Excessive myocardial triglyceride (MTG) content in obesity and type 2 diabetes is associated with impaired cardiac function. This study was conducted to assess influences of moderate dietary weight loss in non-diabetic obese and overweight women on MTG content and cardiac function. A subgroup of 38 women was selected from the B-SMART study population that compared weight loss and associated metabolic and cardiovascular markers with reduced-carbohydrate and reducedfat hypocaloric diets. An average weight reduction of 5.4±4.3kg at six months was associated with a relative decrease of MTG of 25% and the response was similar with carbohydrate and fat restriction. Diastolic function expressed as ratio of peak filling rate in E- and A-Phase (PFR(E)/PFR(A)) was unchanged. Reductions of left atrial size (P = 0.002), the normalized ratio of PFR(E) and early diastolic lengthening velocity PLV (P < 0.001), and fat free mass (P = 0.007) reflected altered cardiac volume loading after diet, but did not correlate to MTG content. Therefore, moderate dietary weight loss reduced MTG content in women with uncomplicated overweight or obesity but dietary macronutrient composition did not affect the extent of MTG reduction. Int J Cardiol. 2012 Apr 9. PMID: 22494864
CoQ10 and Heart Failure
CoQ10 and Heart Failure
A review of evidence
Heart failure is a significant contributor to mortality and morbidity in the U.S. and the developed world. It is typical for heart failure patients to have low levels of coenzyme Q10, which is an integral cofactor for the mitochondrial respiratory chain involved in generating adenosine triphosphate, the major cellular energy source. Coenzyme Q10 is also a potent antioxidant and membranes stabilizer. Since its discovery in 1957, on the basis of a number of studies it has been used therapeutically for heart failure, but this work is largely ignored. Lack of bioavailability is also an issue, but this has been addressed and highly bioavailable products developed. This research is also ignored. It is well known that statin therapy dramatically reduces coenzyme Q10 levels and before the first statin was introduced, inclusion of this coenzyme was considered and rejected. Since coenzyme Q10 is not a prescription drug, most mainstream medical practitioners do not recognize it as a useful supplement or therapeutic agent. Nevertheless, in the context of heart failure, there is evidence that if the blood levels of this cofactor are considerably elevated through supplementation with highly bioavailable products, the impact on the grade of heart failure and the quality of life is highly significant. Beneficial results have been observed when this cofactor is used to treat the side effects of statin treatment and in particular myopathy. The association between dosing levels and outcomes is discussed. There are integrative cardiologists who claim that based on years of clinical experience, they could not effectively practice without this supplement.
Coenzyme Q10 (Q10), also known as ubiquinone, was discovered in 1957 and there followed a considerable amount of research with animals, humans and cell cultures. Many international symposia were held with hard cover proceedings that gather dust on library shelves. Q10 is found in all human cells and is a potent antioxidant, cell membrane stabilizer, and an essential enzyme in the mitochondrial respiratory chain where it is involved in the generation of ATP(Greenberg 1990, Sinatra 2005) Low Q10 has been associated with a number of disorders and its therapeutic use recently reviewed (Villalba 2010). Low levels of Q10 play a role in heart failure, angina and hypertension (Tran 2001). This is important since heart failure contributes significantly to mortality and morbidity in the U.S. and other developed countries. Depending on symptom severity, heart dysfunction, and other factors, in Canada heart failure can be associated with an annual mortality of between 5% and 50% (Arnold 2006). The importance of Q10 for heart function is illustrated by five clinical trials cited by in a recent review with significant improvements found in endpoints of ejection fraction, pulmonary artery pressure, stroke volume, cardiac output, and functional capacity and quality of life associated with supplementation (Lee 2011).
Food is only a minor source of this cofactor. The pathway that leads to the endogenous synthesis of Q10 is also the pathway to cholesterol. Drugs called HMG-CoA reductase inhibitors, better known as statins, inhibit this pathway with the resultant large decrease in both cholesterol and Q10. Concern over the impact of the widespread use of statins in this context is mounting as the pressure intensifies to have everyone on statins, from toddlers to the frail elderly.
Q10 is not viewed with enthusiasm by mainstream medicine, although a systematic review published in 2003 (Rosenfeldt 2003) noted non-significant trends toward increased ejection fraction and reduced mortality associated with Q10 therapy and another study published in 2006 found Q10 enhanced systolic function with HF (Sander 2006). It is not a prescription drug and in the context of heart failure, recent studies can be cited which suggest it is ineffective. As will be discussed below, this conclusion is contrary to a considerable body of older literature and the experience of integrative cardiologists (Sinatra 2005, Sinatra 2009a, Sinatra 2009b). Instead, several anti-hypertension drugs, digoxin and aldosterone receptor antagonists are the standard of practice along with devices that help the heart beat and contract properly. Heart transplants and end-of-life care are discussed in guidelines. Mainstream medicine would no doubt point to the following two recent studies as evidence that Q10 is not an important issue in heart failure.
In 2011 a study was published (Fumagalli 2011) which reported on a small randomized placebo controlled trial involving 67 patients with heart failure (HF) randomized to receive for eight weeks either a placebo plus the usual care or a combination of a Q10 preparation, viewed as of enhanced bioavailability, and creatine plus the usual care. The Q10 dose was 34 mg/day, a rather low dose. Outcomes were exercise tolerance, peak oxygen consumption from an exercise test and what was called a sickness impact profile. Small improvements were found, mostly of no statistical significance or of small and questionable clinical significance. Q10 blood levels were not reported in spite of the novel nature of the Q10 source.
In 2010 a sub-study of the CORONA study examined the impact of rosuvastatin (Crestor) and Q10 levels on heart failure (McMurray 2010). This was an industry-sponsored study with the majority of the investigators having close financial ties to the sponsor. The average age of subjects was about 72. Serum Q10 was measured but the only intervention was with rosuvastatin. All the subjects had significant to severe heart failure. It was observed that patients with lower Q10 levels at baseline were older and had more advanced heart failure (HF). The statin reduced the mean Q10 levels in each of three tertiles of Q10 from 0.49 to 0.35, 0.75 to 0.46 and 1.10 to 0.53 μg/ml respectively. Mortality was significantly higher among patients in the lowest vs. highest Q10 tertile, but the difference was not significant on multivariate analysis and Q10 was not found upon extensive statistical manipulation to be an independent predictor of either worsening or fatal HF, nor did statin treatment result in worse outcomes. The message: concern over statins, Q10 and HF is not justified.
The Q10 levels need to be put in perspective. The range of Q10 serum levels in self-reported normal, healthy individuals is quite large. In one study of healthy individuals, the distribution of blood Q10 levels was found to be <0.4 μg/L, 2%; 0.4-1.6 μg/mL, 81%; and > 1.6 μg/mL 17% (Lu 2007). The age range was infant to 94 years. There was no gender dependence.
A study published in 2008 does not support the view of Q10 as unimportant. This study (Molyneux 2008) examined the relationship between Q10 blood levels and survival among patients with chronic heart failure (CHF). Two hundred thirty six patients, mean age 77, admitted to hospital with HF were followed for a mean of 2.7 years. The mean Q10 blood level was 0.58 μg/mL. They found a significant difference in survival over the period studied (about 4 years) when a cut-point of 0.63 μg/ mL was used (survival of about 65% vs. 45%). Patients below this cut point had a range of Q10 blood levels of 0.11 to 0.63 whereas those above had a range of 0.63 to 1.50 μg/mL.
Important perspective concerning Q10 and HF can be gained by considering the views of Dr. Peter H. Langsjoen, a cardiologist who has been involved on Q10 research since 1985 and has published extensively in this area. He was recently interviewed and the transcript is available on the internet (Langsjoen 2011). He points out that early on, it was believed that if HF patients typically had Q10 levels around 0.5 μg/mL and normal individuals had levels of around 1μg/mL, then when one was trying to treat HF, the use of supplementation to bring the value up to about 1μg/mL was indicated. When this was tried, not much improvement was seen. Further research revealed that there was a significant blood level threshold at about 2.5 μg/ml above which HF patients appeared to have some benefit and severe HF patients were helped by supplementation once the level achieved was greater than 3.5 μg/mL. This was pointed out in a 2008 paper in Biofactors (Langsjoen 2008). Even in an earlier paper (Langsjoen 1999) in the same journal the threshold of > 3.5 μg/mL blood level was discussed and justified. The results of years of research are clearly being ignored.
In the study by Fumagalli et al, Q10 levels were not measured and the dose was very low. It was thus not surprising that small or non-significant results were obtained. The authors were aware of the 1999 paper which recommended and justified therapeutic levels > 3.5 μg/mL for HF patients. But in this study with a novel source of Q10, the levels achieved were not reported and were probably too low. In the study by McMurry et al the range of Q10 serum levels was too low to be of significance in the context of serious HF, especially in multivariate analysis looking for an independent effect and the lowering of Q10 caused by the statin was probably too small to be of significance in this cohort of patients, many with a need for very high levels.
The most interesting study and one which should not have been ignored when the above studies were designed appears to be that of Langsjoen and Langsjoen (Langsjoen 2008). They report on seven consecutive patients who had worsening HF (NYHA Class IV) who were on maximal medical therapy and taking large doses of the ubiquinone form of Q10 which was not, from their point of view concerning the importance of the >3.5 μg/ mL threshold, adequately elevating blood levels in the context of severe HF. Some patients were taking 900 mg/day and still well below the threshold. This could have been written up and presented as a negative study demonstrating that without a doubt Q10 supplementation did not work at all for severe HF. Not so. Patients were switched from an average dose of ubiquinone of 450 mg/day to the ubiquinol form at an average dose of 580 mg/day with a change in average blood Q10 from 1.6 to 6.5 μg/mL. The table below provides the detailed results of ejection fraction (EF) and NYHA class change indicating the decline in the severity of HF after ubiquinol was used rather then ubiquinone. Ubiquinol is the reduced form and the most prevalent form in humans.
The HF class changes listed above are impressive and obviously of huge significance to most of the patients involved. Note the high baseline Q10 levels, which are at the upper extreme of the modern laboratory reference range, and yet the individuals had Class IV HF which was then strongly impacted by changing the supplement to achieve greater bioavailability and thus achieving much higher Q10 levels. Note also the individual variations. This table in fact nicely states the case for treating HF patients to a high target even if their Q10 levels are already high by traditional standards. Four out of seven patients regressed to NYHA I or II. These results suggest the urgent need for a much larger study, but if it has been done it does not appear to have been published.
Even before Langsjoen and Langsjoen carried out their case study, other studies confirmed that while ubiquinone bioavailability was increased when administered in a capsule where the chemical was dispersed in an oil, much higher levels of blood Q10 could be achieved by replacing the oxidized form with ubiquinol (Bhagavan 2007, Hosoe 2007). In fact, Hosoe et al found that supplementation with 300 mg ubiquinol in oil over a period of 28 days raised the Q10 level on average from 0.66 to 7.28 μg/mL. This level was much higher than that achieved with a single dose (2.56 μg/ mL) and thus there is a cumulative effect which must be taken into account in evaluating single dose studies. They found these higher serum levels safe, consistent with a recent safety study (Hidaka 2008). They used Kaneak QH, a Japanese ubiquinol preparation which is available today over the internet from several supplement suppliers. Another comparable preparation is LiQ-NOL CoQ10 which also uses the ubiquinol from Kaneka.
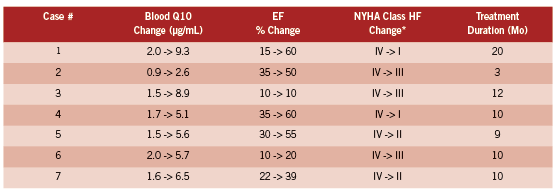
significant limitation of activity due to Symptoms, even to the point of problems walking short distances. Class IV is sufficiently severe as to apply mostly to bed ridden patients.
It has been reported that serious side effects accompany statin use, and that these can be treated and reduced by Q10 supplementation. For example, a small randomized controlled trial reported in 2007 (Caso 2007) was conducted with patients suffering from myopathic symptoms thought to be associated with statin treatment. Supplementation with Q10 (100 mg/day soft-gel) while continuing statin treatment resulted after 30 days in 40% reduction in pain and 38% pain associated interference with daily activities, whereas there were no significant changes in these endpoints in the control group. Similar results were reported in 2005 for patients treated with Q10 after discontinuing statin therapy because of a variety of side effects attributed to this class of drug (Langsjoen 2005).
A more comprehensive alternative approach to heart failure and preventing and treating heart disease in general involves not only Q10 but L-Carnatine and D-Ribose, an approach called Metabolic Cardiology, developed and promoted by the cardiologist Stephen Sinatra on the basis of extensive anecdotal clinical evidence (Sinatra 2005, Sinatra 2009a, Sinatra 2009b). In this context, other micronutrients studied which also appear to provide benefit include omega-3 fatty acids, the B-family of vitamins, and vitamin D (Lee 2011).
