Hospital acquired C difficile infection remains a common cause of morbidity and mortality. An estimated 1% of patients admitted to hospital in Canada acquire C difficile, and an estimated 10% of them die regardless of their basis for admission to the hospital. Bio-K+® is the first natural health product to have a Health Canada approved claim for reducing risk of acquiring C difficile infection. Makers Bio-K Plus International Inc call upon health care providers and hospital administrators across the country to make coadministration of probiotics with antibiotics in hospital settings a standard of care. They estimate adopting such a standard would save the Canadian healthcare system over $200 million per year.
Six medical heroes named Canada’s 2013 Hall of Fame inductees
The Canadian Medical Hall of Fame is the only national Hall of Fame in the world dedicated to celebrating medical heroes. It creates a tribute to those who through discovery and innovation have contributed to better health in Canada and the world. The 2013 inductees are Dr. Antoine Hakim, Dr. David MacLenna, Dr. Claude Roy, Dr. Arnold Naimark, Dr. Bette Stephenson, and the Late Dr. Ian Rusted.The 2013 Induction Ceremony, presented by BMO Harris Private Banking, will be held in Halifax on May 2. Biographies of the current laureates are available on the Canadian Medical Hall of Fame website.
Dr Erika Schwartz, MD
Training integrative healthcare providers for delivery of Bioidentical Hormone Therapy
Dr. Erika Schwartz is the go-to physician for all types of healthcare providers looking to train in prevention and wellness. In that context she is perhaps best known for her role as physician educator in the methodology and science of bioidentical hormone replacement therapy for prevention and wellness. Her career, however, started in a very different area of medicine. At the age of 28, Dr. Erika was the Medical Director of a major trauma centre in New York City. Five years later she went into the private practice of internal medicine and developed a highly successful practice for the following fifteen years affiliated with three hospitals and on the board of a New York medical school. What lead this well- respected, classically- trained physician to seek
a new approach to patient management? “I didn’t like the style of medicine I was practicing… everything was disease focused… I was waiting for people to get sick while doing testing and sending them home to wait for something to go wrong”, was her reply.
Dr. Erika’s career path is as unique as the life path that brought her to her current position. Born in Bucharest, Romania, she graduated from an American high school in Rome, then off to the “Big Apple” to attend college and medical school in New York City. Graduating from medical school in 1975, Dr. Erika’s internship, residency and fellowship focused on internal medicine and critical care, all disease centered practices. Twenty years into the practice of this conventional type of medicine, Dr. Erika found herself asking more questions than the system had answers for… By the early 1990’s, her interest in prevention was firmly established.
Dr. Erika researched physicians in New York and California that were engaged in the practice of integrative medicine and found few credible resources and no formal training available in the area. Dr.Erika found herself spending countless hours researching available data and training in the practices of integrative medicine. Her role today as a leading educator in prevention and bioidentical hormones for clinical practitioners stems from Dr. Erika’s personal frustration with the lack of quality education and training in areas considered outside conventional medicine.
Erika has written a highly successful series of books aimed at educating the general public on principles of wellness and disease prevention. Her first book, entitled Natural Energy (From Tired to Terrific in 10 Days) (1999) reviewed and highlighted the importance of CoEnzyme Q10 and L- carnitine in fighting fatigue. We at IHP are in total agreement that these two agents are crucial in maintaining healthy mitochondrial function, an area receiving more and more attention as the focus of care shifts to health maintenance rather that disease treatment.
Dr. Erika attributes tremendous importance to hormone balance as a means of preventing chronic diseases in the 21st century. “Running out of hormones is unhealthy. Using human identical hormones as replacement opens the door to true disease prevention. Prevention strategies are likely to fail if people feel drained, exhausted, depressed, and generally unwell. Using human identical hormones to create much needed hormone balance leads to improved mood, energy, and general well being allowing a person to be able to address the multiple lifestyle factors (diet, exercise, sleep) responsible for disease prevention”.
Dr. Erika’s second book was published in 2002, and we feel it important to point out it was published three months before the results of the Women’s Health Initiative study demonstrated the use of synthetic (pregnant horse urine) estrogen as a source of higher risk for heart disease, strokes and cancer. In her book, The Hormone Solution, Dr. Erika raised concerns about synthetic hormone replacement therapies (estrogen derived from pregnant mare’s urine), while highlighting the advantages of human identical hormone therapy (also known as bioidentical or natural hormones) and discussed the impact on overall health when individualized hormone therapies are provided for each patient. In 2004 Dr Erika authored her next book “The 30 Day Natural Hormone Plan”, reinforcing the importance of hormone health and simultaneously providing useful tools for implementing health sustaining plans for diet, exercise, relationships, and sleep. In 2007 Erika published Hormone Solution for your Daughter, which showcased the role of hormones in balancing the health of teenagers, and reviewed important skills for parenting, diet, exercise, relationships and sleep.
Treatment with bioidentical hormones has witnessed various methods of usage. Dr. Erika applies a concept of therapy we found incredibly unique; unlike the conventional approach of “treat to a target blood level”, Dr.Erika adopts the approach of “treat until the person feels well and then assess blood levels. Those are then the target blood levels for the individual in question”. It is an approach that may result in “higher than typically used in compounding ” levels of hormones being prescribed, yet is an approach that is truly individualized, and inherently identifies each patient’s particular optimum hormone levels. The approach may be applied to a broad array of treatment plans, not isolated to the application of bioidentical hormones.
“The book is intended for the public, yet delivers an important message to physicians as well; physicians need to change their attitudes. They have to become patient partners and serve the patient rather than their own egos. The book teaches patients to take responsibility for their own care, to trust themselves and not allow others to dictate their path to wellness. Ultimately, there is no reason to be sick!”
Dr. Erika has emerged as a leading authority in the education of healthcare providers. In 2007, along with a few other key opinion leaders in the field of prevention she founded the not for profit organization Bioidentical Hormone Initiative www.bhionline.org www.bioidenticalhormoneinitiative.org Through this organization and its affiliation with a New York medical school, Dr. Erika is able to offer practitioners clinical in office and seminar format training on the use of bioidentical hormone therapy. The seminars Dr. Erika teaches are not just about how to use bioidentical hormones in your practice; they are powerful examples of patient advocacy, and a tremendous and effective tool in bridging the gap between conventional and integrative healthcare providers. The seminars attract practitioners from all disciplines leading to successful much needed inter-professional relationships that result in truly integrative care for the patients and mutual respect and information sharing.
Dr. Erika has a new book in the works. The book focuses on helping patients take responsibility for their own healthcare by giving them the tools and confidence necessary to avoid bad medical decisions. “The book is intended for the public, yet delivers an important message to physicians as well; physicians need to change their attitudes. They have to become patient partners and serve the patient rather than their own egos. The book teaches patients to take responsibility for their own care, to trust themselves and not allow others to dictate their path to wellness. Ultimately, there is no reason to be sick!” she affirms.
IHP is grateful to Dr. Schwartz for taking the time to allow us to showcase her work to you. Dr. Erika is an incredible, energized, brilliant physician whose presence motivates you to ask yourself “what else can I do to help others”… Her parting words for me seem an ideal way to end this story: “you are only as good as the last past patient you helped make well”.
N-acetyl-cysteine A Versatile Intervention
successful completion of the questions at the end of this paper has been approved for continuing education by the bddt-n; 1.0 credit nutritional medicine and by the cnpbc; one ce hour.
Colin MacLeod, ND
Kinesis Health Associates
164-166 Ochterloney St.
Dartmouth, Nova Scotia, Canada
NAC at high oral or intravenous doses is the gold standard treatment for acetaminophen toxicity. NAC antidotes acetaminophen poisoning by repleting glutathione stores and as a result mitigates liver damage. NAC exerts its therapeutic action through glutathione repletion, as well as glutamate modulation and mucous thinning in treating other health conditions. Recently data has surfaced supporting NAC as an effective treatment for a variety of health conditions, including non-acetaminophen-induced acute liver failure, COPD, flu prevention, idiopathic pulmonary fibrosis, acute renal failure due to contrast media, schizophrenia, bipolar disorder, autism, grooming disorders, obsessive compulsive disorder, addiction and angina. Adverse effects of NAC by the oral route are mild at low doses and exclusively gastrointestinal, while IV NAC can lead to anaphylactic reactions, including pruritus, rash, tachycardia, hypotension, bronchospasm and syncope. Care should be taken with concomitant treatment with nitrates and NAC, due to NAC’s potentiating effect on nitrates. It is evident from the published data that NAC is a safe and valuable clinical tool, useful for treating a wide variety of health conditions. 
Introduction
Although widely accepted as the primary antidote to acetaminophen overdose and as a mucolytic agent, N-acetyl-cysteine (NAC) has a variety of clinical applications, some of which have only recently come to light. Cysteine is a non-essential amino acid that is found mostly in animal products. NAC is essentially cysteine with an acetyl group attached. This acetyl group limits digestive degradation and promotes absorption. NAC powder has a white colour and dissolves clear to slightly purple in water. NAC has a sulphur-like odour and unpleasant taste which makes high oral doses challenging to administer.
Standard Dosing
NAC is commonly available without prescription in 500mg or 600mg capsules. NAC is also available at 200mg/ml solution for oral and intravenous use in the treatment of acetaminophen overdose and as a mucolytic in pulmonary conditions with excessive mucous. Dosing of NAC is condition specific and has been best established in the treatment of acetaminophen toxicity.
In the treatment of acetaminophen toxicity in adults, the oral dose of NAC is 1,330 mg/kg (93,100 mg in 70 kg individual) in 18 doses spread over three days: a loading dose of 140 mg/kg followed by 17 doses of 70 mg/kg every four hours (Prescott 2005). The intravenous dose is 300 mg/kg (21,000 mg in 70 kg individual) divided into three separate IV bag drips over 21hours: a loading dose of 150mg/kgin5%dextroseis given over a 15-minute period, followed by 50 mg/kg in four hours and 100 mg/kg in 16 hours, for a total dose of 300 mg/kg in 20 and one quarter hours (Prescott 2005). The intravenous route is used in Canada, Europe and Australia, whereas the oral route is used in the United States.
Liver damage is prevented most effectively when treatment is administered orally (Smilkstein 1988) or intravenously (Whyte 2007) within eight hours of the acetaminophen overdose. However, treatment should not be denied up to 24 hours after poisoning as there is still life-saving potential (Prescott 2005). Most conditions experimentally treated with NAC have used considerably smaller doses than that used for acetaminophen toxicity. Limited data exists on the efficacy and safety of nebulized NAC.
Pharmacokinetics
When 600mg of NAC was administered orally to healthy human volunteers, 6.4-10% was absorbed into the bloodstream intact (Borgström 1986). In an animal model, it has been found that low oral bio-availability is due, in part, to de-acetylation and conversion to other sulphur-containing compounds within the digestive tract, but that a significant amount of NAC reaches the blood as other-sulphur containing compounds (Cotgreave 1987).
The half-life of NAC in healthy adults has been reported differently by different investigators, including 2.27 hours (Borgström 1986) and 5.7 hours (Prescott 1989) for the intravenous route, and 3.7 hours for the oral route (Nolin 2010). The newborn half-life of NAC has been reported to be between 7.8 and 15.2 hours when administered intravenously, but depends largely on weight and gestational age (Ahola 1999). The half-life of NAC is significantly longer when given orally to patients with end-stage renal disease (Nolin 2010), or chronic liver disease (Jones 1997).
Pregnant women with acetaminophen poisoning should be treated with a standard high-dose oral NAC protocol due to its life-saving potential for the mother and fetus (Riggs 1989). If NAC is given to a pregnant woman in high oral or intravenous doses placental transfer will occur (Horowitz 1997). Minimal data exists on the safety and efficacy of IV NAC in pregnant women for treatment of acetaminophen toxicity.
Mechanism of Action
NAC is believed to act through three major mechanisms: providing glutamate for neurotransmission, providing cysteine for the synthesis of glutathione, and cleaving disulphide bonds present in mucous.
Glutathione, the most ubiquitous endogenous antioxidant is composed of three major amino acids: glutamate, glycine and cysteine. Of its three components, cysteine supply is the rate limiting step in glutathione production (Lu 2012, Radtke 2012). Some of the therapeutic effects of NAC have been attributed to its ability to provide glutathione, including in pulmonary conditions and hepatic injury. NAC does not seem to increase plasma glutathione in healthy human controls but will increase plasma glutathione in humans who have depressed glutathione stores from oral acetaminophen dosing (Burgunder 1989).
The activity of NAC in psychiatric conditions has been postulated to be due to its ability to provide glutamate for neurotransmission and in restoring brain glutathione. Altered glutamate status has been reported to be an important factor in psychiatric conditions with an addictive or compulsive component (Dean 2011). NAC’s structure also includes a free sulphhydryl (-SH) group which is thought to act on the relatively weak disulphide bonds within mucous to break it into smaller, less viscous segments which can be more easily cleared.
Clinical Indications
Acetaminophen Toxicity
NAC has been used for over 30 years as an antidote to acetaminophen toxicity (Scalley 1978). NAC’s remarkable efficacy as an acetaminophen antidote can be seen in observational evidence where a delay of NAC administration of even a few hours can be the difference between full recovery and liver failure (Smilkstein 1988). NAC’s ability to replete glutathione is the mechanism by which it antidotes acetaminophen toxicity.
Non-Acetaminophen-Induced Acute Hepatic Failure NAC has proven to be helpful in increasing survival in hepatitis- and drug-induced liver failure at high oral (Mumtaz 2009, Sotelo 2009) and intravenous (Kortsalioudaki 2008, Lee 2009) doses similar to those used in acetaminophen toxicity. Kortsalioudaki found that intravenous NAC administered to children with non-acetominophen acute liver failure resulted in shorter length of hospital stay, higher incidence ofnativeliverrecoverywithouttransplantation,and better survival after transplantation (2008). Much smalleroraldosesof600mg/dayhavebeenassociated with no change in duration of hospitalization compared to controls (Gunduz 2003).
High-dose NAC is a promising treatment in drug- or hepatitis-induced acute hepatic failure. Although no firm dosing schedule can be identified, an oral or intravenous protocol similar to that used in acetaminophen toxicity should be considered due to the increased survival evidenced in four human trials (Mumtaz 2009, Sotelo 2009, Kortsalioudaki 2008, Gunduz2003).
Chronic Obstructive Pulmonary Disease
Several studies have been performed on the effectiveness of NAC for Chronic Obstructive Pulmonary Disease (COPD) using oral doses of 600- 1200mg/day. Some studies (Hansen 1994, Pela 1999, Stav 2009, Völkl 1992, Zuin 2005) though not all (Black 2004, Decramer 2005, Lukas 2005, Schermer 2009) found that use of NAC in addition to conventional treatments is more effective in the management of COPD related symptoms compared to conventional treatment alone. In addition, several trials report improvements in specific measurements of lung function, including FEV1, inspiratory capacity (IC), and functional residual capacity (FRC) (Decramer 2005, Pela 1999, Stav 2009).
It should be noted that although many of the trials utilize FEV1 as the primary measure of effectiveness, failure to show benefit on this outcome should not be strictly interpreted as a failure of treatment success. Stav et al point out that although FEV1 is used to classify disease severity and is a good predictor of COPD mortality, it is a “poor predictor of clinical symptoms, exercise tolerance, and response to bronchodilators in COPD” (2009). Furthermore, COPD is characterized by “disease changes at the level of small airways, which is not usually expressed by the measurement of FEV1… IC [inspiratory capacity], however, is a lung volume measure that has been found to correlate well with patient dyspnea and exercise tolerance (2009). For instance, reduced IC during exercise correlated well with dynamic hyperinflation (Stav 2009). Some trials have shown small but significant improvements in FEV1 associated with NAC treatment (Pela 1999, Völkl 1992, Zuin 2005), while others have not (Decramer 2005).
