C. The acute effects of an extract of Bacopa monniera (Brahmi) on cognitive function in healthy normal subjects. Hum Psychopharmacol. 2001; 16: 345 – 351.
Prabhakar S., Saraf M.K., Pandhi P., Anand A., Bacopa monniera exerts antiamnesic effec on diazepam induced anterograde amnesia in mice. Psychopharmacology. 2008; 200: 27-37.
Pravina K., Ravindra K.R., Goudar D.R., Vind A.J., Joshua P., Wasim K., Venkatehwarlu V.S., Saxena A.A. Safety evaluation of BacoMind in healthy volunteers: A phase I study. Phytomedicine. 2007; 14:301-308.
Raghav S., Singh H., Dalai P.K., Srivastava J.S., Asthana O.P. Randomized controlled trial of standardized Bacopa monniera extract in age- associated memory impairment. Indian J Psychiatry. 2006; 48(4): 238-242
Roodenrys S., Booth D., Bulzomi S., Phipps A., Micallef C., Smoker J. Chronic effects of Brahmi (Bacopa monnieri) on human memory. Neuropsychopharmacology. 2002; 27: 279 – 281.
Statistics Canada – Demographic Change, 2010 [ Accessed online 20 December 2010 at [http://www.statscan.gc.ca/pub/82-229-x/2009001/demo/int1-eng.htm]
Stough C., Lloyd J., Clarke J., Downey L.A., Hutchison C.W., Rodgers T., Nathan P.J. The chronic effects of an extract of Bacopa monniera (Brahmi) on cognitive function in healthy human subjects. Psychopharmacology. 2001; 156:481-484.
Uabundit N., Wattanathorn J., Supaporn M., Kornkanok I. Cognitive enhancement and neuroprotective effects of Bacopa monnieri in Alzheimer’s disease model. Journal of Ethnopharmacology. 2010; 127: 26-31.
Usha P.D., Wasim P., Joshua J.A., Geetharani P., Murali B., Mayachari A.S., Venkateshwarlu K., Saxena V.S., Deepak M., Amit A. BacoMind: A cognitive enhancer in children requiring individual education programme. Journal of Pharmacology and Toxicology. 2008; 3(4): 302-310.