A reasonable conclusion appears to be that HF patients are significantly helped by Q10 supplementation, but the dose must be individualized and blood levels measured to test the impact of the intervention. One can not generalize on the oral dose because of the wide variation in both commercial preparations and individual absorption. Thus monitoring and treating to a target serum levels is essential. As pointed out by Langsjoen in the interview cited above, it took some time before it was demonstrated that using doses of 100 mg/day of ubiquinone was not very effective in many cases of HF and early studies found in many cases only small effects. In his interview, Langsjoen is emphatic that there are no side effects or drug interactions to high doses of Q10. He does however point out that Q10 therapy for HF, once heart function improves, reduces the need for some of the “standard treatment” drugs and in particular blood pressure medications. In fact, Q10 had been used to treat hypertension (Wyman 2010).
REFERENCES
Arnold,P., Liu,P., Demers,C., Dorian,P. and Giannetti,N. Canadian Cardiovascular Society consensus conference recommendations on heart failure 2006. Can J Cardiol 2006; 22(1): 23-45.
Bhagavan,H.N. and Chopra,R.K. Plasma coenzyme Q10 response to oral ingestion of coenzyme Q10 formulations. Mitochondrion 2007; 7 Suppl: S78-S88.
Caso,G., Kelly,P., McNurlan,M.A. and Lawson,W.E. Effect of coenzyme q10 on myopathic symptoms in patients treated with statins. Am J Cardiol 2007; 99(10): 1409-1412.
Fumagalli,S., Fattirolli,F., Guarducci,L., Cellai,T., Baldasseroni,S., Tarantini,F., Di,B.M., Masotti,G. and Marchionni,N. Coenzyme Q10 terclatrate and creatine in chronic heart failure: a randomized, placebo-controlled, double-blind study. Clin Cardiol 2011; 34(4): 211-217.
Greenberg,S. and Frishman,W.H. Co-enzyme Q10: a new drug for cardiovascular disease. J Clin Pharmacol 1990; 30(7): 596-608.
Hidaka,T., Fujii,K., Funahashi,I., Fukutomi,N. and Hosoe,K. Safety assessment of coenzyme Q10 (CoQ10). Biofactors 2008; 32(1-4): 199-208.
Hosoe,K., Kitano,M., Kishida,H., Kubo,H., Fujii,K. and Kitahara,M. Study on safety and bioavailability of ubiquinol (Kaneka QH) after single and 4-week multiple oral administration to healthy volunteers. Regul Toxicol Pharmacol 2007; 47(1): 19-28.
Langsjoen,P. Congestive heart failure and the clinical uses of coenzyme Q10. Interview by K. R. Hamilton. http://www. prescription2000. com/images/stories/ transcripts/2011-09-08-peter-langsjoen-chf-coenzyme-q10-transcript. pdf 2011.
Langsjoen,P.H. and Langsjoen,A.M. Supplemental ubiquinol in patients with advanced congestive heart failure. Biofactors 2008; 32(1-4): 119-128.
Langsjoen,P.H., Langsjoen,J.O., Langsjoen,A.M. and Lucas,L.A. Treatment of statin adverse effects with supplemental Coenzyme Q10 and statin drug discontinuation. Biofactors 2005; 25(1-4): 147-152.
Langsjoen,P.H. and Langsjoen,A.M. Overview of the use of CoQ10 in cardiovascular disease. Biofactors 1999; 9(2-4): 273.
Lee,J.H., Jarreau,T., Prasad,A., Lavie,C., O’Keefe,J. and Ventura,H. Nutritional assessment in heart failure patients. Congest. Heart Fail. 2011; 17(4): 199-203.
Lu,J. and Frank,E.L. Measurement of coenzyme Q10 in clinical practice. Clin Chim. Acta 2007; 384(1-2): 180-181.
McMurray,J.J., Dunselman,P., Wedel,H., Cleland,J.G., Lindberg,M., Hjalmarson,A., Kjekshus,J., Waagstein,F., Apetrei,E., Barrios,V., Bohm,M., Kamensky,G., Komajda,M., Mareev,V. and Wikstrand,J. Coenzyme Q10, rosuvastatin, and clinical outcomes in heart failure: a pre-specified substudy of CORONA (controlled rosuvastatin multinational study in heart failure). J Am Coll Cardiol 2010; 56(15): 1196-1204.
Molyneux,S.L., Florkowski,C.M., George,P.M., Pilbrow,A.P., Frampton,C.M., Lever,M. and Richards,A.M. Coenzyme Q10: an independent predictor of mortality in chronic heart failure. J Am Coll Cardiol 2008; 52(18): 1435-1441.
Rosenfeldt,F., Hilton,D., Pepe,S. and Krum,H. Systematic review of effect of coenzyme Q10 in physical exercise, hypertension and heart failure. Biofactors 2003; 18(1-4): 91-100.
Sander,S., Coleman,C.I., Patel,A.A., Kluger,J. and White,C.M. The impact of coenzyme Q10 on systolic function in patients with chronic heart failure. J Card Fail. 2006; 12(6): 464-472.
Sinatra,S.T., 2005. The Sinatra solution. Basic Health Publications, North Bergen, NJ.
Sinatra,S.T. Metabolic cardiology: an integrative strategy in the treatment of congestive heart failure. Altern. Ther Health Med 2009a; 15(3): 44-52.
Sinatra,S.T. Metabolic cardiology: the missing link in cardiovascular disease. Altern. Ther Health Med 2009b; 15(2): 48-50.
Tran,M.T., Mitchell,T.M., Kennedy,D.T. and Giles,J.T. Role of coenzyme Q10 in chronic heart failure, angina, and hypertension. Pharmacotherapy 2001; 21(7): 797-806.
Villalba,J.M., Parrado,C., Santos-Gonzalez,M. and Alcain,F.J. Therapeutic use of coenzyme Q10 and coenzyme Q10-related compounds and formulations. Expert Opin Investig. Drugs 2010; 19(4): 535-554.
Wyman,M., Leonard,M. and Morledge,T. Coenzyme Q10: a therapy for hypertension and statin-induced myalgia? Cleve. Clin J Med 2010; 77(7): 435-442.
Cancer induced cachexia (CIC)
Cancer induced cachexia (CIC)
Effective integrative management
Abstract
Cancer induced cachexia (CIC) is a syndrome of muscle wasting accompanied by anorexia with or without loss of adipose tissue. Underlying CIC is a complex interaction of proinflammatory cytokines that act centrally on the hypothalamus and peripherally to increase catabolism and resting energy expenditure, while decreasing protein synthesis. The presence of CIC is an important prognostic indicator in cancer patients. Nutritional strategies utilized by naturopathic doctors have been proven to slow down this condition, induce weight gain, and improve cancer related outcomes. Such therapies include high EPA fish oil, melatonin, L-carnitine, and branched chain amino acids and/ or whey protein.
Cancer induced cachexia (CIC) is a multifactorial syndrome characterized by loss of skeletal muscle, accompanied by anorexia and (sometimes) loss of adipose tissue (Fearon 2011). A number of underlying metabolic derangements are implicated in CIC, and associated symptoms may include malnutrition, anemia, fatigue, and decreased muscle strength & impaired physical function. Cachexia, defined as a loss of body weight greater than 5%, leads to poor performance, poor quality of life, increased complications, and higher mortality rate among cancer patients (Burckart 2010, Fearon 2011). Targeted nutritional interventions are an important part of cachexia prevention and management. This paper reviews several key agents that can improve outcomes in cachexia, among them fish derived omega-3 fatty acids (EPA and DHA), melatonin, branched chain amino acids (BCAAs), l-carnitine, and whey protein.
Cachexia is a therapeutic target of high importance in cancer patients due to its strong prognostic significance (Burckhart 2010). It is widely recognized that cachexia leads to poorer cancer outcomes, and is reported as the direct cause of death in up to 20-40% of cancer patients (Fox 2009). Recently, Yang et al found that, among a large cohort of lung cancer patients (n=14,751), those who showed weight loss (15.8% average) at time of diagnosis had significantly shorter survival time compared to those who did not, 6.4 versus 9.2 mo, P < 0.001 (2011). Similar results have been found for other cancers as well, including colorectal cancer and pancreatic cancer, with up to two fold greater increase in risk of death (HR = 2.26; CI 1.18-4.32; P = 0.014) reported (Bachmann 2008, Thoreson 2012). Some teams have used serum albumin as a marker of malnutrition and cachexia, and low levels have been associated with decreased survival in ovarian, breast, and other cancers (Asher 2011, Lis 2003, Polterauer 2010). Cancer related weight loss is associated with decreased tolerance to anticancer therapy, and significantly predicts toxicity from cancer treatment (Fearon 2011, Ross 2004). Naito found that cachexia is associated with altered oxycodone pharmacokinetics due to decreased albumin levels (an important drug binding and transporting protein), resulting in increased levels of free drug and increased incidence of central adverse reactions (2012). Despite this, cachexia remains under-recognized in patients, and under-treated as a clinical entity (Churm 2009, Spiro 2006).
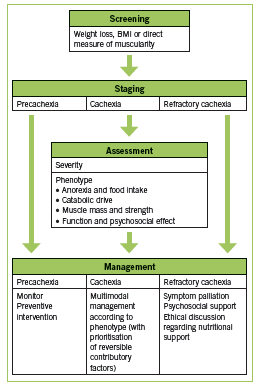
The molecular mechanisms underlying CIC are still being elucidated, however, pro-inflammatory cytokines such as IL-1, IL-2, IL-6, interferon gamma and TNF α have a key role in mediating cancer cachexia (Macdonald 2003). IL-6 is a key cytokine in iniating multiple proinflammatory pathways including the acute phase response, and is known to be produced by the tumor microenvironment (Oshima 2012, Zamarron 2011). The inflammatory basis of CIC distinguishes it from age related sarcopenia or frank starvation (Pepersack 2011), and presents the therapeutic rationale for intervention with agents such as EPA, NSAIDS, and celecoxib, a COX-2 inhibitor. Ultimately, these pro-inflammatory cytokines initiate a cascade of events in the hypothalamus and peripherally that results in detrimental metabolic changes, including increased catabolism (proteolysis and lipolysis) and resting energy expenditure, reduced muscle protein synthesis, and anorexia (Burckart 2010). According to a more simple summation by Fearon, “The pathophysiology [of cachexia] is characterized by a negative protein and energy balance driven by a variable combination of reduced food intake and abnormal metabolism” (2011).
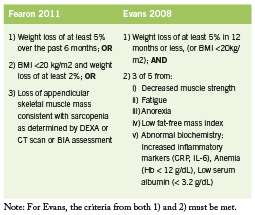
Various definitions of CIC have been used. Two recent sets of diagnostic criteria are outlined in Table 1. These criteria by Fearon are recently proposed in a consensus statement published in Lancet Oncology (2011), while those by Evans are older and incorporate biochemical parameters (2008). Recently, work has been done to establish staging of cachexia. Precachexia is defined as “weight loss ≤5%, with anorexia and metabolic changes” (Fearon 2011). Cachexia is as defined below (Fearon 2011). Refractory cachexia is variable in terms of severity, with the presence of procatabolic state; cancer not responsive to anticancer treatment; low performance score; and <3 months expected survival (Fearon 2011). A proposed algorithm in terms of assessment and management is adapted in Figure 1. (Valium)
Nutritional Interventions Fish Derived Omega-3 Fatty Acids
Fish oil is rich in the omega-3 polyunsaturated fatty acids eicosapentanoic acid (EPA) and docosahexanoic acid (DHA), and has been shown to suppress the production of proinflammatory cytokines in both healthy volunteers and cancer patients, leading to weight stabilization, improved immune function, and better cancer treatment outcomes (Barber 2001, Wigmore 2000). Evidence on use of EPA in various cancer types is summarized in Table 2.
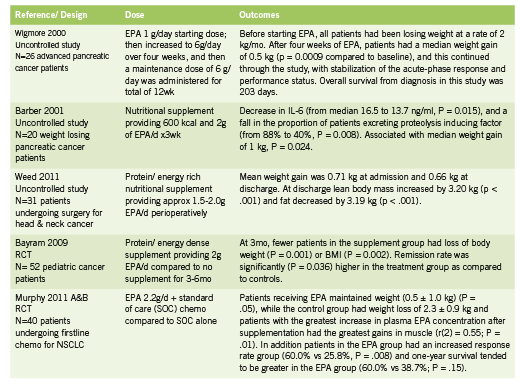
A 2007 systematic review found that oral supplements enriched with omega-3 fatty acids increased weight gain, increased appetite, improved quality of life and reduced post-surgical morbidity in patients with advanced pancreatic and upper gastrointestinal cancer (Colomer 2007). Not all trials have shown such promising effects (Fearon 2006), however, though the reason may have to do with cancer stage, study duration, and differences between cancer types and phenotypes. Although the dosages used in clinical trials shows some variability, a dose of at least 2g EPA per day is recommended. No serious side effects have been reported in the trials reviewed; minor side effects typically include fishy aftertaste and mild nausea or GI symptoms.