The BRONCUS trial was a large (N=523), high quality trial showing no effect of 600mg NAC compared with placebo on the maximal forced expiratory volume in one second (FEV1) and prevention of COPD exacerbations (primary outcomes) when given over a three year period (Decramer 2005). However, a significant reduction in the functional residual capacity (FRC), the volume remaining in the lungs at the end of passive expiration, was seen in the NAC group on secondary analysis: FRC decreased from 4·46L to 4·09L in patients assigned to NAC, p<0.0001 (Decramer 2005). Other trials have also noted improvements in other measures of lung function associated with air trapping, including IC and FVC (Stav 2009). Considering its negligible adverse effects, low cost, and data showing benefit on measures of lung function associated with air trapping, NAC deserves to be considered as an adjunctive treatment option in COPD at oral doses of 600-1200mg/day.
Flu Prevention
One large (N=262), high-quality trial has been performed on the effect of NAC on influenza. Elderly patients were treated prophylactically for six months during flu season with oral NAC at 600mg twice daily or placebo. Although influenza infection rates were not different between treatment and placebo groups, influenza-like episodes, symptom severity and length of time confined to bed in the NAC group were significantly lower. The NAC group was also found to have a significant immune stimulation as compared to baseline and compared to the placebo group, as measured through anergy (De Flora 1997).
Idiopathic Pulmonary Fibrosis
Idiopathic Pulmonary Fibrosis (IPF) is a condition of unknown etiology which is characterized by scarring of the lungs and most commonly affects people 50-70 years of age. One randomized, double-blind placebo- controlled trial (N=182) administered NAC orally at 600mg three times daily as an add-on to standard treatment in patients with IPF. The NAC group had significantly slowed deterioration of lung vital capacity (VC) and diffusion capacity compared to the placebo group (Demedts 2005). Conversely, data on nebulized NAC suggests that it does not improve outcomes in IPF (Bando 2010, Tomioka 2005) and may even lead to acute exacerbations (Bando 2010). On considering these trials, oral NAC at 600mg three times daily should be considered as an add-on to standard medical treatment for IPF. (Tramadol)
Renal Protection from X-ray Contrast Media
Contrast media used during radiographic examination can cause nephropathy and acute renal failure. Risk factors for contrast-induced nephropathy include compromised kidney function, diabetes and reduced intravascular volume. Numerous clinical trials have been conducted over the past decade on prevention of acute renal failure with most using oral doses of 600mg NAC twice daily on the day before and the day of contrast media administration. As a whole the results have been mixed with some studies showing benefit from NAC (Briguori 2011, Koc 2012, Sar 2010) and others showing no effect (Anderson 2011, Gurm 2012, Jaffery 2012).
As is the case with COPD, the evidence is equivocal, but the intervention has negligible adverse effects and the cost is low. With this in mind, oral NAC at 600mg twice daily on the day before and the day of contrast media administration should be considered for patients with compromised kidney function as an add-on to standard treatment for prevention of acute kidney failure.
Nitrate-treated Angina
Sulfhydryls seem to be fundamental to nitrate- induced vasodilation (Mehra 1994). Data also suggests that in angina patients receiving nitrates, NAC given orally may lead to a significant decrease in ischemic effects as assessed by ECG (Boesgaard 1991), myocardial infarction rates (Ardissino 1997), angina symptoms and death rates (Ardissino 1997). Oral dosages in this setting have ranged from 1800- 2400 mg per day in divided doses (Ardissino 1997, Boesgaard 1991). NAC given intravenously may lead to a decreased tolerance to nitrates (Boesgaard 1992, Pizzulli 1997) and a decreased rate of myocardial infarction (Horowitz 1988) in nitrate- treated angina patients. However, not all trials have shown that NAC is of benefit, when given orally (Hogan 1990) or intravenously (Parker 1987).
Considerable care must be taken when giving NAC to nitrate-treated patients orally (Ardissino 1997) or intravenously (Horowitz 1988) due to NACs ability to potentiate associated hypotension and headaches. If prescribed, oral NAC should be started at a low dose (600mg three (Ardissino 1997) or four times daily (Boesgaard 1991)) with careful monitoring of blood pressure and headache occurrence. No clear recommendation can be given for intravenous dosing.
Schizophrenia
Three trials have examined NAC in patients with schizophrenia (Berk 2008, Carmeli 2012, Lavoie 2008). One double-blind, placebo-controlled trial (N= 140) using oral NAC at 1,000mg twice daily showed statistically significant improvements in those treated with NAC across several standardized schizophrenia measurement scales, and akathisia (restless leg syndrome) also improved significantly compared to placebo (Berk 2008). The mechanism by which NAC treats schizophrenia is thought to be through brain glutamate/ NMDA receptor modulation and reduction of oxidative stress (Dean 2011).
Lavoie et al conducted a randomized, double- blind, cross-over trial comparing 2g NAC to placebo for a period of 60 days to investigate N-methyl-D-aspartate (NMDA) receptor function, characteristically low in schizophrenia (2008). Mismatch negativity (MMN) is an auditory evoked potential (AEP) component related to NMDA receptor function that is impaired in patients with schizophrenia (Lavoie 2008). In this study, treatment with NAC significantly improved MMN generation compared with placebo (p=0.025), suggesting improved NMDA receptor activity, but the study was not continued for long enough to observe changes in clinical severity as reported by the same group in Berk 2008 (above) (Lavoie 2008).
Lastly, a recent proof-of-concept study found that NAC was able to significantly increase multivariate phase synchronization (MPS) on EEG testing compared to placebo over the left parieto-temporal, the right temporal, and the bilateral prefrontal regions in patients with schizophrenia (Carmeli 2012). Authors suggested that this may be an early marker of clinically important treatment effects.
Bipolar Disorder
Two small double-blind, placebo-controlled trials (N=75, N=17) in patients diagnosed with bipolar disorder showed very encouraging improvement in measures of symptom severity, functioning and quality of life with oral NAC at 1,000mg twice daily as an add-on to conventional therapy (Berk 2008, Magalhães 2011A). Improvements were evident by eight weeks (Beck 2008). In a sub-analysis of the study by Berk 2008, six of seven participants with bipolar II in the NAC group experienced a full remission of depressive and manic symptoms, compared to only two of seven in the placebo group (p=0.031) (Magalhaes 2011B). In an analysis of the effect of adjunctive NAC treatment on depressive symptoms in bipolar patients, Magalhaes et al reported “very large effect sizes in favor of NAC … for depressive symptoms and functional outcomes …. Eight of the ten participants on NAC had a treatment response at endpoint; the same was true for only one of the seven participants allocated to placebo” (2011A). A report of a larger cohort (N=149) from the two-month open label phase following an RCT showed similar results, which were described by the authors as a “robust decrement in depression scores” associated with NAC treatment (Berk 2011). Adverse effects were non-significant in all three trials.
Autism
A small (N=33), 12-week double-blind randomized placebo-controlled trial treated autisticpatients(3.2- 10.7 years of age) with oral NAC in an escalating dose schedule reaching a maximum of 900mg three times daily. Compared with the placebo group, the NAC group had sign if i can’t improvements on the Aberrant Behaviour Checklist irritability subscale. Adverse effects were mild and exclusively gastrointestinal (Hardan 2012).
Grooming Disorders
There are several case reports and small trials of oral NAC in the treatment of addiction, compulsive disorder and excessive grooming disorders. It has been suggested that the mechanism of action may involve changes to glutamate levels in the brain (Dean 2011).
One small (N=50) randomized, double-blind trial compared NAC 1,200mg or 2,400mg per day to placebo in the treatment of trichotillomania (compulsive hair pulling). Significant improvements were noticed after nine weeks of treatment in hair pulling with 56% of participants in a NAC treated group reporting “much or very much improved” whereas 16% in placebo group reported the same level of improvement (Grant 2009). Another two cases of trichotillomania showed encouraging improvement when treated with oral NAC at 1,200 mg/day for three and six months, respectively (Rodrigues-Barata 2012).
In the case of nail biting, three patients showedcomplete abstinence of nail-biting after oral NAC at 1,000mg bid for four to seven months (Berk 2009). Encouraging improvement was also seen in one case of trichotillomania, one case of nail biting with trichotillomania and one case of skin picking when treated with oral NAC at 1,800-2,400mg/day (Odlaug 2007).
Obsessive Compulsive Disorder
A small (N=36) randomized, double-blind trial compared 600-2,400mg/day NAC and placebo orally for 12 weeks in patients with obsessive compulsive disorder who were refractory to SSRI treatment. The patients in the NAC group had a 52.6% rate of “full response”, while the patients taking placebo had a 15% rate of “full response”. Full response was defined as a 35% or greater reduction on the Yale- Brown Obsessive Compulsive Scale. The Yale-Brown Obsessive Compulsive Scale (Y-BOCS) score was also significantly improved in the NAC group as a whole (p<0.001) (Afshar 2012).
Addiction
Two small (N=13) (N=15) placebo-controlled trials (LaRowe 2006, 2007) and one small (N=23) open- label trial (Mardikian 2006) have shown significant reductions in cocaine craving and usage with oral doses of NAC at 1200-3600mg/day.
Another small (N=116), eight-week double- blind randomized placebo-controlled trial targeted promoting cannabis abstinence in adolescents. Participants were given 1200mg NAC twice daily or placebo orally, along with brief weekly counselling sessions and a small cash-based reward system. When adjusted for cannabis use at baseline, the NAC group had significantly higher odds than the placebo group of negative urine cannabinoid tests during treatment, OR 2.4 (95% CI 1.1-5.2) (Gray 2012).
An open-label trial followed by a double blind placebo-controlled trial investigated the effect of oral NAC at 1,800mg/day on pathological gambling. Significant improvements were seen in the Yale-Brown Obsessive Compulsive Scale Modified for Pathological Gambling in the NAC group compared to the placebo group in both sections of the study (Grant 2007).
Although individually these case studies and clinical trials modestly support NAC use, the totality of evidence supports oral NAC at 1,200-3,600mg/day as a viable treatment option in addiction (specifically cocaine, cannabis and gambling), excessive grooming behaviours (specifically trichotillomania, skin picking and nail biting) and obsessive compulsive disorder.
Adverse Effects and Toxicity
A safety analysis of high-dose NAC examined its effects on 503 patients with acetaminophen toxicity. The oral route caused nausea and vomiting in 23% of patients and anaphylactic reactions in 2%. The IV route caused anaphylactic reactions in 9% of patients and nausea and vomiting in 6% of patients. The anaphylactic reactions were, in decreasing order of occurrence, pruritus, tachycardia, flushing, urticaria, non-urticarial rash, hypotension, bronchospasm and syncope (Bebarta 2010).
One retrospective study of 187 patients with acetaminophen toxicity treated with intravenous NAC found only seven adverse reactions, six being cutaneous and one being life-threatening, but not clearly caused by NAC (Kao 2003). Massive accidental intravenous overdoses have been reported to cause kidney failure (Mullins 2011), severe brain injury (Heard 2011) and death (Bailey 2004, Elms 2011).
Oral and intravenous NAC are considered to be of equal efficacy in treating acetaminophen poisoning (Kanter 2006). The intravenous route is not recommended for patients prone to anaphylaxis or at high risk for bronchospasm, such as asthmatics. The oral route is not recommended for patients with nausea or vomiting severe enough to render oral treatment ineffective(Kanter2006).Oraldosesof600mgtwice dailywereassociatedwithaninsignificantadverseeffect profilecomparedtoplaceboina262persontrial(De Flora 1997).
Conclusion NAC, the standard treatment for acetaminophen toxicity, has several relatively unknown clinical uses, including treatment of acute liver failure, COPD, flu prevention, idiopathic pulmonary fibrosis, acute renal failure due to contrast media, schizophrenia, bipolar disorder, autism, grooming disorders, obsessive compulsive disorder, addiction and angina. Care should be taken to avoid adverse effects associated with nitrate potentiation in angina treatment.
References
Afshar H, Roohafza H, Mohammad-Beigi H, et. al. N-Acetylcysteine Add-On Treatment in Refractory Obsessive-Compulsive Disorder: A Randomized, Double-Blind, Placebo-Controlled Trial. J Clin Psychopharmacol. 2012 Dec;32(6):797-803.
Ahola T, Fellman V, Laaksonen R, et. al. Pharmacokinetics of intravenous N-acetylcysteine in pre-term new-born infants. Eur J Clin Pharmacol. 1999 Nov;55(9):645-50.
Anderson S, Park Z, Patel R. Intravenous N-acetylcysteine in the prevention of contrast media-induced nephropathy. Ann Pharmacother. 2011 Jan;45(1):101-7.
Ardissino D, Merlini PA, Savonitto S, et. al. Effect of transdermal nitroglycerin or N-acetylcysteine, or both, in the long-term treatment of unstable angina pectoris. J Am Coll Cardiol. 1997 Apr;29(5):941-7.
Bailey B, Blais R, Letarte A. Status epilepticus after a massive intravenous N-acetylcysteine overdose leading to intracranial hypertension and death. Ann Emerg Med. 2004 Oct;44(4):401-6.
Bando M, Hosono T, Mato N, et. al. Long-term efficacy of inhaled N-acetylcysteine in patients with idiopathic pulmonary fibrosis. Intern Med. 2010;49(21):2289-96.
Bebarta VS, Kao L, Froberg B, et. al. A multicenter comparison of the safety of oral versus intravenous acetylcysteine for treatment of acetaminophen overdose. Clin Toxicol (Phila). 2010 Jun;48(5):424-30.
Berk M, Copolov DL, Dean O, et. al. N-acetyl cysteine for depressive symptoms in bipolar disorder–a double-blind randomized placebo- controlled trial. Biol Psychiatry. 2008 Sep 15;64(6):468-75.
Berk M, Jeavons S, Dean OM, et. al. Nail-biting stuff? The effect of N-acetyl cysteine on nail-biting. CNS Spectr. 2009 Jul;14(7):357-60.
Berk M, Dean O, Cotton SM, et. al.. The efficacy of N-acetylcysteine as an adjunctive treatment in bipolar depression: an open label trial. J Affect Disord. 2011 Dec;135(1-3):389-94.
Black PN, Morgan-Day A, McMillan TE, Poole PJ, Young RP. Randomised, controlled trial of N-acetylcysteine for treatment of acute exacerbations of chronic obstructive pulmonary disease [ISRCTN21676344]. BMC Pulm Med. 2004 Dec 6;4:13.
Boesgaard S, Aldershvile J, Pedersen F, Pietersen A, Madsen JK, Grande P. Continuous oral N-acetylcysteine treatment and development of nitrate tolerance in patients with stable angina pectoris. J Cardiovasc Pharmacol. 1991 Jun;17(6):889-93.
Boesgaard S, Aldershvile J, Poulsen HE. Preventive administration of intravenous N-acetylcysteine and development of tolerance to isosorbide dinitrate in patients with angina pectoris. Circulation. 1992 Jan;85(1):143-9.
Borgström L, Kågedal B, Paulsen O. Pharmacokinetics of N-acetylcysteine in man. Eur J Clin Pharmacol. 1986;31(2):217-22. Briguori C, Quintavalle C, De Micco F, Condorelli G. Nephrotoxicity of contrast media and protective effects of acetylcysteine. Arch Toxicol. 2011 Mar;85(3):165-73. Burgunder JM, Varriale A, Lauterburg BH. Effect of N-acetylcysteine on plasma cysteine and glutathione following paracetamol administration. Eur J Clin Pharmacol. 1989;36(2):127-31.