Melatonin
Melatonin (N-acetyl-5-methoxytryptamine) is a chronobiological hormone secreted by the pineal gland that is responsible for maintaining circadian rhythms. Melatonin acts as a potent antioxidant, immune modulating agent, antitumor, and antitoxic agent with respect to reducing radiation and chemotherapy induced toxicity (Seely 2011, Vijayalaxmi 2002). A recent meta analysis by Seely et al including 21 RCTs showed that melatonin alongside chemo and/ or radiation therapy improves survival (reduction in one year mortality, RR = 0.60; 95% CI = 0.54-0.67) and decreases toxicities of treatment, including asthenia, leucopenia, nausea and vomiting, hypotension, and thrombocytopenia (2011). Preliminary trials have shown an effect of melatonin on cachexia.
In a randomized controlled trial, 24 patients with advanced gastrointestinal cancer were randomized to receive fish oil or melatonin (Persson 2005). For the fish oil group, 13 patients received the equivalent of 4.9 g EPA and 3.2 g DHA/d, while the melatonin group (n=11) were given 18 mg melatonin/d. After four weeks, five of the 13 patients (38%) in the fish oil group and 3 of 11 patients (27%) in the melatonin group showed weight stabilization or gain. Notably, after combining both interventions, 63% of individuals responded, suggesting an additive effect from the combination.
In another study, Lissoni et al found that among 100 patients with untreatable metastatic solid tumours, those receiving supportive care plus melatonin (20 mg/day orally in the evening x2mo) versus supportive care alone showed slower weight loss and decrease in TNF levels: “The per cent of weight loss greater than 10% was significantly higher in patients treated by supportive care alone than in those concomitantly treated by MLT, with no difference in food intake (P < 0.01)” (1996).
Branched Chain Amino Acids
The branched chain amino acids consist of leucine, isoleucine, and valine, and these may act indirectly by modulating serotonin activity in the hypothalamus. In the brain, the synthesis of neurotransmitters such as serotonin, dopamine, and norepinephrine are dependent upon the availability of the aromatic amino acids (tryptophan, phenylalanine, and tyrosine) respectively (Fernstrom 2005). Branched chain amino acids are thought to counteract the effects of anorexia by competing with tryptophan for entry through the blood brain barrier (Inui 2002). In cancer serotonin levels can be elevated as a result of increased plasma tryptophan. Through competition, BCAAs allow less serotonin in the brain, and result in a reduction in the amount of hypothalamic activity contributing to anorexia (Inui 2002). In addition BCAAs possess anabolic effects peripherally in skeletal muscle (Laviano 2005).
In a double blind prospective study, 28 cancer patients with anorexia were given an oral supplement consisting of branched chain amino acids 4.8 g three times daily for seven days (Le Bricon 1996). In the BCAA- treated group, the incidence of anorexia decreased from 100% to 45% by the end of the study. In the placebo group, the incidence of anorexia remained at 84%. The authors concluded that BCAA supplementation can safely be administered for the treatment of cancer-induced anorexia (Le Bricon 1996).
In a study of patients with intraabdominal adenocarcinoma and receiving total parenteral nutrition (TPN), the effects of a solution containing 19% BCAAs was compared to an isocaloric, isonitrogenous formula containing 50% BCAAs (Hunter 1989). In the high BCAA group, the following outcomes were observed in comparison to the low BCAA group: increase in whole body protein synthesis and breakdown (p<0.05); increased synthesis rate of albumin (P< 0.05); and reduction in tyrosine oxidation, suggesting improved protein utilization.
In terms of BCAA supplementation in increasing lean body mass, clinical trials have been inconsistent in elucidating its effects on skeletal muscle synthesis in humans (Choudry 2006). To date, trials have largely been small and variable and there is no clear positive or negative benefit to BCAA supplementation. The evidence regarding BCAA supplementation in severe catabolic patients is more encouraging. Studies regarding this particular patient population have used more objective outcome measures such as decreases in urinary nitrogen excretion to show benefits.
L-Carnitine
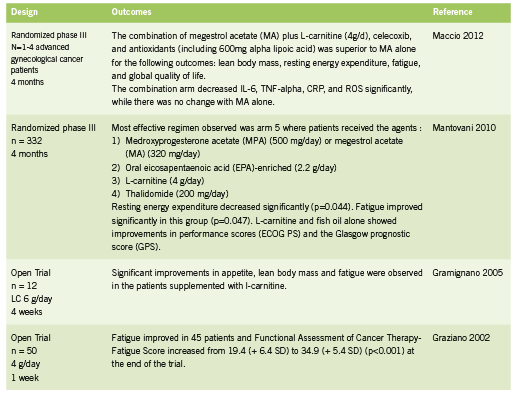
L-carnitine is an amino acid derivative and important cofactor in facilitating mitochondrial fatty acid metabolism within muscle tissue. L-carnitine is responsible for the transportation of fatty acids across the mitochondrial membrane in exchange for acetyl CoA, a byproduct of fatty acid oxidation (Fritz 2011). Maintaining this shuttle system 1. ensures substrate (fatty acids) for the production of ATP through beta oxidation in the mitochondria; and 2. prevents inhibition of glucose metabolism, which could happen through inhibition of pyruvate dehydrogenase by buildup of acetylCoA within the mitochondria (Fritz 2011). Some forms of chemotherapy have been shown to deplete carnitine, in part through impaired renal reabsorption (Hockenberry 2009, Mancinelli 2007). Open label studies of L-carnitine in cancer patients have shown improvements in lean body mass and fatigue (Gramignano 2005, Graziano 2002); RCTs of L-carnitine in combination with other agents in the treatment of cachexia have shown a superior combined effect on measure of cachexia, prognostic scores, and inflammatory cytokines (Maccio 2012, Mantanovi 2010).
Whey Protein Whey protein supplementation contributes to adequate protein intake, while supplying all the essential amino acids in order to maximize muscle protein synthetic activity and optimal immune function. Whey contains high concentrations of the BCAAs as well as beta-lactoglobulin, alpha-lactalbumin, bovine serum albumin, lactoferrin, immunoglobulins, lactoperoxidase enzymes, glycomacropeptides, lactose, and minerals (AltMedRev 2008). Human evidence shows that whey protein is rapidly absorbed and can effectively stimulate muscle protein synthesis (FSR) (Deutz 2011).
Use of a high protein, high leucine whey formula was investigated in 25 cancer patients prior or after chemotherapy treatment (Deutz 2011). The whey group (n = 13) received a formula containing 40g of protein based on casein, whey, and free leucine, while the control group (n =12) was given 24g of casein alone. Results showed that the fractional rate of muscle protein synthesis (FSR) significantly increased from 0.073 (SD: 0.023) to 0.097 (SD: 0.033) (p = 0.0269) in the whey group but not in the control group, showing the superior effects of a whey and leucine containing protein supplement.
A small trial examining the effect of whey on cancer progression was conducted among seven patients with metastatic cancer (five breast, one liver, one pancreas) (Kennedy 1995). Whey protein was supplemented at 30g daily for six months. In six patients, baseline blood lymphocyte GSH (glutathione) levels were substantially elevated, according to authors, “reflecting high tumour GSH levels” (Kennedy 1995). After treatment with whey, two patients exhibited “signs of tumour regression, normalization of haemoglobin and peripheral lymphocyte counts and a sustained drop of lymphocyte GSH levels towards normal” (Kennedy 1995). Two patients showed tumour stabilization and increased haemoglobin levels. Three patients had disease progression. The authors conlcuded that “whey protein concentrate might deplete tumour cells of GSH and [thereby] render them more vulnerable to chemotherapy” (Kennedy 1995). In addition to effects on tumor cell levels of GSH, whey protein has been shown to possess anticancer activity in animals by increasing GSH concentration in healthy tissues, thereby stimulating immune function through the GSH pathway (Bounous 2000). Glutathione is depleted in conditions of immune deficiency, for example HIV and cachexia; under these circumstances, whey protein acts as a cysteine donor to replenish GSH, since cysteine is the crucial limiting amino acid for intracellular GSH synthesis (Bounous 2000).
Conclusion
Naturopathic interventions used for treatment in cancer cachexia are based on good human level evidence and may offer important advantages to cancer patients, particularly in combination. These include high EPA fish oil, melatonin, branched chain amino acids, L-carnitine, and whey protein supplementation. These agents have a good safety profile in that little or no interactions with chemotherapy have been observed in human trials. While evidence suggests benefit from the application of each of these nutrients in isolation, a much stronger impact can reasonably be anticipated from their combined application, thus targeting multiple pathways and factors underlying the pathophysiology of cancer cachexia.
Coauthor Reshi Mehta would like to thank Dr Kieran Cooley, Dr Douglas Andrews, and Dr Philip Rouchotas for their guidance in the compilation of this manuscript.
References:
Altern Med Rev. Whey protein. Monograph. 2008 Dec;13(4):341-7.
Asher V, Lee J, Bali A. Preoperative serum albumin is an independent prognostic predictor of survival in ovarian cancer. Med Oncol. 2011 Jul 7.[Epub ahead of print]
Bachmann J, Heiligensetzer M, Krakowski-Roosen H, Büchler MW, Friess H, Martignoni ME. Cachexia worsens prognosis in patients with resectable pancreatic cancer. J Gastrointest Surg. 2008 Jul;12(7):1193-201.
Barber MD, Fearon KCH, Tisdale MJ, McMillan DC, Ross JA. Effect of a Fish Oil- Enriched Nutritional Supplement on Metabolic Mediators in Patients With Pancreatic Cancer Cachexia. NUTRTION AND CANCER 2001 40(2):118-124
Bayram I, Erbey F, Celik N, Nelson JL, Tanyeli A. The use of a protein and energy dense eicosapentaenoic acid containing supplement for malignancy-related weight loss in children. Pediatr Blood Cancer. 2009 May;52(5):571-4.
Bounous G. Whey protein concentrate (WPC) and glutathione modulation in cancer treatment. Anticancer Res. 2000 Nov-Dec;20(6C):4785-92.
Bruera E, Strasser F, Palmer JL, Willey J, Calder K, Amyotte G, Baracos V. Effect of Fish Oil on Appetite and Other Symptoms in Patients with Advanced Cancer and Anorexia/Cachexia: A Double-Blind, Placebo-Controlled Study. J Clin Oncol 2003 Jan;21(1):129-134
Burckart K, Beca S, Urban RJ, Sheffield-Moore M. Pathogenesis of muscle wasting in cancer cachexia: targeted anabolic and anticatabolic therapies. Curr Opin Clin Nutr Metab Care. 2010 Jul;13(4):410-6.
Choudry HA, Pan M, Karinch AM, Souba WW. Branched-chain amino acidenriched nutritional support in surgical and cancer patients. J Nutr. 2006 jan;136(1 Suppl):314S-8S
Churm D, Andrew IM, Holden K, Hildreth AJ, Hawkins C. A questionnaire study of the approach to the anorexia-cachexia syndrome in patients with cancer by staff in a district general hospital. Support Care Cancer. 2009 May;17(5):503-7.
Colomer R, Moreno-Nogueira JM, Garcia-Luna PP, Garcia-Peris P, Garcia-de- Lorenzo A, Zarazaga A, Quecedo L, del Llano J, Usán L, Casimiro C. N-3 fatty acids, cancer and cachexia: a systematic review of the literature. Br J Nutr. 2007 May;97(5):823-31
Deutz NE, Safar A, Schutzler S, Memelink R, Ferrando A, Spencer H, van Helvoort A, Wolfe RR. Muscle protein synthesis in cancer patients can be stimulated with a specially formulated medical food. Clin Nutr. 2011 Dec;30(6): 759-68
Donohoe C, Ryan AM, Reynolds JV. Cancer Cachexia: Mechanisms and Clinical Implications. Gastroenterol Res Pract. 2011 Jun;2011:1-13
Evans WJ, Morley JE, Argilés J, Bales C, Baracos V, Guttridge D, Jatoi A, Kalantar- Zadeh K, Lochs H, Mantovani G, Marks D, Mitch WE, Muscaritoli M, Najand A, Ponikowski P, Rossi Fanelli F, Schambelan M, Schols A, Schuster M, Thomas D, Wolfe R, Anker SD. Cachexia: a new definition. Clin Nutr. 2008 Dec;27(6):793-9.
Fearon K, Strasser F, Anker SD, Bosaeus I, Bruera E, Fainsinger RL, Jatoi A, Loprinzi C, MacDonald N, Mantovani G, Davis M, Muscaritoli M, Ottery F, Radbruch L, Ravasco P, Walsh D, Wilcock A, Kaasa S, Baracos VE. Definition and classification of cancer cachexia: an international consensus. Lancet Oncol. 2011 May;12(5):489-95.
Fearon KC, Barber MD, Moses AG, Ahmedzai SH, Taylor GS, Tisdale MJ, Murray GD. Double-blind, placebo-controlled, randomized study of eicosapentaenoic acid diester in patients with cancer cachexia. J Clin Oncol. 2006 Jul 20;24(21):3401-7.
Fernstrom JD. Branched-chain amino acids and brain function. J Nutr. 2005 Jun;135(6 Suppl):1539S-46S
Fox KM, Brooks JM, Gandra SR, Markus R, Chiou CF. Estimation of Cachexia among Cancer Patients Based on Four Definitions. J Oncol. 2009;2009:693458.
Fritz, H. L-carinitine: cardiovascular applications. IHP February/March 2011: 40-44.