Carmeli C, Knyazeva MG, Cuénod M, Do KQ. Glutathione precursor N-acetyl-cysteine modulates EEG synchronization in schizophrenia patients: a double-blind, randomized, placebo-controlled trial. PLoS One. 2012;7(2):e29341.
Cotgreave IA, Berggren M, Jones TW, Dawson J, Moldéus P. Gastrointestinal metabolism of N-acetylcysteine in the rat, including an assay for sulfite in biological systems. Biopharm Drug Dispos. 1987 Jul-Aug;8(4):377-86.
Dean O, Giorlando F, Berk M. N-acetylcysteine in psychiatry: current therapeutic evidence and potential mechanisms of action. J Psychiatry Neurosci. 2011 Mar;36(2):78-86.
Decramer M, Rutten-van Mölken M, Dekhuijzen PN, et. al. Effects of N-acetylcysteine on outcomes in chronic obstructive pulmonary disease (Bronchitis Randomized on NAC Cost-Utility Study, BRONCUS): a randomised placebo-controlled trial. Lancet. 2005 Apr 30-May 6;365(9470):1552-60.
De Flora S, Grassi C, Carati L. Attenuation of influenza-like symptomatology and improvement of cell-mediated immunity with long-term N-acetylcysteine treatment. Eur Respir J. 1997 J u l ;10 ( 7 ):1535 – 41.
Demedts M, Behr J, Buhl R, et. al.. High-dose acetylcysteine in idiopathic pulmonary fibrosis. N Engl J Med. 2005 Nov 24;353(21):2229- 42.
Elms AR, Owen KP, Albertson TE, Sutter ME. Fatal myocardial infarction associated with intravenous N-acetylcysteine error. Int J Emerg Med. 2011 Aug 30;4(1):54.
Grant JE, Kim SW, Odlaug BL. N-acetyl cysteine, a glutamate- modulating agent, in the treatment of pathological gambling: a pilot study. Biol Psychiatry. 2007 Sep 15;62(6):652-7.
Grant JE, Odlaug BL, Kim SW. N-acetylcysteine, a glutamate modulator, in the treatment of trichotillomania: a double-blind, placebo-controlled study. Arch Gen Psychiatry. 2009 Jul;66(7):756-63.
Gray KM, Carpenter MJ, Baker NL, et. al. A double-blind randomized controlled trial of N-acetylcysteine in cannabis-dependent adolescents. Am J Psychiatry. 2012 Aug 1;169(8):805-12.
Gunduz H, Karabay O, Tamer A, Ozaras R, Mert A, Tabak OF. N-acetyl cysteine therapy in acute viral hepatitis. World J Gastroenterol. 2003 Dec;9(12):2698-700.
Gurm HS, Smith DE, Berwanger O, et. al. Contemporary use and effectiveness of N-acetylcysteine in preventing contrast-induced nephropathy among patients undergoing percutaneous coronary intervention. JACC Cardiovasc Interv. 2012 Jan;5(1):98-104. Hansen NC, Skriver A, Brorsen-Riis L, et al. Orally administered N-acetylcysteine may improve general well-being in patients with mild chronic bronchitis. Respir Med. 1994 Aug;88(7):531-5. Hardan AY, Fung LK, Libove RA, et. al. A randomized controlled pilot trial of oral N-acetylcysteine in children with autism. Biol Psychiatry. 2012 Jun 1;71(11):956-61. Heard K, Schaeffer TH. Massive acetylcysteine overdose associated with cerebral edema and seizures. Clin Toxicol (Phila). 2011 Jun;49(5):423-5.
Hogan JC, Lewis MJ, Henderson AH. Chronic administration of N-acetylcysteine fails to prevent nitrate tolerance in patients with stable angina pectoris. Br J Clin Pharmacol. 1990 Oct;30(4):573-7.
Horowitz RS, Dart RC, Jarvie DR, Bearer CF, Gupta U. Placental transfer of N-acetylcysteine following human maternal acetaminophen toxicity. J Toxicol Clin Toxicol. 1997;35(5):447-51.
Horowitz JD, Henry CA, Syrjanen ML, et. al. Nitroglycerine/N- acetylcysteine in the management of unstable angina pectoris. Eur Heart J. 1988 Jan;9 Suppl A:95-100.
Jaffery Z, Verma A, White CJ, et. al. A randomized trial of intravenous n-acetylcysteine to prevent contrast induced nephropathy in acute coronary syndromes. Catheter Cardiovasc Interv. 2012 May 1;79(6):921-6.
Jones AL, Jarvie DR, Simpson D, et. al. Pharmacokinetics of N-acetylcysteine are altered in patients with chronic liver disease. Aliment Pharmacol Ther. 1997 Aug;11(4):787-91.
Kanter MZ. Comparison of oral and i.v. acetylcysteine in the treatment of acetaminophen poisoning. Am J Health Syst Pharm. 2006 Oct 1;63(19):1821-7.
Kao LW, Kirk MA, Furbee RB, Mehta NH, Skinner JR, Brizendine EJ. What is the rate of adverse events after oral N-acetylcysteine administered by the intravenous route to patients with suspected acetaminophen poisoning? Ann Emerg Med. 2003 Dec;42(6):741-50.
Koc F, Ozdemir K, Kaya MG, et. al. Intravenous N-acetylcysteine plus high-dose hydration versus high-dose hydration and standard hydration for the prevention of contrast-induced nephropathy: CASIS- -a multicenter prospective controlled trial. Int J Cardiol. 2012 Mar 22;155(3):418-23.
Kortsalioudaki C, Taylor RM, Cheeseman P, et. al. Safety and efficacy of N-acetylcysteine in children with non-acetaminophen-induced acute liver failure. Liver Transpl. 2008 Jan;14(1):25-30.
Larowe SD, Mardikian P, Malcolm R, et. al. Safety and tolerability of N-acetylcysteine in cocaine-dependent individuals. Am J Addict. 2006 Jan-Feb;15(1):105-10.
LaRowe SD, Myrick H, Hedden S, et. al. Is cocaine desire reduced by N-acetylcysteine? Am J Psychiatry. 2007 Jul;164(7):1115-7.
Lavoie S, Murray MM, Deppen P, et. al. Glutathione precursor, N-acetyl-cysteine, improves mismatch negativity in schizophrenia patients. Neuropsychopharmacology. 2008 Aug;33(9):2187-99.
Lee W, Hynan L, Rossaro L, et. al. Intravenous N-acetylcysteine improves transplant-free survival in early stage non-acetaminophen acute liver failure. Gastroenterology. 2009 Sep;137(3):856-64, 864.e1.
Lu SC. Glutathione synthesis. Biochim Biophys Acta. 2012 Sep 17. Epub ahead of print.
Lukas R, Schärling B, Schultze-Werninghaus G, Gillissen A. [Antioxidant treatment with N-acetylcysteine and vitamin C in patients with chronic bronchitis]. Dtsch Med Wochenschr. 2005 Mar 18;130(11):563-7.
Magalhães PV, Dean OM, Bush AI, et. al. N-acetylcysteine for major depressive episodes in bipolar disorder. Rev Bras Psiquiatr. 2011 Dec;33(4):374-8.A
Magalhães PV, Dean OM, Bush AI, et. al. N-acetyl cysteine add-on treatment for bipolar II disorder: a subgroup analysis of a randomized placebo-controlled trial. J Affect Disord. 2011 Mar;129(1-3):317-20.B
Mardikian PN, LaRowe SD, Hedden S, Kalivas PW, Malcolm RJ. An open-label trial of N-acetylcysteine for the treatment of cocaine dependence: a pilot study. Prog Neuropsychopharmacol Biol Psychiatry. 2007 Mar 30;31(2):389-94.
Mehra A, Shotan A, Ostrzega E, et. al. Potentiation of isosorbide dinitrate effects with N-acetylcysteine in patients with chronic heart failure. Circulation. 1994 Jun;89(6):2595-600.
Mumtaz K, Azam Z, Hamid S, et. al. Role of N-acetylcysteine in adults with non-acetaminophen-induced acute liver failure in a center without the facility of liver transplantation. Hepatol Int. 2009 Aug 29.
Mullins ME, Vitkovitsky IV. Hemolysis and hemolytic uremic syndrome following five-fold N-acetylcysteine overdose. Clin Toxicol (Phila). 2011 Oct;49(8):755-9.
Nolin TD, Ouseph R, Himmelfarb J, et. al. Multiple-dose pharmacokinetics and pharmacodynamics of N-acetylcysteine in patients with end-stage renal disease. Clin J Am Soc Nephrol. 2010 Sep;5(9):1588-94.
Odlaug BL, Grant JE. N-acetyl cysteine in the treatment of grooming disorders. J Clin Psychopharmacol. 2007 Apr;27(2):227-9.
Parker JO, Farrell B, Lahey KA, Rose BF. Nitrate tolerance: the lack of effect of N-acetylcysteine. Circulation. 1987 Sep;76(3):572-6.
Pela R, Calcagni AM, Subiaco S, et. al. N-acetylcysteine reduces the exacerbation rate in patients with moderate to severe COPD. Respiration. 1999 Nov-Dec;66(6):495-500.
Pizzulli L, Hagendorff A, Zirbes M, et. al. N-acetylcysteine attenuates nitroglycerin tolerance in patients with angina pectoris and normal left ventricular function. Am J Cardiol. 1997 Jan 1;79(1):28-33.
Prescott L. Oral or intravenous N-acetylcysteine for acetaminophen poisoning? Ann Emerg Med. 2005 Apr;45(4):409-13.
Prescott LF, Donovan JW, Jarvie DR, Proudfoot AT. The disposition and kinetics of intravenous N-acetylcysteine in patients with paracetamol overdosage. Eur J Clin Pharmacol. 1989;37(5):501-6.
Radtke KK, Coles LD, Mishra U, et. al. Interaction of N-acetylcysteine and cysteine in human plasma. J Pharm Sci. 2012 Dec;101(12):4653-9.
Riggs BS, Bronstein AC, Kulig K, et. al. Acute acetaminophen overdose during pregnancy. Obstet Gynecol. 1989 Aug;74(2):247-53.
Rodrigues-Barata AR, Tosti A, Rodríguez-Pichardo A, Camacho- Martínez F. N-acetylcysteine in the Treatment of Trichotillomania. Int J Trichology. 2012 Jul;4(3):176-8.
Sar F, Saler T, Ecebay A, et. al. The efficacy of n-acetylcysteine in preventing contrast-induced nephropathy in type 2 diabetic patients without nephropathy. J Nephrol. 2010 Jul-Aug;23(4):478-82.
Scalley RD, Conner CS. Acetaminophen poisoning: a case report of the use of acetylcysteine. Am J Hosp Pharm. 1978 Aug;35(8):964-7.
Schermer T, Chavannes N, Dekhuijzen R, et. al. Fluticasone and N-acetylcysteine in primary care patients with COPD or chronic bronchitis. Respir Med. 2009 Apr;103(4):542-51.
Smilkstein MJ, Knapp GL, Kulig KW, Rumack BH. Efficacy of oral N-acetylcysteine in the treatment of acetaminophen overdose. Analysis of the national multicenter study (1976 to 1985). N Engl J Med. 1988 Dec 15;319(24):1557-62.
Sotelo N, de los Angeles Durazo M, Gonzalez A, Dhanakotti N. Early treatment with N-acetylcysteine in children with acute liver failure secondary to hepatitis A. Ann Hepatol. 2009 Oct-Dec;8(4):353-8.
Stav D, Raz M. Effect of N-acetylcysteine on air trapping in COPD: a randomized placebo-controlled study. Chest. 2009 Aug;136(2):381-6.
Tomioka H, Kuwata Y, Imanaka K, et. al. A pilot study of aerosolized N-acetylcysteine for idiopathic pulmonary fibrosis. Respirology. 2005 Sep;10(4):449-55.
Völkl KP, Schneider B. Therapy of respiratory tract diseases with N-acetylcysteine. An open therapeutic observation study of 2,512 patients. Fortschr Med. 1992 Jun 30;110(18):346-50.
Whyte IM, Francis B, Dawson AH. Safety and efficacy of intravenous N-acetylcysteine for acetaminophen overdose: analysis of the Hunter Area Toxicology Service (HATS) database. Curr Med Res Opin. 2007 Oct;23(10):2359-68.
Zuin R, Palamidese A, Negrin R, Catozzo L, Scarda A, Balbinot M. High-dose N-acetylcysteine in patients with exacerbations of chronic obstructive pulmonary disease. Clin Drug Investig. 2005;25(6):401-8.
Paradigm Health Group
Paradigm Health Group
Leaders in innovation
By: Angela MacNeil, MSc, ND
IHP is proud to introduce Paradigm Health Group, a 2000 square foot integrative medicine clinic in Calgary, Alberta. Dr. Jason Ahlan and Dr. Trevor Hoffman founded Paradigm after working together at the Integrative Medicine Institute of Canada (IMI), a collaboration amongst medical doctors, naturopathic doctors, dentists, and other healthcare professionals. When IMI closed its doors in 2007, these highly enthusiastic naturopathic doctors opened Paradigm with the vision that “by doing what we love, taking care of our patients one at a time, we can translate that to a positive experience for all.”
Dr. Ahlan and Dr. Hoffman recognized that successful individual naturopathic practices can be extremely difficult to establish. Believing that “there is power in numbers”, they opted to build a strong medical practice comprised of six naturopathic doctors with different backgrounds, strengths, and specialities, who are each held to the same level of excellence. Paradigm offers comprehensive care to patients that strongly consists of in-house cross-referrals. In an attempt to provide patients with the best care possible, Paradigm realizes that practitioners must sometimes acknowledge when another practitioner can provide superior care, especially considering the broad range of services offered, including prolotherapy, photoactivated platelet rich plasma injection therapies, nutrient IVs, chelation therapy, ozone therapy, ultraviolet blood irradiation, neural therapy, and mesotherapy. On the horizon, services offered will also include ultrasound guided injection therapies and adipose derived stem cell therapies. Numerous programs are offered that address stress, sleep, cancer, sports medicine and performance enhancement, pain management, autoimmunity, Firstline Therapy (FLT), allergies/sensitivities and related disorders, and detoxification. Detailed descriptions of these services and programs can be found on Paradigm’s website (http://www.paradigmhealth.ca). Paradigm strives to offer a wide variety of therapies in an effort to address numerous patient needs.
In addition to the naturopathic doctors, the clinic boasts an amazing network of 15 in-house/affiliated medical doctors with different specialities, and a broad referral network that includes chiropractors, massage therapists, physiotherapists, and holistic nutritionists. This network has contributed immensely to Paradigm’s success and the practitioners feel very fortunate to have these ties.
An effective networking strategy employed by Paradigm is the attendance of medical conferences; this serves to further the knowledge base within the clinic while simultaneously increasing contacts. In recent months alone, Paradigm practitioners have attended prestigious conferences world-wide, including the Restorative Medicine Conference, the Ironman Hawaii Sports Medicine Conference, the American College for Advancement in Medicine, and the American Academy of Orthopaedic Medicine. Since naturopathic medicine is a rapidly expanding field, staying on the cutting edge of research requires training from experts around the world. Admittedly, these conferences are costly to attend but the practitioners note that the education, improved experience, and expanded network they confer are priceless and have had tremendous impacts on the practice’s success.
In addition to a strong focus on continuing education, Paradigm is represented at the Canadian Institute of Natural and Integrative Medicine (CINIM), a registered charitable organization that conducts scientific research in complementary/alternative medicine (CAM). CINIM was the first of four Canadian institutions to become involved with the United States Consortium of Academic Health Centres for Integrative Medicine whose membership includes 51 highly esteemed academic medical centers and affiliate institutions, including Harvard, Yale, and the Mayo Clinic. Dr. Ahlan is one of only three naturopathic doctors in this collaboration, representing the University of Calgary Medical School. CINIM was developed in response to public demand for information about alternative therapies and access to these therapies.
Several of Paradigm’s practitioners are also involved with the Pure North S’Energy Foundation, a non-profit initiative that focuses on the delivery of innovative preventative and integrative health and wellness strategies. Pure North includes a comprehensive team of expert health and dental care professionals including physicians, naturopathic doctors and nurses that travel throughout Canada providing first-rate personalized services to populations who cannot afford integrative medicine. At last count, this funded program has taken the team to multiple sites internationally, seeing almost 15,000 patients from the firefighters in the Slave Lake disaster, to the Native American communities in Inuvik, to the off-shore oil sites in the North Sea of Scotland. Paradigm’s practitioners also belong to a wide array of boards and advisory councils that include Winsport Canada, Hockey Canada, Alpine Canada, Bobsleigh Canada, The Calgary Flames Hockey
Club, College of Naturopathic Doctors of Alberta (CNDA), and the Competancy Committee for Restricted Activities for the CNDA.
Paradigm offers patients a broad range of evidence-based diagnostic tests to verify clinical diagnoses and treatment of various health challenges. Diagnostic testing is offered from Rocky Mountain Analytical, Doctor’s Data, PharmaSan Labs, NeuroScience, Genova Diagnostics, Metametrix, and Meridian Valley. Paradigm practitioners consider themselves fortunate as the majority of their patients receive blood work through affiliated medical doctors as part of diagnostic work-ups. The clinic also offers a dispensary that features the best products from a variety of companies, including AOR, Douglas Labs, Metagenics, Priority One, Restorative Formulations, Bioclinic Naturals, BioMed, Genestra, Vitanica, Pascoe, and Heel.
 When Paradigm was established, a focus was placed on billable square feet so that all 10 treatment rooms are full at all times. As a business model, Paradigm focuses on maximizing efficiency, running approximately 100 intravenous treatments per week and seeing roughly 60 patients per day. Paradigm has also fully transitioned to electronic medical records (EMR) and despite the large financial investment and steep learning curve, this has opened many doors for the clinic. Although patient records are paperless, all new patients receive a binder that outlines the clinic’s comprehensive referral network, patient expectations, and success stories. Believing that marketing starts at home, Paradigm attempts to offer services of value, and this means taking care of each and every patient first. Consequently, word of mouth is Paradigm’s most powerful marketing tool.
When Paradigm was established, a focus was placed on billable square feet so that all 10 treatment rooms are full at all times. As a business model, Paradigm focuses on maximizing efficiency, running approximately 100 intravenous treatments per week and seeing roughly 60 patients per day. Paradigm has also fully transitioned to electronic medical records (EMR) and despite the large financial investment and steep learning curve, this has opened many doors for the clinic. Although patient records are paperless, all new patients receive a binder that outlines the clinic’s comprehensive referral network, patient expectations, and success stories. Believing that marketing starts at home, Paradigm attempts to offer services of value, and this means taking care of each and every patient first. Consequently, word of mouth is Paradigm’s most powerful marketing tool.
IHP is thoroughly impressed with the success Paradigm has achieved and is thankful that we were given a glimpse into this cutting edge practice. A nice way to conclude is reiterating a statement from Dr. Ahlan: “[you] must follow what you love, by doing that you will always be successful.”
Organika Health Products
Organika Health Products Inc.
BY SOPHIE KOHN
Tom Chin knows firsthand about the surprising power of natural health remedies. It was his own health scare that set him on the path to becoming CEO and President of Richmond, B.C. based Organika Health Products Inc.
“I started in a strange way,” explains Chin, who launched Organika 23 years ago. Before that, he was working in the finance industry when he developed a kidney stone. The proposed treatment involved an invasive surgical procedure. Curious about the alternatives, he contacted a friend with a natural health bent. The friend instructed Chin to grind up parsley and turmeric, add juice and water, and drink the concoction for three days. Chin took his friend’s advice and passed the stone in one day.
Organika’s inspiration
It was a revelatory moment for Chin. He was astonished by how effective this natural cure had been, and how such a simple remedy let him avoid a painful operation and undoubtedly long recovery.
His only question: Why aren’t more people using these types of solutions? Inspired, he set to work researching natural health products. He quickly found his answer – because natural products are prohibitively expensive for the average person, often carrying a 500 to 600 per cent markup.
Quality and affordability
“You’ve got to make these products available for all,” Chin says emphatically. “So everyone can have a healthy, good life.”
Chin formed Organika with two major goals – to make high quality products, and to make them affordable.
Today, Organika Health Products Inc. operates out of a 76,000 square-foot facility in Richmond. It employs 110 people, exports its products to 22 countries, has 480 NPN numbers, and is expanding and improving constantly. Organika manufactures nutritional supplements, digestion aids, anti-inflammatory remedies and herbal formulas. The bestselling products are enzymes and joint management care, which includes glucosamine and salmon oil capsules.
Organic certification
In June 2011, after a yearlong auditing process, Organika achieved its organic certification and acquired membership in the Pacific Agricultural Certification Society. Organika is now the only “certified organic facility” in Western Canada, meaning that all organic products remain pure during all production stages from raw materials to final product.
Updated facility
Recent improvements to the Richmond facility have supported Chin’s mission. The facility has added two High Performance Liquid Chromatography (HPLC) machines, which test products on three criteria: potency, quality and purity. These machines ensure that each Organika product satisfies the Health Canada guidelines for allowable limits of heavy metals and other foreign matter. Organika sources its raw materials from all over the world, which undergo eight testing methods before using. The company quarantines all raw materials until they pass Health Canada standards. After the product is manufactured, it’s quarantined once again.
New products
Organika has expanded its functional food line to include organic olive oil, and a 100 per cent Organic Greens superfood powder with 17 herbs and vegetables, one of the few greens powders that can boast only organic ingredients.
It also recently launched organic coconut palm sugar, available in bulk or stick packs. Organika started offering coconut palm sugar partly due to its sustainable attributes. A coconut tree can produce sugar with less water and space than it takes to produce cane sugar.
The company also introduced organic coconut vinegar with wild honey, which is selling particularly well. This vinegar is said to be 10 times more nutrient-dense than apple cider vinegar because the type of volcanic soil where the coconut palm grows is extremely rich in amino acids and minerals, which is transferred to the coconut. Similar to apple cider vinegar, it is also alkalizing, which is beneficial for cancer patients and others wanting to reduce food acidity levels.
Chin feels strongly that in order to engage consumers to buy natural health products, he must offer products that don’t disrupt people’s lifestyles.
“People are habitual,” Chin says. “If they’re going to consume sugar, they’re going to consume sugar.” Hence the convenient packet of coconut palm sugar; it’s in the format that people are used to, only it boasts a much lower glycemic index rating.
Evidence-based research
Throughout its 23-year journey, Organika has been fiercely committed to integrating sound science into its product development. This became especially true in 2004, when the Natural Health Product Directorate (NHPD) introduced stricter regulations. For Chin, that meant implementing a stronger focus on science. Today, a fifth of his employees are scientists in Quality Control and Quality Assurance. The team gathers information on the source of raw materials, dosage limits, efficacy, mode of action, safety, side effects, allergy response, contraindications, effects on long-term use, and interactions with other medications.
“We need strong scientists who are also dedicated to natural health,” he stresses. “In the past, we could get away with passionate people who weren’t scientists necessarily. But not anymore.”
Before Organika launches a new product, the product endures a rigorous process. First, scientific evidence must reveal that a new product is necessary. Second, demands of the market are assessed. Third, clinical data on the product safety is conducted. Finally, the collective data must prove that the product offers improvement or greater efficacy within what Chin describes as “the landscape of natural health.”
“‘Science’ is not a bad word,” Chin states. “It means that the product becomes as effective and precise as it can be. It means the product works.”
After 23 years, Chin still sounds committed and passionate about his vision for natural health. “We can be just as good as, and have just as high standards as, a pharmaceutical company,” he concludes.
At a Glance
Organika Health Products Inc.
Brands Organika Professional Line
Address13480 Verdun Place, Richmond, BC V6V 1V2
Phone1-800-663-8880
Website www.organika.com
Staff 110
Products 480
Top-selling product Organika Full Spectrum Plant Enzymes, Organika Pacific Salmon Oil, Organika GLS 500 (glucosamine sulfate)
Melanie Kay and Stephen Rosenhek, La Naturiste
New Beginnings for an Old, Established Chain
Le Naturiste’s new owners Melanie Kau and Stephen Rosenhek plan to revitalize the company
BY CAROL CRENNA
PHOTOGRAPHY BY VADIM DANIEL
When natural health icon Jean-Marc Brunet launched Le Naturiste in 1968, his vision for the future may well have included the goals of the newest owners of the 80-store chain. The enthusiastic and enterprising Montreal-based entrepreneurs Melanie Kau and Stephen Rosenhek, who acquired the company last year, have big plans. Kau, who formerly worked within a family-owned furniture retail business, states, “Donald Trump said, ‘If you are going to be thinking anyway, think big.’ We had great careers before this venture, so if we we’re going to begin a new one, we want to make it fantastic, and have fun doing it.”
Le Naturiste has built strong brand recognition among health-conscious Quebecers; as the first retailer to offer easy access to natural products, it’s established as a “community destination.” The new owners aren’t about to compromise that.
What are key considerations for transforming the 44-year-old chain, while at the same time not alienating existing customers?
REBRANDING PLANS
Since the name has tremendous equity “as a personality,” it won’t change. Kau says, “Customers tell us, ‘We came here as children with our mother and now we bring our kids.’ But we will rebrand to appeal to a wider base of people. We want to continue along the lines of the company’s founder who had a vision to better connect people to their health.”
To do this, the customer experience will be upgraded. They will be re-thinking interior design, signage and layout. “Customers are already waiting to see a new store prototype or flagship location. This will occur in 2013, whether it is a makeover of an existing store or a new store in a new market,” Rosenhek says.
Although all 80 stores are corporately owned, the new owners don’t want Le Naturiste to be pegged as a “big chain.”
Unique product mix
Rosenhek explains, “The amount of products that you wade through in U.S. chains is enormous, with 20 to 30 brands of each supplement or several feet of one brand merchandised together. Our job is to sift through and edit products to buy an intelligent mix, rather than becoming an impersonal environment where a customer asks about a product and the clerk says, ‘They are in aisle 17. Good luck.’ We must also rationalize why these two or three are best, targeted for specific customer’s needs.”
Local focus
Le Naturiste currently offers popular brands like Adrien Gagnon No, Genacol, Genuine Health, New Nordic, EAS and PVL, but the owners are also forging new relationships with smaller local producers within Quebec, and other parts of Canada. Rosenhek says, “We will seek out international specialty items, but for core products, why add environmental impact from something put on a ship or plane that isn’t necessary?”
They won’t give up bestselling Le Naturiste private label products sourced from Europe which have established the company as a leader. Rosenhek says many people rely upon its France-made ampoules (small glass vials that contain a one-dose liquid remedy) formulated for increased energy, weight loss, nutrient absorption and system cleansing.
Kau explains, “Europeans have advanced knowledge in plant extracts since their greater degree of acceptance by consumers creates demand for increased research. The ampoules we buy use a cold-pressed process that has not been replicated in North America.”
Combined experience
Kau, who describes the chain as “the perfect little jewel,” is responsible for anything that touches the customer: consultants, merchandising, in-store experience, branding and marketing. Rosenhek, formerly an accountant, works with research, finance, operations and buying teams.
Although the natural products sector is growing, government regulation and economic barriers frighten some newcomers away. But Rosenhek says they applaud Health Canada regulations because a large percentage of consumers still question the efficacy of natural products. “We were interested in buying Le Naturiste because it has a history in keeping abreast of these issues, and customers trust its quality. New regulations will help promote our company’s scientific basis, and improve the industry’s image.”
Behind the scenes
The company’s rigorous quality control department researches what claims are being made and whether those claims should be made; they consult on product design, and are part of the buying team to decide what makes it onto store shelves. Scientists in external and internal laboratories ensure efficacy, whether it is sampling products purchased from national brands or researching private label ingredients,” explains Rosenhek, who feels that shoppers are looking for greater transparency regarding ingredient sources.
Would Canadians rather shop a chain?
There aren’t many Canadian chains of this size, which has advantages. Customers can count on favourite products being available at every location. The company has the ability to create private label formulas, and have economies of scale for product pricing, unlike most independents. Its community stores also have a long history of personal relationships with clientele.
Rosenhek says, “As a customer, I used to leave my local pharmacy with no product in hand because I was confused. To take products appropriately and effectively, you need to receive proper questioning at the store. Sometimes product A and B work effectively together, but you won’t know it unless told.”
Specialized service
Plans include offering in-house professional practitioners’ services, in-store guest lectures and public health testing, comprehensive written information, and an interactive online experience. Kau, says, “Using technology, we are working on creative ways to enable customers to have live (real-time) health consultations with our corporate naturopath, and to get customers involved in helping to choose products.”
The future?
Rosenhek says, “We have twinkles in our eyes when asked about plans. We have lots of possibilities to expand through strategic alliances and acquisitions with retailers and suppliers. We have been presented opportunities abroad, too.”
The company will introduce professional-level products that consumers don’t usually have access to. European personal care items will be offered at a level not seen in the North American industry including high performance creams with completely natural ingredients. Environmental responsibility will increase. “People now buy products that are healthful but aren’t made with eco-friendly ingredients or manufacturing processes. We are looking at more traditional and more advanced methods that are highly sustainable,” concludes Rosenhek.
Stores may close and open
The chain’s 78 Quebec stores and two New Brunswick stores may change after the owners examine whether each location has evolved with their neighbourhood. “Customers may move away from certain areas; traffic patterns may have changed, and a store may not still be pertinent. Yet some locations have been points of reference for 20 to 30 years so we have to be careful,” Kau says.
Believing their business model can easily be exported, they plan to open new stores where a client base exists. “Ontario is looking pretty good,” Kau admits. Rosenhek qualifies, “As native Quebecers, unlike NBTY, we know this market. We have also worked outside the province so aren’t afraid of expansion, yet are aware that you can’t cookie-cutter stores, and must understand and adapt to customers in different regions.”
They’re not experienced supplement users
Kau feels she represents the majority of occasional shoppers who venture into a health store. “I wasn’t a big user of these products because I never understood how to interact with them. I would use one or two and then stop, not because I wasn’t interested, but because no one who acted like they understood my needs offered advice I could follow, so I could shop with a degree of confidence.”
Rosenhek was similar. “I’d spend more time in the supplement aisles than anywhere trying to find natural ways to deal with ailments I suffered from including migraines, and aches due to overdoing activities like running. Yet I only bought basic vitamins because I never got help. I am now able to deal naturally with health issues that have bothered me for years simply from learning more within this business in the past few months. I am like our customers and Melanie, very interested but never knew how to get to the next level.”