Gramignano G, Lusso MR, Madeddu C, Massa E, Serpe R, Deiana L, Lamonica G, Dessi M, Spiga C, Astara G, Macciò A, Mantovani G. Efficacy of l-carnitine administration on fatigue, nutritional status, oxidative stress, and related quality of life in 12 advanced cancer patients undergoing anticancer therapy. Nutrition. 2006 Feb;22(2):136-45
Graziano F, Bisonni R, Catalano V, Silva R, Rovidati S, Mencarini E, Ferraro B, Canestrari F, Baldelli AM, De Gaetano A, Giordani P, Testa E, Lai V. Potential role of levocarnitine supplementation for the treatment of chemotherapy-induced fatigue in non-anaemic cancer patients. Br J Cancer. 2002 Jun;86(12):1854-7
Gullett N, Rossi P, Kucuk O, Johnstone PA. Cancer-induced cachexia: a guide for the oncologist. J Soc Integr Oncol. 2009 Fall;7(4):155-69.
Hockenberry MJ, Hooke MC, Gregurich M, McCarthy K. Carnitine plasma levels and fatigue in children/adolescents receiving cisplatin, ifosfamide, or doxorubicin. J Pediatr Hematol Oncol. 2009 Sep;31(9):664-9.
Hunter DC, Weintraub M, Blackburn GL, Bistrian BR. Branched chain amino acids as the protein component of parenteral nutrition in cancer cachexia. Br J Surg. 1989 Feb;76(2):149-53.
Inui A. Cancer Anorexia-Cachexia Syndrome: Current Issues in Research and Management. CA Cancer J Clin 2002;52:72-91
Kennedy RS, Konok GP, Bounous G, Baruchel S, Lee TD. The use of a whey protein concentrate in the treatment of patients with metastatic carcinoma: a phase I-II clinical study. Anticancer Res. 1995 Nov-Dec;15(6B):2643-9.
Laviano A, Muscaritoli M, Cascino A, Preziosa I, Inui A, Mantovani G, Rossi-Fanelli F. Branched-chain amino acids: the best compromise to achieve anabolism? Curr Opin Clin Nutr Metab Care. 2005 Jul;8(4):408-14.
Le Bricon T. Effects of administration of oral branched-chain amino acids on anorexia and caloric intake in cancer patients. Clin Nutr. 1996 Dec;15(6):337
Lis CG, Grutsch JF, Vashi PG, Lammersfeld CA. Is serum albumin an independent predictor of survival in patients with breast cancer? JPEN J Parenter Enteral Nutr. 2003 Jan-Feb;27(1):10-5.
Lissoni P, Paolorossi F, Tancini G, Barni S, Ardizzoia A, Brivio F, Zubelewicz B, Chatikhine V. Is there a role for melatonin in the treatment of neoplastic cachexia? Eur J Cancer. 1996 Jul;32A(8):1340-3.
Macciò A, Madeddu C, Gramignano G, Mulas C, Floris C, Sanna E, Cau MC, Panzone F, Mantovani G. A randomized phase III clinical trial of a combined treatment for cachexia in patients with gynecological cancers: evaluating the impact on metabolic and inflammatory profiles and quality of life. Gynecol Oncol. 2012 Mar;124(3):417-25.
MacDonald N, Easson AM, Mazurak VC, Dunn GP, Baracos VE. Understand and managing cancer cachexia. J Am Coll Surg. 2003 Jul;197(1):143-61
Mancinelli A, D’Iddio S, Bisonni R, Graziano F, Lippe P, Calvani M. Urinary excretion of L-carnitine and its short-chain acetyl-L-carnitine in patients undergoing carboplatin treatment. Cancer Chemother Pharmacol. 2007 Jun;60(1):19-26.
Mantovani G. Randomised phase III clinical trial of 5 different arms of treatment on 332 patients with cancer cachexia. Eur Rev Med Pharmacol Sci. 2010 Apr;14(4):292-301.
Murphy RA, Mourtzakis M, Chu QS, Baracos VE, Reiman T, Mazurak VC. Nutritional intervention with fish oil provides a benefit over standard of care for weight and skeletal muscle mass in patients with nonsmall cell lung cancer receiving chemotherapy. Cancer. 2011 Apr 15;117(8):1775-82. A
Murphy RA, Mourtzakis M, Chu QS, Baracos VE, Reiman T, Mazurak VC. Supplementation with fish oil increases first-line chemotherapy efficacy in patients with advanced nonsmall cell lung cancer. Cancer. 2011 Aug 15;117(16):3774-80. B
Naito T, Tashiro M, Yamamoto K, Ohnishi K, Kagawa Y, Kawakami J. Impact of cachexia on pharmacokinetic disposition of and clinical responses to oxycodone in cancer patients. Eur J Clin Pharmacol. 2012 Mar 23. [Epub ahead of print]
Oshima H, Oshima M. The inflammatory network in the gastrointestinal tumor microenvironment: lessons from mouse models. J Gastroenterol. 2012 Feb;47(2):97-106.
Pepersack T. For an operational definition of cachexia. Letter. Lancet Oncol. 2011 May;12(5):423-4.
Persson C, Glimelius B, Rönnelid J, Nygren P. Impact of fish oil and melatonin on cachexia in patients with advanced gastrointestinal cancer: a randomized pilot study. Nutrition 2005 Feb;21(2):170-8.
Polterauer S, Grimm C, Seebacher V, Rahhal J, Tempfer C, Reinthaller A, Hefler L. The inflammation-based Glasgow Prognostic Score predicts survival in patients with cervical cancer. Int J Gynecol Cancer. 2010 Aug;20(6):1052-7.
Ross PJ, Ashley S, Norton A, Priest K, Waters JS, Eisen T, Smith IE, O’Brien ME. Do patients with weight loss have a worse outcome when undergoing chemotherapy for lung cancers? Br J Cancer. 2004 May 17;90(10):1905-11.
Seely D, Wu P, Fritz H, Kennedy DA, Tsui T, Seely AJ, Mills E. Melatonin as Adjuvant Cancer Care With and Without Chemotherapy: A Systematic Review and Metaanalysis of Randomized Trials. Integr Cancer Ther. 2011 Oct 21. [Epub ahead of print]
Spiro A, Baldwin C, Patterson A, Thomas J, Andreyev HJ. The views and practice of oncologists towards nutritional support in patients receiving chemotherapy. Br J Cancer. 2006 Aug 21;95(4):431-4.
Thoresen L, Frykholm G, Lydersen S, Ulveland H, Baracos V, Prado CM, Birdsell L, Falkmer U. Nutritional status, cachexia and survival in patients with advanced colorectal carcinoma. Different assessment criteria for nutritional status provide unequal results. Clin Nutr. 2012 Jun 11.
Tisdale MJ. Mechanisms of cancer cachexia. Physiol Rev. 2009 Apr;89(2):381-410
Vijayalaxmi, Thomas CR Jr., Reiter RJ, Herman TS. Melatonin: from basic research to cancer treatment clinics. J Clin Oncol. 2002 May;20(10):2575-601
Weed HG, Ferguson ML, Gaff RL, Hustead DS, Nelson JL, Voss AC. Lean body mass gain in patients with head and neck squamous cell cancer treated perioperatively with a protein- and energy-dense nutritional supplement containing eicosapentaenoic acid. Head Neck. 2011 Jul;33(7):1027-33.
Wigmore SJ, Berber MD, Ross JA, Tisdale MJ, Fearon KC. Effect of oral eicosapentaenoic acid on weight loss in patients with pancreatic cancer. Nutr Cancer. 2000 36(2):177-84
Yang R, Cheung MC, Pedroso FE, Byrne MM, Koniaris LG, Zimmers TA. Obesity and weight loss at presentation of lung cancer are associated with opposite effects on survival. J Surg Res. 2011 Sep;170(1):e75-83.
Zamarron BF, Chen W. Dual roles of immune cells and their factors in cancer development and progression. Int J Biol Sci. 2011;7(5):651-8.
Galectin-3
Galectin-3
An emerging treatment target; role of MCP
Chronic illnesses such as cancer, cardiovascular disease, diabetes and others share common metabolic and inflammatory derangements. Novel epidemiological forecasts predict that a new category of diseases may soon be established, classifying these chronic illnesses as “Elevated Galectin-3 Diseases.”
Galectin-3 is a beta-galactoside binding lectin, ie, a carbohydrate binding protein with an affinity for beta-galactoside containing glycans (Iurisci 2000), expressed by epithelial and immune cells (Yu 2010). Several ligands for galectin-3 have been described, including lysosomal-associated membrane proteins 1 and 2, IgE, laminin & fibronectin, and Mac-2 BP (aka 90K) (Iurisci 2000, Yu 2010); and the biological function of galectin-3 is the subject of increasing research. Although the molecule is normally present in the body at relatively low concentrations, levels are greatly increased in a variety of chronic diseases, granting galectin-3 utility as a marker of disease risk and progression (Iurisci 2000). Notably, galectin-3 appears to be an active biomarker, as opposed to other surrogates such as C-reactive protein (CRP).
Galectin-3 has been shown to be involved in a large number of biological processes, including inflammation, proliferation, and fibrosis. In cardiovascular disease, it is implicated in mediating fibrosis (Lin 2009), while in cancer it appears to be active in promoting metastasis and angiogeneisis (Iurisci 2000). For instance, galectin-3 on the cell surface promotes interactions with glycans on adjacent cell surfaces, promoting tumor cell adhesion, invasion, and dissemination (Yu 2011). Conversely, suppression of galectin-3 expression in animal models results in reduced tumor growth and metastasis (Honjo 2001). Circulating galectin-3 may also play a role in the inhibition of anti-tumor T-cell activity (Peng 2008).
In 2011, the groundbreaking PREVEND study (Prevention of REnal and Vascular ENd-stage Disease) found that elevated serum galectin-3 predicted a two-fold increase in all cause mortality among the general population (deBoer 2011, 2012). A total of 7968 people were following for ten years, with overall mortality being 15.6% in the highest quintile, compared to 7.7% in the lowest quintile (De Boer 2011, 2012). For each standard deviation increase in galectin-3, there was a corresponding 46% increase risk in all cause mortality, HR 1.46 (95% CI 1.37-1.56, unadjusted), and this remained upon adjustment for age, gender, and classical risk factors (but not hs-CRP), HR 1.09 (1.01-1.19, p=0.036) (deBoer 2012).
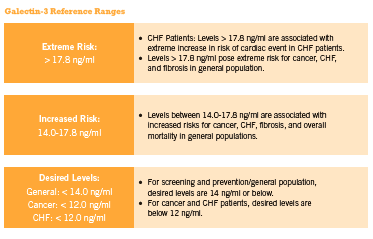
Galectin-3 can be expressed in the nucleus, cytoplasm, mitochondrion, cell surface, extracellular space, circulate freely in the blood stream (Brown 2012), and can be measured in blood samples as well as in tumor tissue samples. Normal serum levels range from 5.4-26.2 ng/mL in 95% of the population based on an analysis conducted by the BGM lab on 1099 samples from healthy patients (BG Medicine 2011). Recently, a blood test for galectin-3 has been approved by the FDA for use in assessing prognosis in patients with chronic heart failure (FDA 2012).
The Role of Galectin-3 in Chronic Disease Processes Cancer
Galectin-3 is over-expressed on the surface of cancer cells (Saleh 2009), where it affects tumor growth through several mechanisms. First, galectin-3 acts as a cell surface adhesion molecule that allows cancer cells to aggregate (Tinari 2001). Second, galectin-3 promotes the dissemination of cancer through the circulatory system, representing an important mechanism for cancer metastasis (Wang 2012). Finally, galectin-3 is also involved in angiogenesis (Nangia-Makker 2000, Yu 2007, Wang 2009, Zhao 2009).
Evaluation of galectin-3 is gaining recognition as an assessment tool in oncology, in particular as a diagnostic marker and as a prognostic marker for different cancers (Brown 2012, Chiu 2010). Studies of galectin-3 in oncology to date have focused predominantly but not exclusively on levels of galectin-3 expression in tumor tissue, rather than on circulating galectin-3, which is the basis of the test in heart failure. Tumor expression of galectin-3 may be elevated or suppressed depending on the cancer type and/ or stage; ie., “decreased expression of galectin-3 … detected in breast cancer, colonic cancer, prostate cancer and head and neck cancer compared to that of corresponding normal tissues, [and increased] galectin-3 expression … in pancreatic, vulvar and colonic carcinomas compared to normal tissues” (Brown 2012). More research on the topic is anticipated to be published in the very near future, however, studies of circulating galectin-3 thus far demonstrate an association with increased risk of cancer, and particularly with increased risk of metastasis. The reason for the apparent lack of consistency in effect between tissue expression levels and circulating levels in not known at this time.
As reported above, the PREVEND study found increased all cause mortality associated with circulating galectin-3 (deBoer 2012); for cancer specific mortality, there was a 41% increased risk among the highest quintile, HR 1.41 (1.28-1.56), unadjusted; though this relationship became insignificant after adjustment.