Medium Chain Triglycerides (MCT’s)
Medium Chain Triglycerides (MCT’s)
An alternative approach to the treatment of Alzheimer’s disease
If one takes a realistic view, modern medicine appears to have little of real significant to offer patients with Alzheimer’s disease. Treatments are symptomatic and do not significantly decelerate or prevent progression. Behavioural and psychiatric challenges of great concern to caregivers are attacked with powerful drugs not approved for these indications, and have serious and sometimes permanent adverse effects. A new approach appears desperately needed. While it has been known for decades that Alzheimer’s diseases is associated with impaired cerebral glucose metabolism, only recently has this observation begun to attract serious attention and now some even call Alzheimer’s disease Type 3 diabetes. This article addresses therapeutic approaches to cerebral hypoglycemia which directly or indirectly involve providing an alternative brain fuel to counteract decreased glucose metabolism. The alternative fuel consists of ketone bodies produced by ketogenesis in the liver. Clinical studies and limited randomized controlled trials have provided evidence for both the biological plausibility of this alternative approach and its efficacy and safety in the treatment of mild cognitive impairment and Alzheimer’s disease. In addition, what appear to be highly satisfactory oral precursors of the required ketone bodies are natural products readily available in many health food stores. This has generated considerable anecdotal evidence recently reviewed in a book by a respected physician that makes an important contribution to this field.
Introduction
Mainstream medicine treats early to moderate stages of Alzheimer’s disease (AD) with cholinesterase inhibitors (Aricept, Exelon, Razadyne and rarely Cognex). For moderate to severe stages, memantine (Ebixa), a drug that regulates the activity of glutamate, a chemical involved in learning and memory, is frequently employed. While aggressive combination therapies can produce small decreases in the frequently relentless increase of dementia or deteriation in daily living scores, progression remains (Atri 2008). “Currently available treatments for AD are symptomatic and do not decelerate or prevent the progression of the disease” (Herrmann 2011). There is considerable support for this view (Farrimond 2012, Galvin 2012, Lanctot 2009, Popp 2011).
In the US there are no drugs approved for dealing with behavior and psychiatric challenges associated with AD which cause significant problems for caregivers. However, the whole arsenal of antidepressants, anxiolytics and antipsychotics are widely used off-label. The problems and side effects associated with these drugs, both transient and permanent, can be significant (Breggin 2008). While these drugs modify symptoms, their adverse effect profile casts doubt on their true utility. Conventional therapies and the small changes they can affect provide false hope (Breggin 2008, Herrmann 2011). A new approach seems desperately needed.
The Brain Hypometabolism Hypothesis
There is growing interest in this hypothesis, initiated in 2005 by Suzanne de la Monte, that Alzheimer’s disease and type 2 diabetes have a number of pathologic features in common (de la Monte 2005). It was suggested that impaired glucose metabolism and impaired insulin action in the brain, which in part leads to mitochondrial dysfunction and decreased energy production, could be significantly responsible for the pathology observed as AD develops. This led to the suggestion that AD be called Type 3 diabetes. However, the notion that there is a connection between AD and decreased glucose metabolism (hypometabolism) in the brain goes back at least several decades (Hoyer 1970), and there is growing interest in this phenomenon (Yao 2011). Abnormalities in metabolism have been linked to brain insulin and insulin-like growth factor resistance with disruption of signalling pathways that regulate neuronal survival, energy production, gene expression and brain plasticity (de la Monte 2012b). The use of PET scans with a metabolic tracer have provided significant and extensive evidence supporting the presence of hypometabolism and the notion of Type 3 diabetes, and its mechanistic details are the subject of current research (de la Monte 2012a).
A recent review of brain hypometabolism (Cunnane 2011) points out the potential for a vicious cycle generated by brain damage associated with hypometabolism. The authors suggest that this cycle can be effectively broken by sustained improvement in brain metabolism, which can be achieved with ketone bodies acting as replacement fuel. Brain hypometabolism now appears to be an attractive target in AD therapy (Costantini 2008).
Ketone Bodies–A Replacement Brain Fuel
The three so-called ketone bodies are water soluble compounds (acetone, acetoacetate and beta-hydroxybutyrate) produced in the liver from fatty acids (ketogenesis). Ketone bodies bypass insulin controlled cellular entry and can serve as energy sources in the mitochondria of tissues including the brain. It was once accepted that upon weaning, the brain was limited solely to glucose for metabolic energy. This view has changed as the neuroprotective potential of ketone body administration has been shown for neurodegenerative conditions, epilepsy, hypoxia/ischemia and traumatic brain injury, and that there is an age-related therapeutic potential for ketone bodies as an alternative substrate (Prins 2008, White 2011). Also dietary ketosis has been shown to enhance memory in mild cognitive impairment, and there is evidence of protection against neuronal insults, an increase in metabolic efficiency relative to glucose, and in general such diets appear to some extent to mitigate neurodegenerative mechanisms (Krikorian 2012, Veech 2004). Perspective regarding the role of ketones in brain metabolism can be gained by considering that during starvation or very low carbohydrate intake, the liver turns to making ketones (Owen 1967). This defence mechanism evolved eons ago and was likely necessary for human survival.
Starvation is obviously not a practical therapeutic intervention. Neither is a severely ketogenic diet for many patients. Adherence to a classical ketogenic diet is a serious problem since about 90% of energy must come from fat. An obvious solution is an oral supplement or “medicinal food” that provides a source of the needed ketone bodies. Either natural or synthetic medium chain triglycerides (MCT) are obvious sources.
Oral Medium-Chain Triglycerides To Treat Alzheimer’s Disease
One of the prominent researchers in the field of brain hypometabolism, Samuel Henderson, was instrumental in the development of a MCT-based product to treat AD called AC-1202, which contained one MCT and could be taken as a dissolved powder. It was patented and tested in several trials and recently a multicenter randomized controlled trial reported (Costantini 2008, Henderson 2009). The compound rapidly elevated serum ketone bodies in patients with AD and resulted in significant improvement in cognitive scores in those with mild to moderate AD when compared to a placebo. MCT oil ingestion has also been found in a placebo controlled study to preserve brain function during hypoglycaemia in intensively treated patients with Type 1 diabetes (Page 2009). Some would consider these results convincing evidence concerning the biological plausibility of the hypo metabolic hypothesis.
AC-1202 is available only by prescription and approved by the FDA as a “medicinal food” called Axona. It contains only one ketone generating MCT. Coconut oil and MCT oil are natural sources generating several ketone bodies and offering flexible dosing throughout the day.
The leading advocate of MCT oil and coconut oil is Dr. Mary Newport, M.D., a paediatrician and director of a neonatal ICU at a Florida hospital. In 2008 her husband had moderately advanced AD that was progressing significantly. She first heard of the use of MCTs for AD in 2008 when she came across reference to Henderson’s patent, found AC-1202 contained a MCT and ascertained that MCTs were present in coconut oil which was available at natural food and health food stores. She decided there was nothing to lose by trying coconut oil. This was a turning point in her husband’s life as well as hers. Results were remarkably rapid, AD progression apparently stopped, and over the following three to four years many cognitive and behavioural deficits were significantly reduced and many lost functions recovered. In her words, she “got her husband back.” This is detailed in Dr. Newport’s recent book (Newport 2011).
One of the most fascinating aspects of Dr. Newport’s husband’s change was that initially he was totally unable to draw a clock, a standard test in mental assessment, and a few weeks after starting coconut oil his clock, while not perfect, was significantly improved. This is illustrated in Dr. Newport’s book and in the article on the internet (Newport 2008). In addition, sequential MRI studies revealed that the progression of brain atrophy had been totally halted. It appears that conventional, approved treatments have never produced such a result, or in a significantly sustained manner, any of the above results, especially when the patient has fairly severe AD. Today, most of the interest in this intervention is among lay persons who become aware of MCT oil and coconut oil while doing an internet search for natural AD treatments, through the social media, by word-of-mouth or from Dr. Newport’s book (Newport 2011).
In this book, Dr. Newport provides details and a summary of anecdotal caregiver reports (60 cases) she received after the do-it-yourself treatment became popular. Unfortunately, there is uncertainty as to dose, source of MCTs, adherence, severity of the disease and the quality of reportage from caregivers concerning changes. Nevertheless, 90% of those who tried MCTs were observed to be improved, as indicated by better scores on tests, improved clock drawing, better cognition, more alertness, brighter outlook, improved awareness, less foggy or hazy behaviour, being able again to recognize people or places, being less distractible, and having a better sense of direction.
MCT oil yields higher serum ketone body levels than coconut oil, but the decline over time is more rapid than seen with coconut oil. Thus coconut oil taken with MCT oil helps smooth out daily fluctuations with higher average levels and coconut oil may have some other benefits not provided by the commercial MCT oils since it contains other fatty acids (Newport 2011). This influenced Newport’s final protocol which involved using an approximately 1:1 mixture of coconut oil and MCT oil at a dose of three tablespoons with each meal and two at bedtime. To avoid diarrhea, the only known adverse effect of MCTs reported, dose escalation is sometimes needed. It is important to note that while attention was paid to a healthy diet, the macronutrient intake of Dr. Newport’s husband was not drastically altered. In fact, the use of MCTs has the advantage of not requiring significant diet changes and can even be used along with conventional treatments.
Ketogenic Diets
An alternative to ingesting MCTs is to use a ketogenic diet. Newport claims that the ketogenic diet is capable of producing up to 10 times or more ketone bodies than possible with the doses of MCTs commonly being used, and this becomes an attractive next step for those who do not respond to MCT therapy (Newport 2011). The ketogenic diet has been used to treat epilepsy and especially patients who failed to respond to antiepileptic drug therapy. The initial interest occurred in the early 1920s and then there was a long hiatus followed by recent renewed interest. There is now considerable evidence of efficacy in epileptic adults and children (Auvin 2012, Lambrechts 2012, Nam 2011). There has also been recent interest in ketogenic diets to treat Alzheimer’s disease (Kossoff 2012, McPherson 2012, Yao 2011). It has also been found that dietary ketosis enhances memory in mild cognitive impairment (Krikorian 2012). In addition, improvement in memory impaired adults has been correlated with orally induced increases in the serum level of the ketone body beta-hydroxybuterate (Reger 2004).
There are four recognized major ketogenic diets, the classical diet with 90% of calories from fat, the MCT diet, the modified Atkins diet and the low glycemic index diet. The MCT diet has been used since the 1970s and involves using coconut oil while limiting carbohydrates. The modified Atkins diet uses a very low carbohydrate induction diet (70% calories from fat, 24% from protein) as the permanent intervention, whereas the low glycemic diet has 45% from fat and 28% from protein with emphasis on carbohydrates selected for low glycemic index (Kossoff 2012).
Conclusions
What might be called the alternative fuel approach to treating AD has strong biological plausibility. Limited results from clinical and randomized controlled trials provide evidence of efficacy and safety. Treating hypometabolism with a prescription MCT (Axona) has FDA approval for the indication of cognitive impairment. When trial data is combined with a large body of anecdotal evidence, the picture emerges suggesting AD patients appear to obtain much more benefit from this alternative approach than from conventional medical therapy.
Individuals with AD frequently have a mixture of vascular and AD-type abnormalities and some have dementia associated with Lewy bodies. Hypothyroidism, micronutrient deficiencies, heavy metal overload, etc., may also contribute to the symptomatic presentation and the alternative fuel approach would not be expected to impact some of these abnormalities. There is also an issue with genetics. In the AC-1202 clinical studies patients not carrying the E4 variant of the apolipoprotein gene (about half of those with AD) showed the highest response (Costantini 2008, Henderson 2009). However, Dr Newport’s husband is ApoE4+ and showed remarkable regression (Newport 2011).
It is disappointing that there is not more clinical trial evidence, but therapies involving natural MCT oil and coconut oil are of no interest to pharmaceutical companies, and the very nature of this therapy invites the criticism of not being adequately evidence based and in fact simply another quack treatment. Those who believe in the future of alternative treatments must recognize the necessity of tolerating these hostile views because meeting the demands of so-called evidence-based medicine will be a slow and frustrating adventure, and individuals with AD and their caregivers may be actively discouraged from trying a therapy with the potential for greater benefit than appears attainable by any other means. Judging by what Dr. Newport reports, many AD caregivers will not settle for what mainstream medicine has to offer and seek to circumvent the hostile attitude of modern medicine. They simply adopt the approach that “What is there to loose? We can do this at home…”; a view directly applicable to MCT therapy. Everything needed is available online or at the local health food store, including a mixture of MCT and coconut oil in the proportions used by Dr. Newport (sold as a salad dressing). Finally, the use of MCTs in primary prevention as one ages remains to be studied but intuitively would appear to have merit.
The website of the company making Axona mentions caution when patients have a history of gastrointestinal problems, metabolic syndrome, uncontrolled diabetes and/or kidney disease. The extent to which these warnings apply to mixed MCT oil and coconut oil is unknown, but Dr. Newport reports only diarrhea. Guidance and monitoring by a physician is always highly desirable.
References
Atri, A, Shaughnessy, LW, Locascio, JJ and Growdon, JH. Longterm course and effectiveness of combination therapy in Alzheimer disease. Alzheimer Dis Assoc Disord. 2008; 22(3): 209-221.
Auvin, S. Should we routinely use modified Atkins diet instead of regular ketogenic diet to treat children with epilepsy? Seizure. 2012; 21(4): 237-240.
Breggin, P, 2008. Brain-Disabling Treatments in Psychiatry. Springer Publishing Co., New York.
Costantini, LC, Barr, LJ, Vogel, JL and Henderson,ST. Hypometabolism as a thera-peutic target in Alzheimer’s disease. BMC Neurosci 2008; 9 Suppl 2: S16.
Cunnane, S, Nugent, S, Roy, M, Courchesne-Loyer, A, Croteau, E, Tremblay, S, Castel-lano, A, Pifferi, F, Bocti, C, Paquet, N, Begdouri, H, Bentourkia, M, Turcotte, E, Allard, M, Barberger- Gateau, P, Fulop, T. and Rapoport, SI. Brain fuel metabolism, aging, and Alz-heimer’s disease. Nutrition 2011; 27(1): 3-20.
de la Monte, SM. Brain insulin resistance and deficiency as therapeutic targets in Alz-heimer’s disease. Curr Alzheimer Res 2012a; 9(1): 35-66.
de la Monte,SM. Metabolic derangements mediate cognitive impairment and Alzhei-mer’s disease: role of peripheral insulinresistance diseases. Panminerva Med 2012b; 54(3): 171-178.
de la Monte,SM and Wands, JR. Review of insulin and insulin-like growth factor ex-pression, signaling, and malfunction in the central nervous system: relevance to Alzhei-mer’s disease. J Alzheimers. Dis 2005; 7(1): 45-61.
Farrimond, LE, Roberts, E and McShane, R. Memantine and cholinesterase inhibitor combination therapy for Alzheimer’s disease: a systematic review. BMJ Open. 2012; 2(3).
Galvin, JE. Optimizing diagnosis and management in mild-tomoderate Alzheimer’s disease. Neurodegener. Dis Manag. 2012; 2(3): 291-304.
Henderson, ST, Vogel,JL, Barr, LJ, Garvin, F, Jones, JJ and Costantini, LC. Study of the ketogenic agent AC-1202 in mild to moderate Alzheimer’s disease: a randomized, double-blind, placebocontrolled, multicenter trial. Nutr Metab (Lond) 2009; 6: 31.
Herrmann, N, Chau, SA, Kircanski, I and Lanctot, KL. Current and emerging drug treat-ment options for Alzheimer’s disease: a systematic review. Drugs 2011; 71(15): 2031-2065.
Hoyer, S. Brain metabolism and the incidence of cerebral perfusion disorders in organic psychoses. Dtsch. Z Nervenheilkd. 1970; 197(4): 285-292.
Kossoff, EH and Hartman, AL. Ketogenic diets: new advances for metabolism-based therapies. Curr Opin Neurol 2012; 25(2): 173-178.