Iurisci et al examined circulating galectin-3 in 99 cancer patients compared with 50 healthy subjects, finding that levels were significantly higher in cancer patients (p=0.014) (2000). Interestingly, when breast cancer patients only (n=35) were compared to normal subjects, there was no significant difference in galectin-3 levels, however, patients with metatstatic breast cancer had significantly higher galectin-3 compared to those with non-metastatic disease (p<0.032) (Iurisci 2000). Similar results were found for patients with gastrointestinal (GI) cancer (n=25), non-samll cell lung cancer (n=26). Among healthy subjects, median levels were 62ng/mL (range 20-313, 95th percentile 184.3 ng/mL), while maximum levels were found among patients with metastatic GI cancers: median 320ng/mL, range 20-950m/L) (Iurisci 2000).
Saussez et al evaluated circulating galectin-3 in 102 patients with head and neck squamous cell carcinomas (HNSCCs) (2008). Galectin-3 levles were significantly higher in cancer patients compared to controls, median 3.2 and 2.39 ng/mL, respectively (p=0.03), and a threshold value of 4.3 ng/mL enabled discrimination between groups at 90% specificity and 36% sensitivity (Saussez 2008). Notably, galectin-3 levels were significantly higher among five patients with metastatic disease compared with the 97 with localized tumors. Further analysis showed a “weak, but nevertheless significant, prognostic value in terms of periods of survival for HNSCC patients” based on being over or under 4.3 ng/mL (Saussez 2008). Finally, levels of galectin-3 were found to decrease in response to treatment (chemo and/ or radiation and/ or surgery) of the primary tumor, compared to pre-treatment (p=0.001).
Other members of the galectin family have been found to be elevated in patients with breast and colorectal cancer (Barrow 2011).
More research is clearly needed to better evaluate the best use of this test in routine practice, however in the meantime galectin-3 holds immense potential as a cancer prognostic marker. In addition, human trials monitoring the effect on galcetin-3 levels in response to cancer treatment are currently lacking (Pieters 2006), however this presents an intriguing area for future research. It is also worth noting that there is little report in the medical literature of any pharmaceutical galectin-3 inhibitors; instead, the primary agent discussed in review articles thus far has been a natural agent, modified citrus pectin (Pieters 2006).
Cardiovascular Disease
As mentioned above, the galectin-3 blood test has recently received FDA approval as a prognostic test for chronic heart failure (2012). Several large studies have shown that elevated levels of plasma galectin-3 are significantly associated with higher risk of death in patients with acute decompensated and chronic heart failure (De Boer 2011a, DeBoer 2011b, Lok 2010, Shah 2010).
In the CORONA (Controlled Rosuvastatin Multinational Trial in Heart Failure) study, 1492 patients with ischaemic systolic heart failure were found to differentially respond to statin drugs according to their levels of galectin-3 (Gullestad 2010). Among patients who received rouvastatin, those whose plasma galectin-3 was below the median (≤19.0 ng/mL) had a 1) lower event rate [hazard ratio (HR) 0.65; 95% confidence interval (CI), 0.46-0.92; P= 0.014] and 2) lower total mortality (HR 0.70; 95% CI, 0.50-0.98; P= 0.038) compared with placebo. Notably, there was no benefit from rouvastatin in patients with higher levels of galectin-3.
Another key study was the HF-ACTION study (Felker 2012). In this study, galectin-3 levels were analyzed in 895 patients with chronic heart failure caused by left ventricular systolic dysfunction. Higher galectin-3 levels were associated with measures of heart failure severity, including “higher New York Heart Association class, lower systolic blood pressure, higher creatinine, higher amino-terminal proB-type natriuretic peptide (NTproBNP), and lower maximal oxygen consumption” (Felker 2012).
Cardiac fibrosis is gaining significant attention as an important risk factor in cardiac disease, in particular chronic heart failure (CHF) (deFilippi 2010), and galectin-3 plays a central role in mediating fibrosis (Liu 2009). At a site of injury/inflammation, galectin-3 is secreted into the extracellular space, activating resting fibroblasts into matrix-producing fibroblasts, thus promoting fibrosis (Liu 2009). Galectin-3 has also been found to beneficially impact other mechanisms associated with heart failure, including myofibroblast proliferation, inflammation and fibrogenesis, tissue repair, and ventricular and tissue remodeling (Liu 2009).
Other Chronic Disease Preclinical evidence suggests that elevated galectin-3 levels may be linked to an array of other chronic diseases, including autoimmune disease. Levels of circulating galectin-3 have been associated with active disease in patient’s with Behcet’s disease (Ozden 2011), juvenille arthritis (Ezzat 2011), and inflammatory bowel disease (Frolova 2009); while increased local levels have been documented in patients with liver disease (Honsawek 2011, Wanninger 2011) and asthma/ chronic obstructive pulmonary disease (Pilette 2007). Preliminary in vivo studies have found that achieving reductions in circulating galactin-3 levels delivers therapeutic benefit for the diseases in question: arthritis (Forsman 2011, Wang 2010A), inflammatory gastrointestinal conditions (Fowler 2006, Srikanta 2010), hepatic disease (Honsawek 2011, Iacobini 2011), and asthma (Zuberi 2004).
FDA Approved Galectin-3 Serum Assay The direct link between galectin-3 and numerous acute and chronic disease states gives this novel molecule an important role in diagnostics and cardiovascular therapeutics, and this is becoming more widely accepted among the medical community, especially with the advent of a galectin-3 serum assay that can now accurately measure this “active” biomarker.
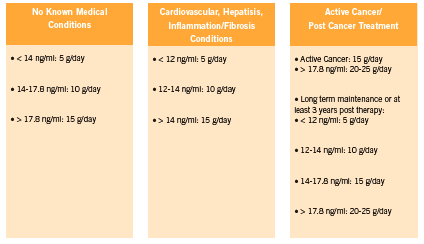
This inexpensive blood test can also be used by practitioners to assess cancer risk and progression, cirrhosis of the liver, kidney fibrosis, and other inflammation/fibrosis related conditions for which early detection is critical for optimal clinical outcome. Approximately 20% of patients exhibit changes in their galectin-3 levels every three months, and current evidence supports checking glactin-3 levels twice annually in those with stable disease (McCullough 2011). Furthermore, according to McCullough “doubling in galectin-3 level over the course of 6 months, irrespective of baseline value, identifies a high-risk patient in whom additional care … could be warranted” (2011). (See Figure 1 below outlining the reference ranges for normal and elevated serum galectin-3 in relation to disease risk).
Unlike the “bystander” biomarker C-reactive protein (CRP) that only indicates the presence of inflammation, elevated circulating galectin-3 is recognized as an “active” or “culprit” biomarker, since research shows that it actively mediates progression of numerous chronic illnesses. This means that galectin-3 is also a potentially important therapeutic target.
Targeting Galectin-3 with Modified Citrus Pectin
Although there are strong associations between galectin-3 levels and heart failure prognosis, and an impressive body of preclinical research linking galectin-3 levels to a variety of diseases, at present there is a lack of human interventional data on therapies that can lower galectin-3. There is preliminary but promising data coming out of preclinical studies however, with respect to modified citrus pectin for galectin-3 inhibition (Kidd 1996, Nangia-Makker 2002B, Olano-Martin 2003). (See Figure 2 for recommended dosages).
MCP is a form of citrus pectin that has been modified to a specific molecular weight and structure which allows it to be absorbed into the circulation and lends it its specific therapeutic properties. MCP is a complex polysaccharide fiber of repeating galacturonic acid groups with neutral sugar side chains. Regular (unmodified) citrus pectin derived from the pith of citrus fruit has a molecular weight of about 50-300 kiloDaltons, and a degree of esterification over 70%. These characteristics make regular pectin far too large to enter the blood stream. MCP can easily enter the circulation, however, when modified through a specific enzymatic process to achieve a molecular weight of 3-13 kiloDaltons, a degree of esterification under 10%, and its present specific structure (Eliaz 2006, Zhao 2008, Pieters 2006, Kidd 1996). Since galectin-3 is a beta-galactoside binding protein, and MCP is rich in beta-galactose, MCP has the ability to bind galectin-3, blocking galectin-3’s harmful effects (Nangia- Makker 2002A).
MCP is the only natural galectin-3 inhibitor demonstrated in published research that can modify the expression of galectin-3 through the natural galectin-binding affinity of its specific molecular structure, based on these authors’ literature review. For instance, a study in prostate cancer cells found that Gal-3 inhibition by antagonist GCS-100, a modified citrus pectin (MCP) product, increased cisplatin-induced apoptosis of PC3 cells (Wang 2010B). A mouse study found that MCP reduced colon cancer metastases to the liver (Liu 2008). In an animal model of an endothelial cancer, modified citrus pectin (MCP) caused a dose-dependent reduction in cancer cell survival by blocking galectin-3’s anti-apoptotic function (Johnson 2007). Furthermore, MCP sensitized cancer cells to doxorubicin such that the in vitro IC(50) of doxorubicin was reduced by 10.7-fold (Johnson 2007). Another study using breast and colon cancer xenografts models (transplanting cancer cell lines into animals and assessing growth) found that MCP was able to reduce growth, angiogenesis, and metastasis (Nangia- Makker 2002B). Another study found that MCP was able to decrease the adhesion of cancer cells to matrix (Inohara 1994). One study found that MCP can induce activation of NK cells in a leukemia model (Ramachandran 2011). Yan found that MCP has antiproliferative effects in prostate cancer cells (2010). In addition, by inhibiting galectin-3’s anti-apoptotic function and enhancing apoptosis induced by cytotoxic drugs, MCP holds the potential to dramatically increase the efficacy of conventional chemotherapy (Najmeh 2012), as well as natural and botanical compounds. These synergistic effects have been demonstrated in preclinical studies (Johnson 2007, Wang 2010B) and warrant further investigation in human clinical trials.
MCP: Beyond galectin-3
Two uncontrolled studies have examined the effect of MCP in cancer patients (Azemar 2007, Guess 2003). One study found a significantly higher PSA doubling time while on MCP for one year, compared to baseline in seven of 10 subjects (Guess 2003), while Azemar found that MCP increased ratings of quality of life overall after eight weeks (2007). Anti cancer effects were variable, with 11 of 29 assessable patients rated as having stable disease after 8 weeks (2 cycles), while 15 had progressive disease. Side effects included mild GI upset (flatulence, dyspepsia) and pruritis.
MCP has been shown to possess chelating activity in humans. As reported previously in IHP, MCP is a safe and potentially effective chelator of heavy metals and radioactive particles (Gallant 2010). MCP does not deplete essential minerals as other chelation therapies often do, and four uncontrolled trials show that patients reduced their toxic metal load by up to 76% (Eliaz 2006, Zhao 2008). MCP was dosed as 5g three times daily for three to six months.
Conclusion
As this important body of research continues to expand, galectin-3 testing is expected to become an integral component of cardiovascular and other screening panels — as routine as assessing cholesterol levels. There are a number of major laboratories that currently offer galectin-3 testing and with this simple assay, practitioners can gain more accurate insight into the risk, progression and advancement of numerous chronic inflammatory diseases. With the emergence of further research, galectin-3 is poised to become an important marker of cancer prognosis. Conversely, we can observe our patients experiencing significant clinical improvements, through firstline lifestyle modifications as well as through use of agents that can reduce inflammation and expression of galectin-3 – key among them, use of MCP.
References
Azemar M, Hildenbrand B, Haering B, Heim ME, Unger C. Clinical benefit in patients with advanced solid tumors treated with modified citrus pectin: a prospective pilot study. Clin Med: Oncol 2007, 1:73-80
Barrow H, Guo X, Wandall HH, Pedersen JW, Fu B, Zhao Q, Chen C, Rhodes JM, Yu LG. Serum galectin-2, -4, and -8 are greatly increased in colon and breast cancer patients and promote cancer cell adhesion to blood vascular endothelium. Clin Cancer Res. 2011 Nov 15;17(22):7035-46.
BG Medicine Inc. BGM Galectin-3 (Galectin-3 Assay). Document: LAB-IVD-001 R07. March 2011.
Brown ER, Doig T, Anderson N, Brenn T, Doherty V, Xu Y, Bartlett JM, Smyth JF, Melton DW. Association of galectin-3 expression with melanoma progression and prognosis. Eur J Cancer. 2012 Apr;48(6):865-74.
Chiu CG, Strugnell SS, Griffith OL, Jones SJ, Gown AM, Walker B, Nabi IR, Wiseman SM. Diagnostic utility of galectin-3 in thyroid cancer. Am J Pathol. 2010 May;176(5):2067-81.
deFilippi , CR, Felker, GM. Galectin-3 in Heart Failure—Linking Fibrosis, Remodeling, and Progression. U.S. Cardiology. 2010;7;1: 3–6.
de Boer RA, van Veldhuisen DJ, Gansevoort RT, Muller Kobold AC, van Gilst WH, Hillege HL, Bakker SJ, van der Harst P. The fibrosis marker galectin-3 and outcome in the general population. J Intern Med. 2012 Jul;272(1):55-64.
De Boer RA. Galectin-3 Levels & Mortality from All Causes in the General Population: PREVEND The Prevention of Renal and Vascular End-stage Disease (PREVEND) study results presented at the European Society of Cardiology (ESC) Congress (Aug) 2011a, in Paris, France. “Galectin-3, Cardiovascular Risk Factors and Outcome in the General Population.”
De Boer RA, Lok DJ, Jaarsma T, van der Meer P, Voors AA, Hillege HL, et al. Predictive value of plasma galectin-3 levels in heart failure with reduced and preserved ejection fraction. Ann Med. 2011b;43;1:60-8.
De Boer RA, Voors AA, Muntendam P, van Gilst WH, van Veldhuisen DJ. Galectin-3: a novel mediator of heart failure development and progression. Eur J Heart Fail. 2009;11:811–7. 37.
Weigert J, Neumeier M, Wanninger J, Bauer S, Farkas S, Scherer MN, et al. Serum galectin-3 is elevated in obesity and negatively correlates with glycosylated hemoglobin in type 2 diabetes. J Clin Endocrinol Metab. 2010;95;3:1404-11.
Eliaz I, Hotchkiss A, Fishman M, Rode D. The effect of modified citrus pectin on urinary excretion of toxic elements. Phytother Res 2006, 20:859-864.
Ezzat MH, El-Gammasy TM, Shaheen KY, Osman AO. Elevated production of galectin-3 is correlated with juvenile idiopathic arthritis disease activity, severity, and progression. Int J Rheum Dis. 2011 Oct;14(4):345-52.
Felker GM, Fiuzat M, Shaw LK, Clare R, Whellan DJ, Bettari L, Shirolkar SC, Donahue M, Kitzman DW, Zannad F, Piña IL, O’Connor CM. Galectin-3 in ambulatory patients with heart failure: results from the HF-ACTION study. Circ Heart Fail. 2012 Jan 1;5(1):72-8.
FDA, Food and Drug Administration. Galectin-3 in vitro diagnostic assay. Updated 17 June 2012. URL http://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfpcd/ classification.cfm?ID=627 Accessed 20 June 2012.
Forsman H, Islander U, Andréasson E, Andersson A, Onnheim K, Karlström A, et al. Galectin 3 aggravates joint inflammation and destruction in antigen-induced arthritis. Arthritis Rheum. 2011;63;2:445-54.
Fowler M, Thomas RJ, Atherton J, Roberts IS, High NJ. Galectin-3 binds to Helicobacter pylori O-antigen: it is upregulated and rapidly secreted by gastric epithelial cells in response to H. pylori adhesion. Cell Microbiol. 2006;8;1:44-54.
Frol’ová L, Smetana K Jr, Borovská D, Kitanovicová A, Klimesová K, Janatková I, Malícková K, Lukás M, Drastich P, Benes Z, Tucková L, Manning JC, André S, Gabius HJ, Tlaskalová-Hogenová H. Detection of galectin-3 in patients with inflammatory bowel diseases: new serum marker of active forms of IBD? Inflamm Res. 2009 Aug;58(8):503-12.
Gallant J, Rouchotas P. Modified citrus pectin (MCP): heavy metal chelator, possible cancer treatment. IHP Sept 2010: 58-61.
Guess BW, Scholz MC, Strum SB, Lam RY, Johnson HJ, Jennrich RI. Modified citrus pectin (MCP) increases the prostate-specific antigen doubling time in men with prostate cancer: a phase II pilot study. Prostate Cancer Prostatic Dis 2003;6:301-304.
Gullestad L, Ueland T, Kjekshus J, Nymo SH, Hulthe J, Muntendam P, Adourian A, Böhm M, van Veldhuisen DJ, Komajda M, Cleland JG, Wikstrand J, McMurray JJ, Aukrust P; on behalf of the CORONA Study Group. Galectin-3 predicts response to statin therapy in the Controlled Rosuvastatin Multinational Trial in Heart Failure (CORONA). Eur Heart J. 2012 Apr 26.
Honjo Y, Nangia-Makker P, Inohara H, Raz A. Down-regulation of galectin-3 suppresses tumorigenicity of human breast carcinoma cells. Clin Cancer Res. 2001 Mar;7(3):661-8.
Honsawek S, Chongsrisawat V, Praianantathavorn K, Theamboonlers A, Poovorawan Y. Elevation of serum galectin-3 and liver stiffness measured by transient elastography in biliary atresia. Eur J Pediatr Surg. 2011;21;4:250-4.
Iacobini C, Menini S, Ricci C, Fantauzzi CB, Scipioni A, Salvi L, et al. Galectin-3 ablation protects mice from diet-induced NASH: a major scavenging role for galectin-3 in liver. J Hepatol. 201;54;5:975-83.
Inohara H, Raz A. Effects of natural complex carbohydrate (citrus pectin) on murine melanoma cell properties related to galectin-3 functions. Glycoconj J. 1994 Dec;11(6):527-32.
Iurisci I, Tinari N, Natoli C, Angelucci D, Cianchetti E, Iacobelli S. Concentrations of galectin-3 in the sera of normal controls and cancer patients. Clin Cancer Res. 2000 Apr;6(4):1389-93
Johnson KD, Glinskii OV, Mossine VV, Turk JR, Mawhinney TP, Anthony DC, Henry CJ, Huxley VH, Glinsky GV, Pienta KJ, Raz A, Glinsky VV. Galectin-3 as a potential therapeutic target in tumors arising from malignant endothelia. Neoplasia. 2007 Aug;9(8):662-70.
Kidd P. A new approach to metastasis cancer prevention: modified citrus pectin (MCP), a unique pectin that blocks cell surface lectins. Altern Med Rev 1996;1:4-10.
Lin YH, Lin LY, Wu YW, Chien KL, Lee CM, Hsu RB, Chao CL, Wang SS, Hsein YC, Liao LC, Ho YL, Chen MF. The relationship between serum galectin-3 and serum markers of cardiac extracellular matrix turnover in heart failure patients. Clin Chim Acta. 2009 Nov;409(1-2):96-9.
Liu HY, Huang ZL, Yang GH, Lu WQ, Yu NR. Inhibitory effect of modified citrus pectin on liver metastases in a mouse colon cancer model. World J Gastroenterol 2008, 14:7386-7391.
Liu YH, D’Ambrosio M, Liao TD, Peng H, Rhaleb NE, Sharma U, André S, Gabius HJ, Carretero OA. N-acetyl-seryl-aspartyl-lysyl-proline prevents cardiac remodeling and dysfunction induced by galectin-3, a mammalian adhesion/growth-regulatory lectin. Am J Physiol Heart Circ Physiol. 2009 Feb;296(2):H404-12.
Lok DJ, Van Der Meer P, de la Porte PW, Lipsic E, Van Wijngaarden J, Hillege HL, et al. Prognostic value of galectin-3, a novel marker of fibrosis, in patients with chronic heart failure: data from the DEAL-HF study. Clin Res Cardiol. 2010;99;5:323-8.
McCullough PA, Olobatoke A, Vanhecke TE. Galectin-3: a novel blood test for the evaluation and management of patients with heart failure. Rev Cardiovasc Med. 2011;12(4):200-10.
Najmeh T, Houri S, Parvin M, Firouzeh B, Arash HN, Abdolfattah S, Ebrahim H. Combination effect of PectaSol and Doxorubicin on viability, cell cycle arrest and apoptosis in DU-145 and LNCaP prostate cancer cell lines. Cell Biology International 2012, Immediate Publication, doi:10.1042/CBI20110309.
Nangia-Makker P, Conklin J, Hogan V, Raz A. Carbohydrate-binding proteins in cancer, and their ligands as therapeutic agents. Trends Mol Med 2002; 8:187-192.A
Nangia-Makker P, Hogan V, Honjo Y, Baccarini S, Tait L, Bresalier R, et al. Inhibition of human cancer cell growth and metastasis in nude mice by oral intake of modified citrus pectin. J Natl Cancer Ins 2002;94:1854-1862.B
Nangia-Makker P, Honjo Y, Sarvis R, Akahani S, Hogan V, Pienta KJ, et al. Galectin-3 induces endothelial cell morphogenesis and angiogensis. Am J Pathol 2000, 156:899-909.
Olano-Martin E, Rimbach GH, Gibson GR, Rastall RA. Pectin and pecticoligosaccharides induce apoptosis in in vitro human colonic adenocarcinoma cells. Anticancer Res 2003;23:341 346.
Özden MG, Caycı YT, Tekin H, Çoban AY, Aydın F, Sentürk N, Bek Y, Cantürk T, Turanlı AY. Serum galectin-3 levels in patients with Behçet’s disease: association with disease activity over a long-term follow-up. J Eur Acad Dermatol Venereol. 2011 Oct;25(10):1168-73.
Peng W, Wang HY, Miyahara Y, Peng G, Wang RF. Tumor-associated galectin-3 modulates the function of tumor-reactive T cells. Cancer Res. 2008 Sep 1;68(17):7228-36.
Pienta KJ, Naik H, Akhtar A, Yamazaki K, Replogle TS, Lehr J, et al. Inhibition of spontaneous metastasis in a rat prostate cancer model by oral administration of modified citrus pectin. J Natl Cancer Inst 1995;87:348-353.
Pieters RJ. Inhibition and detection of galectins. Chembiochem. 2006 May;7(5):721-8.
Pilette C, Colinet B, Kiss R, André S, Kaltner H, Gabius HJ, Delos M, Vaerman JP, Decramer M, Sibille Y. Increased galectin-3 expression and intra-epithelial neutrophils in small airways in severe COPD. Eur Respir J. 2007 May;29(5):914-22.
Ramachandran C, Wilk BJ, Hotchkiss A, Chau H, Eliaz, I Melnick SJ. Activation of human T-helper/inducer cell, T-cytotoxic cell, B-cell, and natural killer (NK)-cells and induction of natural killer cell activity against K562 chronic myeloid leukemia cells with modified citrus pectin. BMC Complementary and Alternative Medicine. 2011;11:59.
Saleh HA, Feng J, Tabassum F, Al-Zohaili O, Husain M, Giorgadze T. Differential expression of galectin-3, CK19, HBME1, and Ret oncoprotein in the diagnosis of thyroid neoplasms by fine needle aspiration biopsy. Cytojournal. 2009 Sep 18;6:18.
Saussez S, Lorfevre F, Lequeux T, Laurent G, Chantrain G, Vertongen F, Toubeau G, Decaestecker C, Kiss R. The determination of the levels of circulating galectin-1 and -3 in HNSCC patients could be used to monitor tumor progression and/or responses to therapy. Oral Oncol. 2008 Jan;44(1):86-93.
Shah RV, Chen-Tournoux AA, Picard MH, van Kimmenade RR, Januzzi JL. Galectin-3, cardiac structure and function, and long-term mortality in patients with acutely decompensated heart failure. Eur J Heart Fail. 2010;12;8:826-32.
Srikanta BM, Sathisha UV, Dharmesh SM. Alterations of matrix metalloproteinases, gastric mucin and prostaglandin E(2) levels by pectic polysaccharide of swallow root (Decalepis hamiltonii) during ulcer healing. Biochimie. 2010;92;2:194-203.
Tinari N, Kuwabara I, Huflejt ME, Shen PF, Iacobelli S, Liu FT. Glycoprotein 90K/ MAC-2BP interacts with galectin-1 and mediates galectin-1-induced cell aggregation. Int J Cancer. 2001 Jan 15;91(2):167-72.
Wang YG, Kim SJ, Baek JH, Lee HW, Jeong SY, Chun KH. Galectin-3 Increases the Motility of Mouse Melanoma Cells by Regulating MMP-1 Expression. Exp Mol Med. 2012 Mar 21. [Epub ahead of print]
Wang CR, Shiau AL, Chen SY, Cheng ZS, Li YT, Lee CH, et al. Intra-articular lentivirus-mediated delivery of galectin-3 shRNA and galectin-1 gene ameliorates collagen-induced arthritis. Gene Ther. 2010;17;10:1225-33.A
Wang Y, Nangia-Makker P, Balan V, Hogan V, Raz A. Calpain activation through galectin-3 inhibition sensitizes prostate cancer cells to cisplatin treatment. Cell Death Dis. 2010 Nov 18;1:e101.B
Wang Y, Nangia-Makker P, Tait L, Balan V, Hogan V, Pienta KJ, et al. Regulation of prostate cancer progression by galectin-3. Am J Pathol. 2009;174;4:1515-23.
Wanninger J, Weigert J, Wiest R, Bauer S, Karrasch T, Farkas S, Scherer MN, Walter R, Weiss TS, Hellerbrand C, Neumeier M, Schäffler A, Buechler C. Systemic and hepatic vein galectin-3 are increased in patients with alcoholic liver cirrhosis and negatively correlate with liver function. Cytokine. 2011 Sep;55(3):435-40.
Weigert J, Neumeier M, Wanninger J, Bauer S, Farkas S, Scherer MN, et al. Serum galectin-3 is elevated in obesity and negatively correlates with glycosylated hemoglobin in type 2 diabetes. J Clin Endocrinol Metab. 2010;95;3:1404-11.
Yan J, Katz A. PectaSol-C modified citrus pectin induces apoptosis and inhibition of proliferation in human and mouse androgen-dependent and- independent prostate cancer cells. Integr Cancer Ther 2010;9:197-203.