Krikorian, R, Shidler, MD, Dangelo, K, Couch, SC, Benoit, SC and Clegg, DJ. Dietary ketosis enhances memory in mild cognitive impairment. Neurobiol. Aging 2012; 33(2): 425-427.
Lambrechts, DA, Wielders, LH, Aldenkamp, AP, Kessels, FG, de Kinderen, RJ and Majoie, MJ. The ketogenic diet as a treatment option in adults with chronic refractory epilepsy: efficacy and tolerability in clinical practice. Epilepsy Behav 2012; 23(3): 310-314.
Lanctot, KL, Rajaram, RD and Herrmann, N. Therapy for Alzheimer’s Disease: How Effective are Current Treatments? Ther Adv Neurol Disord. 2009; 2(3): 163-180.
McPherson, PA and McEneny, J. The biochemistry of ketogenesis and its role in weight management, neurological disease and oxidative stress. J Physiol Biochem 2012; 68(1): 141-151.
Nam, SH, Lee, BL, Lee, CG, Yu, HJ, Joo, EY, Lee, J and Lee, M. The role of ketogenic diet in the treatment of refractory status epilepticus. Epilepsia 2011; 52(11): e181-e184.
Newport, M, 2008. What if there was a cure for Alzheimer’s disease and no one knew?
Newport, M, 2011. Alzheimer’s Disease. What If There Was A Cure? Basic Health Publi-cations, Inc, Laguna Beach, CA.
Owen, OE, Morgan, AP, Kemp, HG, Sullivan, JM, Herrera, MG and Cahill, GF, Jr. Brain metabolism during fasting. J Clin Invest 1967; 46(10): 1589-1595.
Page, KA, Williamson, A, Yu, N, McNay, EC, Dzuira, J, McCrimmon,RJ. and Sherwin, RS. Medium-Chain Fatty Acids Improve Cognitive Function in Intensively Treated Type 1 Diabetic Patients and Support In Vitro Synaptic Transmission During Acute Hypoglycemia. Diabetes 2009; 58(5): 1237-1244.
Popp, J and Arlt,S. Pharmacological treatment of dementia and mild cognitive impair-ment due to Alzheimer’s disease. Curr Opin Psychiatry 2011; 24(6): 556-561.
Prins,ML. Cerebral metabolic adaptation and ketone metabolism after brain injury. J Cereb Blood Flow Metab 2008; 28(1): 1-16.
Reger, MA, Henderson, ST, Hale, C, Cholerton, B, Baker, LD, Watson, GS, Hyde, K, Chapman, D. and Craft, S. Effects of betahydroxybutyrate on cognition in memory-impaired adults. Neurobiol. Aging 2004; 25(3): 311-314.
Veech, RL. The therapeutic implications of ketone bodies: the effects of ketone bodies in pathological conditions: ketosis, ketogenic diet, redox states, insulin resistance, and mitochondrial metabolism. Prostaglandins, Leukotrienes and Essential Fatty Acids 2004; 70(3): 309-319.
White, H and Venkatesh, B. Clinical review: ketones and brain injury. Crit Care 2011; 15(2): 219.
Yao, J, Rettberg, JR, Klosinski, LP, Cadenas, E and Brinton, RD. Shift in brain metabolism in late onset Alzheimer’s disease: implications for biomarkers and therapeutic interventions. Mol Aspects Med 2011; 32(4-6): 247-257.
Molecular chaperones as 21st century solutions for neurodegenerative disorders
Molecular chaperones as 21st century solutions for neurodegenerative disorders
A look at catecholamine-regulated protein 40 (CRP40)
The aging population in developed countries is experiencing an increase in the prevalence of neurodegenerative disorders that are placing a high burden on healthcare systems. The combined problems of lack of sustainable therapies and the absence of validated biomarkers for early diagnosis and intervention make these debilitating disorders a high priority for research. However, before biomarkers and therapies can be developed, scientists need to establish a clearer and more complete understanding of the molecular mechanisms behind neurodegeneration. Recently, researchers have linked the pathogenesis of many neurodegenerative disorders such as Parkinson’s (PD) and Alzheimer’s disease (AD) to abnormal protein folding, oxidative stress, and mitochondrial dysfunction. A specific class of proteins, called molecular chaperones (also known as heat-shock proteins), have emerged as potential targets for neuroprotection because of their ability to modulate abnormal protein folding and aggregation in disease states. We have discovered in our laboratory a novel molecular chaperone protein called Catecholamineregulated protein 40 (CRP40; 40kDa) that is expressed solely in the central nervous system (CNS) and blood. Our preliminary findings indicate CRP40 is an alternative splice variant of a larger, ubiquitously-expressed protein called mortalin, which has been linked to protein homeostasis, pathophysiology of neurodegenerative disorders, and has been shown to protect neurons from oxidative stress. Studies on CRP40 have revealed the presence of a conserved heat-shock motif, upregulated expression under cell stress, as well as its ability to decrease aggregation of proteins, suggesting heat-shock like functions of this novel protein. Interestingly, evidence has revealed that CRP40 may play a role in neurodegenerative disorders characterized by impairments of the dopamine (DA) system, such as Schizophrenia (SCZ) and PD, through its ability to bind DA and colocalize with components of the DA-synthesis pathway. Our group has also discovered that CRP40 expression is dysregulated in human post-mortem SCZ and Parkinson’s brain samples – a compelling finding that requires further study. In this review, we present evidence that CRP40 may play a role in CNS disorders, particularly disorders related to abnormal protein folding and impairments of the dopaminergic system. Further study of CRP40 and its mechanisms may aid in increasing the understanding of molecular chaperone function in neurodegenerative disorders, which is essential for accelerating future developments of biomarkers and sustainable therapeutics.
Neurodegenerative disorders:How molecular chaperones play a role
Neurodegenerative diseases are increasing in prevalence in many developing countries and this trend is set to continue due to the rapidly aging population. They encompass a heterogeneous group of disorders that can be described by progressive and selective loss of neuronal systems that are anatomically or physiologically related. Despite research advances over the past several decades, critical challenges still remain in understanding the pathology and underlying mechanisms behind these debilitating disorders. Converging lines of evidence have suggested a common “protein-conformational” pathogenic pathway for AD, PD, familial amyotrophic lateral sclerosis (FALS), Huntington’s disease (HD) and related polyglutamine (polyQ) expansion diseases. Collectively, these disorders are characterized by the abnormal accumulation of insoluble neuronal or extracellular protein aggregates in the CNS (Hartl 2011, Muchowski 2005). However, the significance of these phenomena to the process of neurodegeneration is incompletely understood. Several groups have proposed that the formation of protein aggregates may be neuroprotective (Caughey 2003, Sisodia 1998). However, any perturbation of protein homeostasis can impair the ability of proteins to function properly and perform their diverse biological roles that are essential for life.
There is still some debate about whether the aggregates formed by disease proteins are toxic. Evidence has suggested that misfolded proteins can induce excitotoxicity or apoptosis via activation of caspase 3, and also affect neuronal synaptic function and axonal transport (Forman 2004). As well, toxic species in neurodegenerative disorders have been linked to various inflammatory responses, including increased oxidative stress, and mitochondrial dysfunction (Lin 2006). Indeed, there seems to be a common link between misfolded proteins and the resulting aberrations that occur in neurodegenerative disorders.
Protein quality or proteostasis is regulated by an integrated network of proteins known as molecular chaperones. Molecular chaperones perform diverse roles in the cell including assisting with de novo protein folding, refolding of denatured proteins, protein trafficking, and degradation (Hartl 2011, Soti 2005). Numerous classes of molecular chaperones have been described thus far. Many of these proteins are also known as heat-shock proteins (Hsp) or stress proteins because it has been found that various cellular stresses upregulate their expression. These may include heat-shock, glucose deprivation, exposure to heavy metals, protein kinase C (PKC) stimulators, amino acid analogues, calcium increasing agents, ischemia, microbial infections and various hormones (Ellis 2006).
Different families of molecular chaperone proteins are classified according to their molecular weights and include the Hsp40, Hsp60, Hsp70, Hsp90, Hsp100, and small Hsp proteins (Ellis 2006). Heat-shock proteins are expressed in the cytosol, nucleus, mitochondria, and endoplasmic reticulum, and typically have long half-lives of about 48 hours (Ellis 2006). Although the function of these proteins is broad, the Hsp70s, Hsp90s, and the chaperonins (Hsp60s) are specifically involved in de novo protein folding and refolding. These classes of proteins are characterized as multicomponent molecular machines that have the ability to recognize exposed hydrophobic amino-acid side chains of misfolded proteins and guide proper folding through adenosine triphosphate (ATP)- and cofactor- mediated mechanisms (Hartl 2011). Due to their various roles, molecular chaperones are currently being investigated in a variety of disorders.
Indeed, molecular chaperone proteins are emerging as important players in a number of neurodegenerative diseases characterized by aberrations in protein conformation. The purported roles of molecular chaperones in neurodegeneration may include the following: 1) control of potentially toxic structural changes in disease-related polypeptides; 2) regulation of aggregation of proteins by directing towards formation of less toxic species; 3) control of degradation of toxic agents and preservation of cellular degradation systems; 4) involvement in cell signalling and apoptotic events stimulated by abnormal protein aggregates, along with preservation of survival pathways; and 5) protection of mitochondria and neurons against oxidative stress evidenced in neurodegenerative disorders (Muchowski 2005). Further understanding of cellular chaperone networks will help to define their roles in neurodegenerative diseases where protein homeostasis has been impaired, and elucidate ways in which they may be manipulated to serve as possible therapeutic factors in these debilitating disorders.
Mortalin: an essential mitochondrial heat-shock protein
The heat-shock protein 70 (Hsp70; 70kDa) family represents one of the most ubiquitous classes of molecular chaperones. Mortalin, also known as the mitochondrial heat-shock protein (also referred to as mtHsp70 or Grp75), is an essential ubiquitously expressed molecular chaperone with multiple roles in mitochondrial biogenesis, maintenance of mitochondrial protein integrity, and regulation of import of mitochondrial proteins into the matrix (Deocaris 2008). Recently, mortalin has been implicated in neurogenesis and neurodegeneration processes and linked to AD, PD, as well as HD (Deocaris 2008). Also, since mortalin has been found to exert various cytoprotective functions that permit cell survival under stressful conditions, it has been implicated in cancer and aging systems (Kaul 2007).
Evidence for the involvement of mortalin in several human diseases has been accumulating over the past decade. Gabriele and colleagues (2010) studied the possible involvement of mortalin in the pathogenesis of SCZ and have shown that antisense knockdown of mortalin in the medial prefrontal cortex resulted in impairments of DA-mediated behaviours including prepulse inhibition and social interaction deficits (Gabriele 2010). Mortalin has also been implicated in AD through several studies. Osorio and colleagues (2007) showed differential expression of mortalin isoforms in hippocampi of AD patients. In another study using an animal model of AD, the ApoE knockout mouse model, it was shown that mortalin sustained ADassociated oxidative damage, suggesting the involvement of this protein in the pathogenesis of AD (Choi 2004). Mortalin has also been linked to neurodegeneration in PD based on interactions with PD-associated proteins, including the redox-sensing DJ-1 protein that maintains mitochondrial function (Kaul 2007).
Mortalin is expressed from chromosome 5q31.1 (Kaul 1995). A recent study found that PD patients display mutations in the mortalin gene that have been associated with impaired mitochondrial function that is critical in protection from neurodegeneration (Burbulla 2010). Two of these mutations were located in the carboxyl-terminal substrate-binding domain of mortalin, indicating that this functional region of mortalin further characterization. The combined evidence from literature underscores the critical role of mortalin as an important mitochondrial stress protein in a variety of neurodegenerative disorders that needs to be investigated in more detail.
Interestingly, several studies have suggested that mortalin may play a role in brain ischemia, as it is upregulated following brain injury (Deocaris 2008). This protein may serve a protective function in ischemia by limiting the accumulation of reactive oxygen species in neurons (Liu 2005). Further, in vivo overexpression of mortalin in rat brain neurons and astrocytes following induction of ischemia significantly reduced infarct volume, improved neurological function and provided protection from oxidative damage in animals (Xu 2009).
Mortalin has been found to interact with the apoptosis-inducing factor (AIF) (Sun 2006). Normally, when AIF is released from the mitochondria, it may be translocated to the nucleus where it can cause caspase-independent apoptosis. It has been suggested that mortalin sequesters AIF and thus suppresses cell death (Sun 2006). Interestingly, this protective ability of mortalin was not retained in Hsp70 deletion mutants lacking the carboxyl-terminal (Ravagnan 2001). The functionality of the mortalin carboxyl-terminal was further confirmed in studies by Sun and colleagues (2006) who found that Hsp70 mutants lacking the entire amino-terminal domain and Hsp70 ATPasedeficient point mutants retained the ability to protect primary astrocytes against ischemic insults in vitro as well as to inhibit AIF translocation to the nucleus. This indicated that the carboxyl-terminal was indeed sufficient for protection against ischemic brain injury (Sun 2006).
Indeed, the carboxyl-terminal of mortalin seems to serve a specific function in neurodegenerative disorders and oxidative stress. McMaster University researchers have recently discovered a novel protein that is related to the carboxyl-terminal of mortalin, and this protein is under investigation for its possible roles in neurodegeneration.
Catecholamine-regulated protein 40 (CRP40): A novel molecular chaperone
The Catecholamine-Regulated Protein 40 is a novel heat-shock like protein that was discovered and characterized by Drs. Mishra and Gabriele of McMaster University (Hamilton, Canada) (Gabriele 2009, Nair 2001). CRP40 is a splice variant of mortalin, showing significant homology to mortalin’s carboxyl-terminus. Specifically, the sequence between exons 10-17 is identical to mortalin, with a promoter region at intron 9 (Gabriele 2009). Interestingly, this carboxyl-terminus homology means that the above discussed PD-related mortalin mutations would affect CRP40 function as well. Like mortalin, CRP40 belongs to the Hsp70 family, as evidenced by the following characteristics: 1) CRP40 exhibits the characteristic and conserved heat-shock motif; 2) expression of CRP40 is inducible by elevated temperature; and 3) the organellar localization motif, which allows Hsp70s to translocate between organelles, is conserved (Gabriele 2009, Nair 2001). Although the full mechanistic functions of CRP40 have not yet been elucidated, evidence suggests that it may play a role as a molecular chaperone, not unlike mortalin. Further study is required to determine how CRP40 functions in protein folding and proteostasis as related to neurodegenerative conditions.
One major distinction between CRP40 and mortalin lies in their localization. While mortalin is expressed in all cells, CRP40 is found exclusively in the CNS and in specific blood cells (Ross 1995). This pattern of expression makes CRP40 of particular interest to research in neurological disorders, especially neurodegeneration, as CRP40 may serve a more specific function in CNS disorders.
CRP40 may have similar roles to mortalin as a chaperone protein, but CRP40 also displays some functions in neuroprotection. Thus far, studies have shown that upregulation of CRP40 in the presence of excess DA can actually be inhibited by treatment of cells with antioxidants, suggesting that CRP40 is a reactant protein to the oxidative stress caused by the natural oxidation of DA (Nair 2001). Further, heat-shocked cells that were subsequently treated with CRP40 protein showed decreased denaturation and aggregation of proteins in comparison to untreated cells — a finding that highlights CRP40 as a heat-shock protein and molecular chaperone (Gabriele 2009, Nair 2001).
Unlike other members of the Hsp70 family, CRP40 contains the tyrosine and aspartate residues necessary for DA binding (Gabriele 2009, Nair 2001). Staining studies show that CRP40 colocalizes in neuronal cells with DA and Tyrosine Hydroxylase (TH), the ratelimiting molecule in DA synthesis (Gabriele 2009, Goto 2001, Nair 2001). This protein actually displays covalent interactions with the catecholamine neurotransmitters, but does not interrelate with serotonin or other amines (Nair 2001, Ross 1993, Ross 1995). In fact, CRP40 expression is inducible by presence of excess DA, which makes it an interesting target for research related to the neurodegenerative disorders with dopaminergic components, like SCZ and PD (Nair 2001).
Recent reports have revealed the importance of CRP40 to these dopaminergic degenerative conditions. Gabriele et al. (2005) conducted a study using human post-mortem SCZ samples obtained from the Stanley Foundation Neuropathology Consortium. Results showed that CRP40 is dysregulated in the affected brain regions and suggest its involvement in the pathogenesis of SCZ. In fact, there was a significant decrease in the protein expression of CRP40 in patients with SCZ (Gabriele 2005). Similarly, in studies using human postmortem PD brain samples, Mortalin/CRP40 were found to be dysregulated in PD-specific brain regions (Jin 2006, Shi 2008). Specifically, there is a quantitative decrease in Mortalin/CRP40 expression with progression of the disease (Shi 2008). These reports present important evidence that CRP40 may act as a potential biomarker for neurological disorders. Future studies will be conducted to determine whether CRP40 and its dysregulation in neurological disorders may be exploited for early disease-detection and monitoring of disease progression.
Since the discovery of CRP40, new findings and evidence have converged to reveal its involvement in neurodegenerative disease pathology. Specifically, CRP40 seems to have great significance to CNS disorders involving DA. As well, CRP40 may be involved in the crucial process of proteostasis that is impaired in neurodegenerative diseases. The Gabriele group is moving forward with this important research and has engaged in studies of both basic and clinical sciences in order to elucidate potential breakthroughs of CRP40 in the realm of neurodegenerative disorders.
References
Burbulla LF, Schelling C, Kato H, Rapaport D, Woitalla D, Schiesling C, Schulte C, Sharma M, Illig T, Bauer P, Jung S, Nordheim A, Schöls L, Riess O, Krüger R. Dissecting the role of the mitochondrial chaperone Mortalin in Parkinson’s disease: functional impact of disease-related variants on mitochondrial homeostasis. Hum Mol Genet. 2010;19(22):4437-4452.
Caughey B, Lansbury PT. Protofibrils, pores, fibrils, and neurodegeneration: separating the responsible protein aggregates from the innocent bystanders. Annu Rev Neurosci. 2003;26:267-298.
Choi J, Forster MJ, McDonald SR, Weintraub ST, Carroll CA, Gracy RW. Proteomic identification of specific oxidized proteins in ApoE-knockout mice: relevance to Alzheimer’s disease. Free Radic Biol Med. 2004;36(9):1155-1162.
Deocaris CC, Kaul SC, Wadhwa R. From proliferative to neurological role of an Hsp70 stress chaperone, mortalin. Biogerontol. 2008;9(6):391-403.
Ellis RJ. Molecular chaperones: assisting assembly in addition to folding. Trends Biochem Sci. 2006;31(7):395-401.
Forman MS, Trojanowski JQ, Lee VM. Neurodegenerative diseases: a decade of discoveries paves the way for therapeutic breakthroughs. Nat Med. 2004;10(10):1055-1063.
Gabriele JP, Chong VZ, Pontoriero GF, Mishra RK. Decreased expression of a 40-kDa catecholamine-regulated protein in the ventral striatum of schizophrenic brain specimens from the Stanley Foundation Neuropathology Consortium. Schizophr Res. 2005;74(1):111-119.
Gabriele N, Pontoriero GF, Thomas N, Shethwala SK, Pristupa ZB, Gabriele JP. Knockdown of mortalin within the medial prefrontal cortex impairs normal sensorimotor gating. Synapse. 2010;64(11):808-813.
Gabriele J, Pontoriero GF, Thomas N, Thomson CA, Skoblenick K, Pristupa ZB, Mishra RK. Cloning, characterization, and functional studies of a human 40-kDa catecholamine-regulated protein: implications in central nervous system disorders. Cell Stress Chaperon. 2009;14(6):555-567.
Goto A, Doering L, Nair VD, Mishra RK. Immunohistochemical localization of a 40-kDa catecholamine regulated protein in the nigrostriatal pathway. Brain Res. 2001;900(2):314-319.
Hartl FU, Bracher A, Hayer-Hartl M. Molecular chaperones in protein folding and proteostasis. Nature. 2011;475(7356):324-332.
Jin J, Hulette C, Wang Y, Zhang T, Pan C, Wadhwa R, Zhang J. Proteomic identification of a stress protein, mortalin/mthsp70/GRP75: relevance to Parkinson disease. Mol Cell Proteomics. 2006;5(7):1193- 1204.
Kaul SC, Deocaris CC, Wadhwa R. Three faces of mortalin: a housekeeper, guardian and killer. Exp Gerontol. 2007;42(4):263-274.
Kaul SC, Wadhwa R, Matsuda Y, Hensler PJ, Pereira-Smith OM, Komatsu Y, Mitsui Y. Mouse and human chromosomal assignments of mortalin, a novel member of the murine hsp70 family of proteins. FEBS Lett. 1995;361(2-3):269-272.
Lin MT, Beal MF. Mitochondrial dysfunction and oxidative stress in neurodegenerative diseases. Nature. 2006;443(7113):787-795.
Liu Y, Liu W, Song XD, Zuo J. Effect of GRP75/mthsp70/PBP74/ mortalin overexpression on intracellular ATP level, mitochondrial membrane potential and ROS accumulation following glucose deprivation in PC 12 cells. Mol Cell Biochem. 2005;268(1-2):45-51.
Muchowski PJ, Wacker JL. Modulation of neurodegeneration by molecular chaperones. Nat Rev Neurosci. 2005;6(1):11-22.
Nair VD, Mishra RK. Molecular cloning, localization and characterization of a 40-kDa catecholamine-regulated protein. J Neurochem. 2001;76(4):1142-1152.
Osorio C, Sullivan PM, He DN, Mace BE, Ervin JF, Strittmatter WJ. Mortalin is regulated by APOE in hippocampus of AD patients and by human APOE in TR mice. Neurobiol Aging. 2007;28(12):1853-1862.
Ravagnan L, Gurbuxani S, Susin SA, Maisse C, Daugas E, Zamzami N, Mak T, Jaattela M, Penninger JM, Garrido C, Kroemer G. Heat-shock protein 70 antagonizes apoptosis-inducing factor. Nat Cell Biol. 2001;3(9):839-843.
Ross GM, McCarry BE, Mishra RK. Covalent Affinity Labeling of Brain Catecholamine-Absorbing Proteins Using a High-Specific- Activity Substituted Tetrahydronaphthalene. J Neurochem. 1995;65(6):2783-2789.
Ross GM, McCarry BE, Thakur S, Mishra RK. Identification of novel catecholamine absorbing proteins in the central nervous system. J Mol Neurosci. 1993;4(3):141-148.
Sisodia SS. Nuclear inclusions in glutamine repeat disorders: are they pernicious, coincidental, or beneficial? Cell. 1998;95(1):1-4.
Shi M, Jin J, Wang Y, Beyer RP, Kitsou E, Albin RL, Gearing M, Pan C, Zhang J. Mortalin: A Protein Associated With Progression of Parkinson Disease? J Neuropathol Exp Neurol. 2008;67(2):117-124.
Soti C, Nagy E, Giricz Z, Vígh L, Csermely P, Ferdinandy P. Heat shock proteins as emerging therapeutic targets. Br J Pharmacol. 2005;146(6):769-780.
Sun Y, Ouyang YB, Xu L, Chow AM, Anderson R, Hecker JG, and Giffard RG. The carboxyl-terminal domain of inducible Hsp70 protects from ischemic injury in vivo and in vitro. J Cereb Blood Flow and Metab. 2006;26(7):937-950.
Xu L, Voloboueva LA, Ouyang Y, Emery JF, Giffard RG. Overexpression of mitochondrial Hsp70/Hsp75 in rat brain protects mitochondria, reduces oxidative stress, and protects from focal ischemia. J Cereb Blood Flow and Metab. 2009;29(2):365-374. Lubarda.
DHEA
DHEA
Outcomes in female fertility
DHEA and DHEA-S are together the most abundant steroid hormones in the body, and function as weak androgens. In women, DHEA is mainly converted to testosterone, and to a lesser degree estrogen (Buster 1992). Androgen stimulation is thought to play a key role in early follicular development, with androgen receptor deficient mice showing problems with follicular development, ovulation, and ovarian response to gonadotropins that are similar to problems experienced by women with diminished ovarian reserve (DOR). Fuelled by an early case report showing a “dramatic and continuous” (Barad 2005) improvement in ovarian function over an 11 month selftreatment period with DHEA alongside weekly acupuncture, subsequent studies have since shown statistically significant improvements in clinical pregnancy rates, live birth rates, miscarriage rates, increased antral follicle count, number of retrievable mature oocytes and high quality embryos, reduced aneuploidy, and a reduction of IVF cycle cancellations. DHEA is currently in use as a treatment for DOR, and further investigations are warranted to determine whether DHEA may be of benefit in other related conditions such as premature ovarian failure.
Introduction
Dehydroepiandrosterone (DHEA), and its sulphated form DHEA-S, are weak adrenal androgens and the most abundant steroid hormone in the body (Andus 2003). DHEA acts as a precursor for other steroid hormones in the body, predominantly estradiol and testosterone via androstenedione (Gleicher 2012, Olech 2005). In women, DHEA is mainly converted to testosterone, and to a lesser degree estrogen (Buster 1992, Gleicher 2012). As a result, DHEA is used as a hormone replacement for older women, in which circumstance it has been shown to benefit bone health (Weiss 2009). DHEA has also been shown to possess anti-inflammatory effects through inhibition of NF-kappaB activation, as well as inhibition of IL-6 and IL-12 secretion via PPAR-alpha (Andus 2003), and has on the basis of this activity been utilized in various autoimmune conditions (Petri 2004, Andus 2003). These applications will be covered in detail in part II of this review. This review focuses on DHEA supplementation as a treatment for diminished ovarian reserve (DOR) and female infertility.
Review of Reproductive Physiology
Grynnerup defines the follicle as “the basic functional unit in the ovaries” (2012). The follicle consists of the oocyte in association with surrounding theca cells and granulosa cells; during follicular development, granulosa cells support the maturation of and interact with the oocyte, producing growth factors and hormones to direct follicular development. After ovulation, the granulosa cells become the corpus luteum and secrete progesterone. The number of follicles is set during embryonic development; over the course of a woman’s life, resting primordial follicles are recruited to become growing follicles, a process called initial recruitment that continues until the follicle pool is exhausted (Grynnerup 2012). Growing follicles either undergo atresia (most) or they undergo cyclic recruitment, an ongoing process that begins at puberty, which involves recruitment by FSH. A cohort of follicles is selected with each “cycle” of recruitment, only one of which is selected to become the dominant follicle that will reach ovulation. Figure 1 depicts the stages of follicular maturation. During early follicular development (pre-antral and antral stages), the granulosa cells secrete anti-Mullerian hormone (AMH). AMH inhibits excessive recruitment by FSH, which would lead to premature exhaustion of the follicle pool (Grynnerup 2012); however, since it is produced by the early pre-antral follicles, AMH can be used as a marker of the remaining growing follicular pool, and by extension the remaining primordial follicle pool, or ovarian reserve (Grynnerup 2012). Similarly, inhibin B is a less studied but similar marker that is produced during the early to mid stages of follicular development; it inhibits pituitary release of FSH and may also serve as a marker of ovarian reserve (Sill 2009).
The mechanism by which DHEA impacts folliculogenesis is not well defined. Until recently, DHEA was thought to promote follicular development during the very early stages of follicular maturation (Gleicher 2012). This is perhaps surprising since androgens are considered detrimental in other conditions of impaired fertility such as polycystic ovarian syndrome (PCOS). Nonetheless, evidence based on animal studies shows that androgen receptor mediated function in granulosa cells is crucial to the development of preantral follicles at early stages of development and the prevention of follicular atresia (Gleicher 2012, Sen 2010). In mouse models, androgen receptor knock out animals (ARKO) share striking resemblances with women who suffer from DOR: they become subfertile, have defective folliculogenesis, lower numbers of antral (pre-ovulatory) follicles, fewer corpora lutea, higher rates of granulosa cell apoptosis, and ultimately develop premature ovarian failure (Hu 2004, Kimura 2007, Sen 2010, Shiina 2006, Walters 2007). When subjected to superovulation through gonadotropin administration, these mice ovulate fewer oocytes (Hu 2004).
In humans, intrafollicular androgen levels have been shown to increase granulosa cell AMH (anti-Mullerian hormone) production and decrease inhibin-B levels (Andersen 2008). The authors of this study speculate that this seemingly opposite effect on AMH and inhibin B respectively may reflect a dual activity of androgens on ovarian function: during the early stage of follicular differentiation, androgens induce FSHreceptor expression on granulosa cells, promoting FSH- stimulated follicular differentiation; however in the later (pre-ovulatory) stages, excess androgens may induce atresia of granulosa cells and inhibit LH receptor formation, thus inhibiting ovulation (Andersen 2008). This type of activity does appear to be profiled in conditions of androgen excess most notably PCOS, with hyper-development of follicles but failure of ovulation.
More recently, Hyman et al demonstrated that DHEA therapy was able to improve antral follicle count but not change AMH or inhibin B, suggesting that DHEA does not so much influence the early stages of follicular development (AMH and inhibin B are secreted at these stages) as it increases the rescue rate of the small antral follicles (later stage) from atresia (increased AFC) (2013). Other earlier studies showed conflicting results with respect to AMH, with one RCT reporting no change and one retrospective study reporting a large improvement (Gleicher 2010A, Yeung 2012). It is hoped that more research will be able to more clearly define the mechanism(s) of DHEA.
DHEA. Over the past ten years or so, several investigations into the effect of DHEA supplementation in women with DOR and/ or poor responders to IVF have been conducted, following upon the heels of an initial case series in 2000 and an impressive case report in 2005 (Barad 2005, Casson 2000; both in Table 1). There are only two RCTs among 15 identified studies, in part due to the ethical difficulty of randomizing this particular population, with a highly time-sensitive condition, to an inactive arm (Gleicher 2011). Instead study designs include prospective, uncontrolled trials and case-control studies in which DHEA- treated cycles are compared to non-DHEA treated cycles in the same group of women.We include all human level interventional evidence pertaining to the treatment of infertility identified in Pubmed up to January 2013, using the terms “DHEA supplementation”.
Diminished Ovarian Reserve (DOR)
According to the Society of Assisted Reproductive Technology (SART), DOR does not consist of clearly defined criteria; rather it describes the number and quality of eggs remaining in a woman’s ovaries as assessed or estimated through a number of tests and prognostic factors (Practice Committee 2012).
The SART document describes DOR as referring to “women of reproductive age having regular menses whose response to ovarian stimulation or fecundity is reduced compared to those women of comparable age” (2012).
Tests/ factors often used to assess DOR include: day 3 FSH (>10 is considered a poor prognostic) and estradiol; antral follicle count (follicles measuring 2-10mm, normal count is >10); anti-mullerian hormone (AMH) (<1ng/ mL is considered low); clomiphene citrate challenge (FSH >10 following treatment with clomiphene); age >35 years; family history of early menopause; single ovary or history of ovarian surgery, chemotherapy, or pelvic radiation (Practice Committee 2012).