Yu LG, Andrews N, Zhao Q, McKean D, Williams JF, Connor LJ, et al. Galectin-3 interaction with Thomsen-Friedenreich disaccharide on cancerassociated MUC1 causes increased cancer cell endothelial adhesion. J Biol Chem. 2007;5;282(1):773-81.
Yu LG. Circulating galectin-3 in the bloodstream: An emerging promoter of cancer metastasis. World J Gastrointest Oncol. 2010 Apr 15;2(4):177-80.
Zaia Povegliano L, Oshima CT, de Oliveira Lima F, Andrade Scherholz PL, Manoukian Forones N. Immunoexpression of galectin-3 in colorectal cancer and its relationship with survival. J Gastrointest Cancer. 2011 Dec;42(4):217-21.
Zhao ZY, Liang L, Fan X, Yu Z, Hotchkiss AT, Wilk BJ, et al. The role of modified citrus pectin as an effective chelator of lead in children hospitalized with toxic lead levels. Altern Ther Health Med 2008, 14:34-38.
Zhao Q, Guo X, Nash GB, Stone PC, Hilkens J, Rhodes JM, et al. Circulating galectin-3 promotes metastasis by modifying MUC1 localization on cancer cell surface. Cancer Res. 2009;69;17:6799-806.
Zuberi RI, Hsu DK, Kalayci O, Chen HY, Sheldon HK, Yu L, et al. Critical role for galectin-3 in airway inflammation and bronchial hyperresponsiveness in a murine model of asthma. Am J Pathol. 2004;165;6:2045-53.
Low Dose Naltrexone
Low Dose Naltrexone
Role as adjunct cancer treatment
Abstract
Research in the area of naltrexone has shown both promise and controversy for cancer treatment. Ongoing validation regarding the differing mechanisms of action, the dosage, and the timing of the dose is required. In reviewing the mechanism of action for low-dose naltrexone, more were identified than originally anticipated, which range from a vague endogenous immune response to a complex mechanisms involving LDN affecting a blockade at the OGF-OGFr axis. This article presents the historical use of naltrexone, the current information about the low-dose options and use recommendations, the receptors involved with some controversial effects on cancer with respect to mechanisms of action, and calls for more evidence in the form of phase three clinical trials.
Naltrexone – Mechanism of Action in Treatment of Cancer
The task of bringing together single dimension information from laboratory research examining cellular, vascular, and endocrine events to understand the three dimensional mechanisms of disease in the human body is daunting. The reliance upon research and scientific principles weighs heavy in cancer where much is unknown about disease progression, especially in the form of metastasis or recurrence. Currently information is elucidated about mechanisms of action, from which arise more questions and theories, which in turn provokes the critically-thinking reader to utilize the available information to the best of their abilities. An example of the evolution of a treatment that was first presented as novel and which is now entering the realm of evidence-based medicine is that of low dose Naltrexone (LDN) for use in autoimmune disease and cancer. For the purpose of this article, LDN and its effects in cancer will be the primary focus.
There is little clinical or research evidence beyond the level of dramatic reports from Dr.’s Bihari, Berkson and Rubin (Berkson 2009) to support the use of LDN in treatment of cancer. The authors present case reports of three people diagnosed with pancreatic cancer, treated successfully with the use of Alpha Lipoic Acid and LDN, and call for clinical trials for further investigation of their protocol. In elucidating the mechanism of action for this success they suggested an endogenous immune response. Suffice to say it is a start, and a proposed mechanism of action may eventually create further research to prove or disprove. Further, there is recent evidence in cell studies elucidating more than one anti-cancer mechanism of LDN. Low-dose Naltrexone (LDN) presents itself with complexity: there are different receptors involved, and the resulting effects of LDN on cancer have varied widely. This article reviews the recent data regarding the mechanisms of action and the proposed resulting effects of LDN in treatment of cancer.
Naltrexone History in Brief Naltrexone is used to treat opioid substance abuse and acts by blocking opioid receptors in the brain. It was approved by the US FDA in 1984 in 50 mg dose increments as a competitive narcotic antagonist, for the purpose of stopping or reducing the effects of a heroin or opium overdose and/or for the reduction of opioid withdrawal symptoms (Lowdosenaltrexone.org May 4 2010). For the purposes of opioid blockade this 50mg ‘high dose’ Naltrexone is in tablet form for oral consumption away from meals. Depending upon the type of addiction there are different prescription regimens ranging from daily to every other day schedules (PubMed Health 2011). There is very little understood about Naltrexone beyond opioid blockade as the patent expiry led to it becoming dormant as a prescription item for a number of years. For the treatment of addiction, ‘high-dose’ Naltrexone exerts opposite effects on the Mu Opioid Protein (MOP) receptor at the plasma membrane, and can inhibit secretion of opioids in both brain and adrenal glands via beta endorphins and enkephalins (Figure 1). Naltrexone has been used to treat opioid overdose, opioid addiction (Gonzalez 2004) and addictions to other substances such as alcohol (Chick 2000, Davidson 1999). Naltrexone is conventionally dosed up to a maximum of 350 mg QD. This high-dose use is limited by the adverse effects which are similar to opioid withdrawal and these effects can even occur in patients without previous exposure to opioids (Hollister 1981).
In contrast the Low-dose naltrexone (LDN) dose ranges from 0.1-4.5 mg QD and has not produced adverse effects. The off-label use of low-dose Naltrexone started with Dr. Bihari and has been in use since the 1980’s, subsequently described via case report as an anti-cancer strategy in 2009 by Berkson and Rubin. LDN has been used off-patent for treatment of autoimmune illnesses and cancer since the late 1980’s. It is compounded into tablets or a powder which is mixed with water for oral consumption. Comparative absorption data and information on suppository or injectable doses is limited and unreliable at this time. LDN interacts with opioid analgesics such as Hydrocodone, Oxycodone, Oxymorphone and other opiate/opioid narcotics and thus should be avoided in such instances. It is recommended to gradually reduce the intake and then eliminate the use of any opioid narcotic for 10-12 days before starting LDN. The compounded powder for oral consumption of LDN has a shorter, temperature-sensitive shelf-life and a bitter taste when taken on its own, in comparison to the compounded LDN tablet for oral consumption (Figure 2) (Fawcett 1997).
Receptor Review
Transmembrane receptors in the human body regulate the majority of cellular metabolism and physiology. The largest group of receptors belongs to a group coupled to G-proteins (G-Protein-Coupled Receptors, or GPCRs) (Lefkowitz 2007). The superfamily of GPCRs regulates metabolism, including cellular replication; they are the targets for drug therapy in treatment of cancer and autoimmune disease (Jacoby 2006, Pierce 2002). The genes encoding μ-, δ-, κ-, ζ- and nociceptin Opioid Proteins (MOP- Figure 1, DOP, KOP, OGFr, NOP respectively) belong in the super-family of the GPCRs (Brown 2008, Corbett 2006, Wang 2001) are the transmembrane receptors targeted in the treatment of narcotic abuse (Wang 2001), and are suggested to be the target in the treatment of autoimmune disease (Zagon 2009) and cancer with LDN (Brown 2008). However, more recent research has indicated that the regulatory effect on cancer cell growth extends beyond that of the MOP and to an endogenous opioid factor also referred to as Met5-Enkephalin or Opioid Growth Factor (OGF, Figure 3), and its receptor the ζ-opioid receptor or Opioid Growth Factor Receptor (OGFr). OGF has a role in growth inhibition and OGFr determines the properties of cell proliferation (Zagon 2003). Ongoing phase one and two trials on squamous cell carcinoma of the head and neck and hepatocellular cancer respectively are investigating how OGF binds and activates the OGFr to inhibit angiogenesis and tumor cell growth (Clinicaltrials.gov 2010A, Clinicaltrials.gov 2010B).
Naltrexone – Past, Present and Future
Naltrexone (Figure 2) was approved by the US FDA in 1984 in 50mg doses (max dose 350mg) and acts as a narcotic antagonist. It is used to treat and stop heroin or opium overdoses and withdrawal (Gonzalez 2004), and to treat substance addiction such as alcoholism (Chick 2000, Davidson 1999). Since the patent expired other uses have thus been investigated. In the 1990’s Dr. Bihari first used LDN to treat people with AIDS and then later on to treat people with cancer (Bihari 1995, Lowdosenaltrexone.
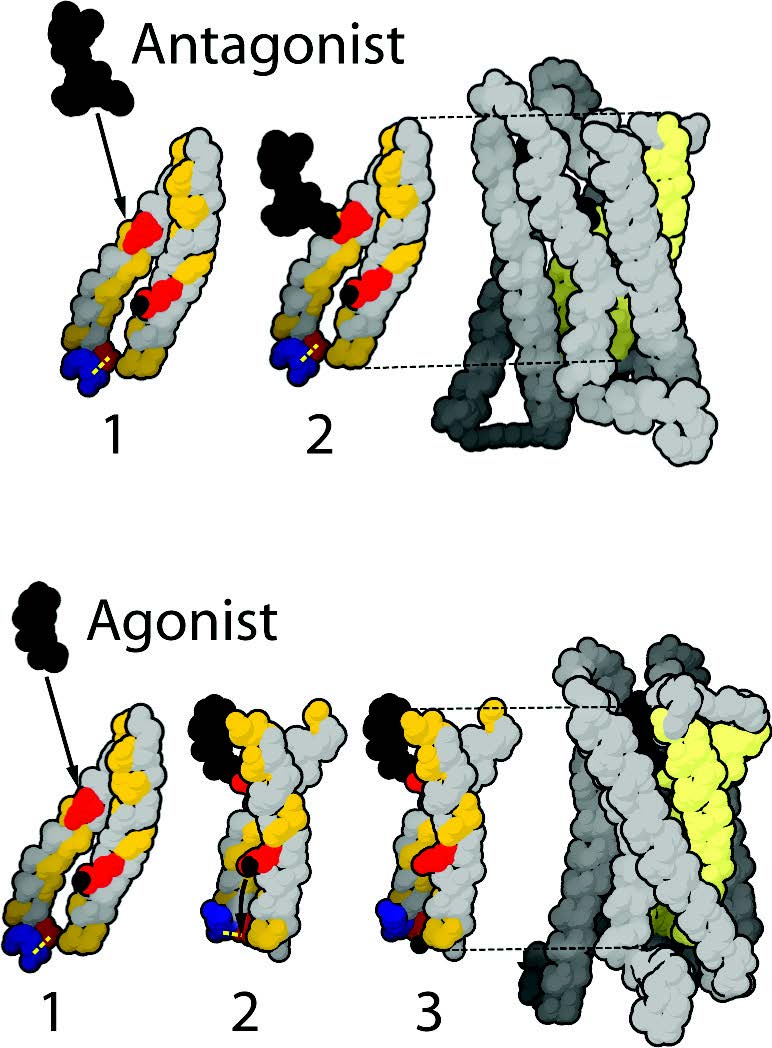
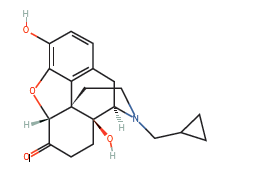
org May 18 2010). There have been reports of successful treatment of various cancers (Berkson 2009, Dagleish 2005, Hytrek 1996, Zagon 2000), AIDS (Bihari 1995, Gekker 2001), Multiple Sclerosis (Cree 2010), Autism (Panskepp 1991), and autoimmune disorders such as Crohn’s Disease (Smith 2007) and fibromyalgia (Brown 2008). Sustained release naltrexone (32mg) has now completed investigation in Phase 2 trials with bupropion for the treatment of obesity (Clinicaltrials.gov 2010B).
Naltrexone – Mechanisms of Action
As an opioid antagonist, high dose (50mg – 350mg) naltrexone blocks secretion of opioids in the brain and adrenal glands via the MOP. At low dose (3-4.5mg) nocturnal dose-timing of naltrexone is hypothesized to block opioid receptor expression (MOP, DOP, KOP) and increases circulation of met-enkephalin (Figure 3) and beta-endorphin for up to six hours. The MOP is hypothesized to play a role in the various responses of the immune system to stress, infection and malignant transformation (Makman 1994). LDN has several proposed theoretical mechanisms of action depending on whether the treatment is targeted for cancer or autoimmune disease. An recent cell study investigation revealed controversy. In the anti-cancer role of LDN the OGF/OGFr axis can be allosterically modulated to create multi-dimensional effects on signaling pathways for cellular replication. Allosteric modulation is also demonstrated by the human immune system and readers will recall that the subtypes of Th cells are allosterically modulated to create immune functionality. According to the investigator Dr. Ian Zagon at Penn State University (e-mail communication May 12, 2010): “All human cancers are equipped with the OGF/OGFr axis and depend on it for regulation of cell proliferation. NTX (naltrexone) works by blocking the OGF-OGFr axis. It causes an up-regulation. If the dose is low enough, you allow time for OGF to interact with OGFr to cause a depression in cell proliferation. If you give repeated low doses or a high dose you get up-regulation but no interaction between peptide and receptor. Since the OGF-OGFr axis is tonically active – it is on all the time and watching over the pace of cell proliferation by inhibitory pathways, blockade of OGF-OGFr by naltrexone continuously allows cells to be unregulated in cell division and you have more cells produced.” For the purpose of illustrating further the complexity of this drug there are several mechanisms of action proposed in treatment of the following different types of cancer:
•In neuroblastoma, the low (3-4.5mg) and high dose (100 mg) naltrexone administered demonstrated a direct modulating effect on oncogenesis. The low dose naltrexone elevated opiate receptors and circulating beta endorphin and met-enkephalin through the four to six hour receptor blockade (McLaughlin 1987) and resulted in decreased oncogenesis.