Clinical Evidence
Collectively, the 14 reviewed studies reported the following outcomes in women described as having DOR or as poor responders to IVF:
• Higher clinical pregnancy rates between 22.1- 28.4% with DHEA supplemented cycles (Barad 2007, Gleicher 2012, Gleicher 2010A, Sonmezer 2009, Weghofer 2012). Three of these trials were uncontrolled; in the one casecontrol study, the control group had a clinical pregnancy rate of 11.9%, versus 28.4% in the DHEA group, HR pregnancy 3.8 (95% CI 1.2-11.8, p<0.05) (Barad 2007). In the second study, the DHEA supplemented cycle resulted in pregnancy rates of 44.4% versus 0% after embryo transfer compared to the first unsupplemented cycle (p<0.01) (Sonmezer 2009).
• Higher live birth rate: 23.1% with DHEA compared to 4.0% (p=0.05) without DHEA (RCT evidence) (Wiser 2010).
• Lower miscarriage rate: 15.1% with DHEA, which was lower than the national average; odds of miscarriage was reduced by approximately half: OR 0.49 (0.25- 0.94, p=0.04) (Gleicher 2009). The miscarriage rate was reduced at all ages but most especially so in women over 35 years of age.
• Improvements in the follicular microenvironment due to a better blood supply to the ovaries as measured by FF HIF1 levels (Artini 2012).
• Greater numbers of mature oocytes retrieved from selected follicles following ovarian stimulation (Artini 2012); and increased number of follicles >15mm (p=0.004), 17mm (p<0.05) and 10mm respectively (Hyman 2013, Sonmezer 2009, Yeung2012 RCT evidence).
• No significant change in AMH or FSH levels, but significantly increased antral follicle count and ovarian volume at wk 12 and 20 (RCT evidence, Yeung 2012; and Hyman 2013).
• Reduction in the number of aneuploidy embryos (p<0.029) (Gleicher 2010B) and increased embryo quality (p=0.04) (RCT evidence) (Wiser 2010).
• Increased good quality embryos at day two and three (p<0.05) (Barad 2006, Sonmezer 2009).
• Decrease in mean day three estradiol (p<0.01) (Sonmezer 2009).
• Reduction in cycle cancellation rates, 5.3% versus 42.1%, p<0.01 (Hyman 2013, Sonmezer 2009).
Of note, Gleicher et al investigated the effect of DHEA conversion to testosterone as a prognostic marker of the response to DHEA (2012). Androgen conversion was compared between conception and non-conception cycles. They found that the beneficial effect of DHEA on pregnancy rates was statistically associated with the efficiency of androgen conversion from DHEA, and the amplitude of testosterone gain; both younger women and certain FMR1 genotypes were found to convert more efficiently (Gleicher 2012).
In an excellent review, Gleicher and Barad briefly discuss other fertility related applications, for example the use of DHEA for idiopathic infertility and premature ovarian failure (POF) as distinct from DOR; however there is a lack of clinical evidence regarding these applications and the focus of published papers to date is on DOR (2011). Anecdotally, however, Gleicher et al report a handful of cases of POF at their and colleagues’ fertility centers that achieved pregnancy following several months use of DHEA, and are currently investigating this more rigorously through a clinical trial(ClinicalTrials.gov ID#NCT00948857) (Gleicher2011, Mamas 2009).
Criticism
Some have been critical of the use of DHEA in clinical practice for the treatment of DOR. In a commentary article, Urman et al argue that the evidence in support of DHEA is not of sufficient quality to merit use in practice (2012). At the time of publication there was only retrospective or uncontrolled studies and one RCT in existence (Wiser 2010), the latter of which Urman critiques as underpowered to detect the effects on pregnancy and other endpoints that were reported (2012). Although one additional placebo controlled RCT has since reported similar benefit of DHEA on DOR (Yeung 2012), there is admittedly still a need for further RCT level evidence. In addition, Urman is of the opinion that DOR with low AMH and few antral follicles will not be amenable to pharmacological therapy of any sort: “It is unlikely that any medication will be of benefit in a woman harboring ovaries devoid of antral follicles. After all, one cannot stimulate what is not there” (Urman 2012). This statement does not however sufficiently explain the observations reported in several studies of DHEA that have been published, the majority of which have documented some level of improvement in markers of ovarian function associated with use of DHEA.
A recent study exploring the mechanism of DHEA in DOR suggested that rather than somehow increasing the set number of follicles a woman possesses, DHEA may instead increase the quality of the few existing, antral follicles (Hyman 2013). This study found that although DHEA had no impact on baseline AMH or inhibin B levels, which are secreted by early to mid stage follicles, the antral follicle count nonetheless improved. Authors concluded that “DHEA does not appear to exert influence via recruitment of pre-antral or very small antral follicles (no change in AMH and inhibin B), but rather by rescue from atresia of small antral follicles (increased AFC)” (Hyman 2013). There is clearly a need for further research to confirm the effect and mechanism of DHEA, however in the meantime a basis to utilize this promising therapy does appear to exist, particularly in women whose existing therapeutic options are limited.
Dosing
The dosage generally used by studies reviewed here is 25mg three times per day. This is beyond that considered to be physiologic replacement dosages, typically considered to be 20-50mg/d for men and 10-30mg/d for women; however Olech points out that “higher dosages may be necessary for increasing suppressed DHEA and DHEAS levels secondary to chronic disease, adrenal exhaustion, and corticosteroid therapy” (2005). Indeed, higher dosages have been used in lupus, as will be reviewed in part II of this series.
Adverse Effects
No serious side effects were reported by the studies included in this review. Gleicher et al report having treated over 1000 women with DOR with DHEA 25mg three times daily and observing no serious side effects other than acne, hair loss, or oily skin (2011).
Conclusion
DHEA is a weak adrenal androgen that appears to positively influence early follicular development in women with diminished ovarian reserve (DOR). Two RCTs and 13 non-randomized studies in women with DOR show a promising role for DHEA in improving ovarian function and fertility. These studies suggest that DHEA can be dosed at 25mg three times daily for a minimum of six weeks up to three to four months for DOR. DHEA is a relatively safe agent with few side effects that may include acne, hair loss and oily skin.
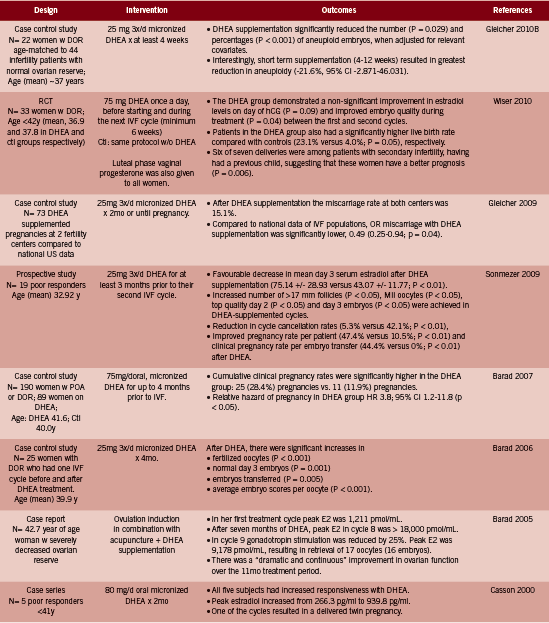
and Embryology; FF follicular fluid; HIF1 hypoxia inducible factor 1; NR not reported; POA premature ovarian aging; POI premature ovarian
insufficiency; w with; w/o without.
References
Andersen CY, Lossl K. Increased intrafollicular androgen levels affect human granulosa cell secretion of anti-Müllerian hormone and inhibin-B. FertilSteril. 2008 Jun;89(6):1760-5.
Andus T, Klebl F, Rogler G, Bregenzer N, Schölmerich J, Straub RH. Patients with refractory Crohn’s disease or ulcerative colitis respond to dehydroepiandrosterone: a pilot study. Aliment Pharmacol Ther. 2003 Feb;17(3):409-14.
Artini PG, Simi G, Ruggiero M, Pinelli S, Di Berardino OM, Papini F, Papini S, Monteleone P, Cela V. DHEA supplementation improves follicular microenviroment in poor responder patients. GynecolEndocrinol. 2012 Sep;28(9):669-73.
Barad D, Brill H, Gleicher N. Update on the use of dehydroepiandrosterone supplementation among women with diminished ovarian function. J Assist Reprod Genet. 2007 Dec;24(12):629-34.
Barad D, Gleicher N. Effect of dehydroepiandrosterone on oocyte and embryo yields, embryo grade and cell number in IVF. Hum Reprod. 2006 Nov;21(11):2845-9.
Barad DH, Gleicher N. Increased oocyte production after treatment with dehydroepiandrosterone. FertilSteril. 2005 Sep;84(3):756.
Buster JE, Casson PR, Straughn AB, Dale D, Umstot ES, Chiamori N, Abraham GE. Postmenopausal steroid replacement with micronized dehydroepiandrosterone: preliminary oral bioavailability and dose proportionality studies. Am J Obstet Gynecol. 1992 Apr;166(4):1163-8; discussion 1168-70.
Casson PR, Lindsay MS, Pisarska MD, Carson SA, Buster JE.Dehydroepiandrosterone supplementation augments ovarian stimulation in poor responders: a case series. Hum Reprod. 2000 Oct;15(10):2129-32.
Gleicher N, Kim A, Weghofer A, Shohat-Tal A, Lazzaroni E, Lee HJ, Barad DH. Starting and resulting testosterone levels after androgen supplementation determine at all ages in vitro fertilization (IVF) pregnancy rates in women with diminished ovarian reserve (DOR). J Assist Reprod Genet.2012 Dec 5. [Epub ahead of print]
Gleicher N, Barad DH. Dehydroepiandrosterone (DHEA) supplementation in diminished ovarian reserve (DOR). ReprodBiolEndocrinol. 2011 May 17;9:67.
Gleicher N, Weghofer A, Barad DH. Dehydroepiandrosterone (DHEA) reduces embryo aneuploidy: direct evidence from preimplantation genetic screening (PGS). ReprodBiolEndocrinol. 2010 Nov 10;8:140.
Gleicher N, Weghofer A, Barad DH. Improvement in diminished ovarian reserve after dehydroepiandrosterone supplementation. Reprod Biomed Online. 2010 Sep;21(3):360-5. B
Gleicher N, Ryan E, Weghofer A, Blanco-Mejia S, Barad DH. Miscarriage rates after dehydroepiandrosterone (DHEA) supplementation in women with diminished ovarian reserve: a case control study. ReprodBiolEndocrinol. 2009 Oct 7;7:108. doi: 10.1186/1477-7827-7-108.
Grynnerup AG, Lindhard A, Sørensen S. The role of anti-Müllerian hormone in female fertility and infertility – an overview. Acta Obstet Gynecol Scand. 2012 Nov;91(11):1252-60.
Hu YC, Wang PH, Yeh S, Wang RS, Xie C, Xu Q, Zhou X, Chao HT, Tsai MY, Chang C. Subfertility and defective folliculogenesis in female mice lacking androgen receptor. ProcNatlAcadSci U S A. 2004 Aug 3;101(31):11209-14.
Hyman JH, Margalioth EJ, Rabinowitz R, Tsafrir A, Gal M, Alerhand S, Algur N, Eldar-Geva T. DHEA supplementation may improve IVF outcome in poor responders: a proposed mechanism. Eur J Obstet Gynecol Reprod Biol. 2013 Jan 8. [Epub ahead of print]
Kimura S, Matsumoto T, Matsuyama R, Shiina H, Sato T, Takeyama K, Kato S. Androgen receptor function in folliculogenesis and its clinical implication in premature ovarian failure. Trends EndocrinolMetab. 2007 Jul;18(5):183-9.
Mamas L, Mamas E. Premature ovarian failure and dehydroepiandrosterone.Fertil Steril. 2009 Feb;91(2):644-6.
Olech E, Merrill JT. DHEA supplementation: the claims in perspective. Cleve Clin J Med. 2005 Nov;72(11):965-6, 968, 970-1 passim.
Petri MA, Mease PJ, Merrill JT, Lahita RG, Iannini MJ, et al. Effects of prasterone ondisease activity and symptoms in women with active systemic lupus erythematosus. Arthritis Rheum. 2004 Sep;50(9):2858-68.
Practice Committee of American Society for Reproductive Medicine. Diagnostic evaluation of the infertile female: a committee opinion. FertilSteril. 2012 Aug;98(2):302-7.
Sen A, Hammes SR. Granulosa cell-specific androgen receptors are critical regulators of ovarian development and function. MolEndocrinol. 2010 Jul;24(7):1393-403.
Shiina H, Matsumoto T, Sato T, Igarashi K, Miyamoto J, Takemasa S, Sakari M, Takada I, Nakamura T, Metzger D, Chambon P, Kanno J, Yoshikawa H, Kato S. Premature ovarian failure in androgen receptor-deficient mice. ProcNatlAcadSci U S A. 2006 Jan 3;103(1):224-9.
Sills ES, Alper MM, Walsh AP. Ovarian reserve screening in infertility: practical applications and theoretical directions for research. Eur J Obstet Gynecol Reprod Biol. 2009 Sep;146(1):30-6.
Sönmezer M, Ozmen B, Cil AP, Ozkavukçu S, Taşçi T, Olmuş H, Atabekoğlu CS. Dehydroepiandrosterone supplementation improves ovarian response and cycle outcome in poor responders. Reprod Biomed Online. 2009 Oct;19(4):508-13.
Traish AM, Kang HP, Saad F, Guay AT. Dehydroepiandrosterone (DHEA)—a precursor steroid or an active hormone in human physiology. J Sex Med. 2011 Nov;8(11):2960-82; quiz 2983.
Urman B, Yakin K. DHEA for poor responders: can treatment be justified in the absence of evidence? Reprod Biomed Online. 2012 Aug;25(2):103-7.
Walters KA, Allan CM, Jimenez M, Lim PR, Davey RA, Zajac JD, Illingworth P, Handelsman DJ. Female mice haploinsufficient for an inactivated androgen receptor (AR) exhibit age-dependent defects that resemble the AR null phenotype of dysfunctional late follicle development, ovulation, and fertility. Endocrinology. 2007 Aug;148(8):3674-84.
Weghofer A, Kim A, Barad DH, Gleicher N. The impact of androgen metabolism and FMR1 genotypes on pregnancy potential in women with dehydroepiandrosterone (DHEA) supplementation. Hum Reprod. 2012 Nov;27(11):3287-93.
Weiss EP, Shah K, Fontana L, Lambert CP, Holloszy JO, Villareal DT. Dehydroepiandrosterone replacement therapy in older adults: 1- and 2-y effects on bone. Am J Clin Nutr. 2009 May;89(5):1459-67.
Weissman A, Horowitz E, Ravhon A, Golan A, Levran D. Dehydroepiandrosterone supplementation increases baseline follicular phase progesterone levels. GynecolEndocrinol. 2011 Dec;27(12):1014-7.
Wiser A, Gonen O, Ghetler Y, Shavit T, Berkovitz A, Shulman A. Addition of dehydroepiandrosterone (DHEA) for poorresponder patients before and during IVF treatment improves the pregnancy rate: a randomized prospective study. Hum Reprod. 2010 Oct;25(10):2496-500.
Yeung TW, Li RH, Lee VC, Ho PC, Ng EH. A Randomized Double-Blinded Placebo-Controlled Trial on the Effect of Dehydroepiandrosterone for 16 Weeks on Ovarian Response Markers in Women with Primary Ovarian Insufficiency. J ClinEndocrinolMetab. 2012 Nov 8. [Epub ahead of print]