• A small study of 14 patients with untreatable metastatic solid tumors treated with melatonin (20mg hs) and naltrexone(100mgQD) to augment Interleukin-2 therapy demonstrated that lymphocyte production is amplified compared with melatonin and IL-2 therapy alone (Lissoni 2002).
• In Nude mice with colon cancer (HT-29) treated with 0.1mg/ kg naltrexone demonstrated that tumorigenicity was inhibited by opioids via a 2.5 increase in plasma met5-enkephalin in naltrexone-treated mice and an 85% reduction of binding capacity of 3H-Met5-enkephalin in tumor tissue when compared to the control group (Hytrek 1996).
• In pancreatic cancer MIA PaCa-2 clonal cell lines the singlelow- dose naltrexone-induced amplification of the OGFr axis delayed the G1/S inter-phase of the cell cycle via CDK inhibition of p16 or p21, causing a decrease in CDK4/CDK2 activity and RB protein phosphorylation, thus halting DNA synthesis and subsequent cell growth (Zagon 2007).
It remains to be determined how LDN exerts its effects via either an independent or combined extracellular or cytoplasmic effect on cancer cell replication. These give rise to the confirmation that often LDN is not a single therapy provided in isolation of other cancer treatment, and that there are pharmacokinetic properties to LDN which affect enkephalin production, lymphocyte production, and cyclin dependent kinases on control of the cell cycle.
Summary The number of cancer survivors alive compared to those seeking a cure can be described as an inverse relationship. There is a distinct lack of cancer survivors, in spite of all the enormity of efforts ongoing to seek a cure. Research in the area of naltrexone has shown both promise and controversy for cancer treatment and requires ongoing validation regarding the differing mechanisms of action, the dosage, and the timing of the dose and the type of cancer. In reviewing the mechanism of action for low-dose naltrexone, more were identified than originally elucidated by historical data and case reports. An endogenous immune response has been suggested via case report as one possible theory, as well as an increase in enkephalins, lymphocytes, and allosteric blockade at the OGF-OGFr axis. It is a daily challenge to understand the intended and untoward influences of naltrexone upon tumor cell growth because of the differing receptors involved and to tailor a specific protocol based upon the current evidence for people with cancer. In looking to the future use of LDN, there is a need for collaborative effort between the research bench and the patient that may provide more information regarding the mechanisms of tumor initiation, progression and promotion to help extend the limit of one’s knowledge, skills and judgment when treating the complex disease of cancer.
References
Berkson BM, Rubin DM, Berkson AJ. Revisiting the ALA/N (alpha-lipoic acid/lowdose naltrexone) protocol for people with metastatic and nonmetastatic pancreatic cancer: a report of 3 new cases. Integr Cancer Ther. 2009 Dec;8(4):416-22.
Bihari B. Efficacy of low dose naltrexone as an immune stabilizing agent for the treatment of HIV/AIDS [letter]. AIDS Patient Care. 1995 Feb;9(1):3.
Brown N, Panskepp J. Low-dose naltrexone for disease prevention and quality of life. Med Hypotheses. 2008;72(3):333-7.
Chick J, Anton R, Checinski K, Croop R, Drummond DC, Farmer R, Labriola D, Marshall J, Moncrieff J, Morgan MY, Peters T, Ritson B. A multicentre, randomized, double-blind, placebo-controlled trial of naltrexone in the treatment of alcohol dependence or abuse. Alcohol Alcohol. 2000 Nov-Dec;35(6):587-93.
Clinicaltrials.gov. http://www.cancer.gov / clinicaltrials /search /view?cdrid = 599953&ver sion =HealthProfessional & protocolsearchid = 8291854. Accessed Nov 2010A.
Clinicaltrials.gov. http://clinicaltrials.gov/ct2/show/NCT00711477. Accessed Nov 2010B.
Corbett AD, Henderson G, McKnight AT, Paterson SJ. 75 years of opioid research: the exciting but vain quest for the Holy Grail. Br J Pharmacol. 2006 Jan;147 Suppl 1:S153-62.
Cree BA, Kornyeyeva E, Goodin DS. Pilot trial of low-dose naltrexone and quality of life in multiple sclerosis. Ann Neurol. 2010 Aug;68(2):145-50.
Dalgleish A, Whelan M. Novel immunotherapeutic approaches to prostate cancer. Curr Opin Mol Ther. 2005 Feb;7(1):30-4.
Davidson D, Palfai T, Bird C, Swift R. Effects of naltrexone on alcohol selfadministration in heavy drinkers. Alcohol Clin Exp Res. 1999 Feb;23(2):195-203.
Fawcett JP, Morgan NC, Woods DJ. Formulation and stability of naltrexone oral liquid for rapid withdrawal from methadone. Ann Pharmacother. 1997 Nov;31(11):1291-5.
Gekker G, Lokensgard J, Peterson PK. Naltrexone potentiates anti-HIV-1 activity of antiretroviral drugs in CD4+ lymphocyte cultures. Drug Alc Depend. 2001;64:257-63.
Gonzalez G, Oliveto A, Kosten TR. Combating opiate dependence: a comparison among the available pharmacological options. Expert Opin Pharmacother. 2004 Apr;5(4):713-25.
Hollister LE, Johnson K, Boukhabza D, Gillespie HK. Aversive effects of naltrexone in subjects not dependent on opiates. Drug Alcohol Depend. 1981 Aug;8(1):37-41.
Hytrek SD, McLaughlin PJ, Lang CM, Zagon IS. Inhibition of human colon cancer by intermittent opioid blockade with naltrexone. Cancer Lett. Mar 29, 1996;101(2):159-64.
Jacoby E, Boulehal R, Gerspacher M, Seuwen K. The 7 TM G-protein-coupledreceptor target family. ChemMedChem. 2006;1(8):761-82.
Lefkowitz RJ. Seven transmembrane receptors: something old, something new. Acta Physiol (Oxf). 2007 May;190(1):9-19.
Lissoni P, Malugani F, Malysheva O, Kozlov V, Laudon M, Conti A, Maestroni G. Neuroimmunotherapy of untreatable metastatic solid tumors with subcutaneous lowdose interleukin-2, melatonin and naltrexone: modulation of interleukin-2-induced antitumor immunity by blocking the opioid system. Neuro Endocrinol Lett. 2002 Aug;23(4):341-4.
Lowdosenaltrexone.org. www.lowdosenaltrexone.org. Accessed May 4 2010.
Lowdosenaltrexone.org. www.lowdosenaltrexone.org/ldn_and_cancer.htm. Accessed May 18 2010.
Makman M. Morphine receptors in immunocytes and neurons. Adv Neuroimmunol. 1994;4:69-82.
McLauMghlin PJ, Zagon I. Modulation of human neuroblastoma transplanted into nude mice by endogenous opioid systems. Life Sci. 1987;41:1465-72.
Panksepp J, Lensing P, Leboyer M, Bouvard MP. Naltrexone and other potential new pharmacological treatments of autism. Brain Dysfunc. 1991;4:281-300.
Pierce KL, Premont R, Lefkowitz RJ. Seven transmembrane receptors. Nat Rev Mol Cell Biol. 2002;3(9):639-50.
PubChem. http://pubchem.ncbi.nlm.nih.gov. Accessed Nov 2010.
PubMed Health. http://www.ncbi.nlm.nih.gov/pubmedhealth/PMH0000853/ accessed July 17 2011.
Smith J, Stock H, Bingaman S, Mauger D, Rogosnitzky M, Zagon I. Low-dose naltrexone therapy improves active Crohn’s disease. Amer J Gastroenterology. 2007;102(4):820-8.
Wang D, Rachal KM, Bilsky EJ, Sadee W. Inverse Agonists and neutral antagonists at mu opioid receptor (MOR): possible role of basal receptor signaling in narcotic dependence. J Neurochem. 2001;77(6):1590-600.
Zagon IS, McLaughlin P. Opioids and the apoptotic pathway in human cancer cells. Neuropeptides. 2003;37:79-88.
Zagon IS, Rahn KA, Turel KP, McLaughlin PJ. Endogenous Opioids Regulate Expression of Experimental Autoimmune Encephalomyelitis: A New Paradigm for the Treatment of Multiple Sclerosis. Exp Biol Med. 2009;234(11):1383-92.
Zagon IS, Roesener CD, Verderame MF, Ohlsson-Wilhelm BM, Levin RJ, McLaughlin PJ. Opioid growth factor regulates the cell cycle of human neoplasias. Int J Oncol. 2000;17:1053-61.
Zagon IS, Verderame MF, Hankins J, McLaughlin PJ. Overexpression of the opioid growth factor receptor potentiates growth inhibition in human pancreatic cancer cells. International Journal of Oncology. 2007;30(4):775-783.
Study doubting niacin’s heart effects challenged
A high-profile U.S. study raising doubts about the heart-protective effects of niacin came under severe criticism at a recent American Heart Association (AHA) meeting. The Aim-High study (n = 3414) found that adding Abbott Laboratories Inc.’s Niaspan, a long-acting version of niacin, to cholesterol-lowering statins did not further prevent heart attacks, strokes, and other adverse events. However, Dr. Philip Barter of the Heart Research Institute in Sydney, Australia, said that this study was not properly designed to test the benefits of niacin. “This trial disturbs me greatly,” said Barter, who was designated by the AHA conference to critique the study. “The design was such that in no way could it test the hypothesis of raising HDL [high-density lipoprotein] or the benefit of niacin.” Barter said any conclusions on niacin must be suspended until the results of a much larger study involving a Merck & Co drug that includes niacin (n = 25 000) are released. That study is expected to be completed next year. “If that trial doesn’t show a positive effect, niacin is finished,” Barter said. “We shouldn’t draw any conclusions or change practice based on this.”
Cytochroma announces positive Phase 2b results for CTAP101 Capsules
Cytochroma has announced positive Phase 2b clinical trial results for its CTAP101 Capsules that effectively and safely lowered elevated plasma intact parathyroid hormone (iPTH) in patients with Stage 3 or 4 chronic kidney disease (CKD) and vitamin D insufficiency. “CTAP101 Capsules performed well above our expectations in the Phase 2b study,” stated Dr. Charles W. Bishop, President and CEO of Cytochroma. “No marketed therapy both effectively controls elevated serum parathyroid hormone levels and corrects the underlying vitamin D insufficiency in CKD patients. These strong data indicate that CTAP101 Capsulesisahighlydifferentiatedproduct compared to current treatment options. We look forward to initiating Phase 3 trials later this year.”
New diagnostic tool for Tuberculosis in the North
A new diagnostic device that will help fight the spread of tuberculosis (TB) in theNorthhasbeenunveiled.Theportable and automated device, Xpert MTB/RIF has recently been approved by Health Canada to diagnose TB. It can identify TB and also drug resistant TB from a sputum sample in only 90 minutes, which is much faster than current laboratory methods. The technology is based on a new molecular method that identifies the TB bacteria’s specific DNA in the sample and is thought to be more accurate and specific than current diagnostic methods.
Express Scripts Canada releases findings of 2011 Drug Trend Report
Express Scripts Canada has announced the findings of the Express Scripts Canada 2011 Drug Trend Report, an annual report that provides one of the country’s most comprehensive analyses into the use of prescription drugs and related trends in the private sector throughout Canada. Among the primary findings of the report is that while drug spend remained flat in 2011, private drug plans in Canada wasted approximately $5 billion in drug spend for the 2011 calendar year. The report defines waste as “spending that does not result in improvements to health outcomes”. Other key findings included: 1) increased average annual drug spend perclaimant ($761 in 2011 vs $757 in 2010); 2) increased utilization by ~2.5% every year (average number of prescriptions per claimant in 2011 was 12.6 vs 12.3 in 2010); 3) decreased cost per prescription ($60.45 in 2011 vs $61.55 in 2010); 4) increased average dispensing fee ($10.74 in 2011 vs $10.64 in 2010); 5) decreased generic prices from approximately 60% of brand price to between 30%-45%; and 6) increased average price per brand prescription.
mihealth introduces mihealthView TM app to the app store
mihealth Global Systems has announced thelaunchofarevolutioninpersonalized healthcare communications with the mihealthView TM app, which is available on the App Store for iPhone, iPad, and iPod touch. Patients can now manage and store their personal medical information and histories on their iPhone, iPad, and iPod touch and have safe, secure, and global access to their personal health records. Patients can track their own health information, including current prescriptions and medications, allergies, test results, and vaccination histories. The mihealthView TM app is available for $9.99 at www.itunes.ca/appstore. For more information visit: www.mihealth.com