Part 1 — Pycnogenol® & Menopause Series
Why non-hormonal support matters now
By 2025, more than one billion women will be in menopause or transitioning toward it, most between ages 45–55, with symptoms that can persist for years [1–3]. Vasomotor symptoms—hot flashes and spontaneous sweats—affect up to 80% of perimenopausal women and commonly disturb sleep and mood, with downstream impacts on fatigue, memory and productivity [3–6]. While menopausal hormone therapy can be effective, it carries risks (e.g., venous thromboembolism, stroke, and breast cancer) that prompt many women to seek non-hormonal options [7].
What is Pycnogenol®?
Pycnogenol® is a standardized extract of French maritime pine bark with antioxidant and anti-inflammatory activity. It has been studied across multiple health areas relevant to menopause without affecting sex-hormone levels [8–10].
Clinical signal: symptom relief without altering hormones
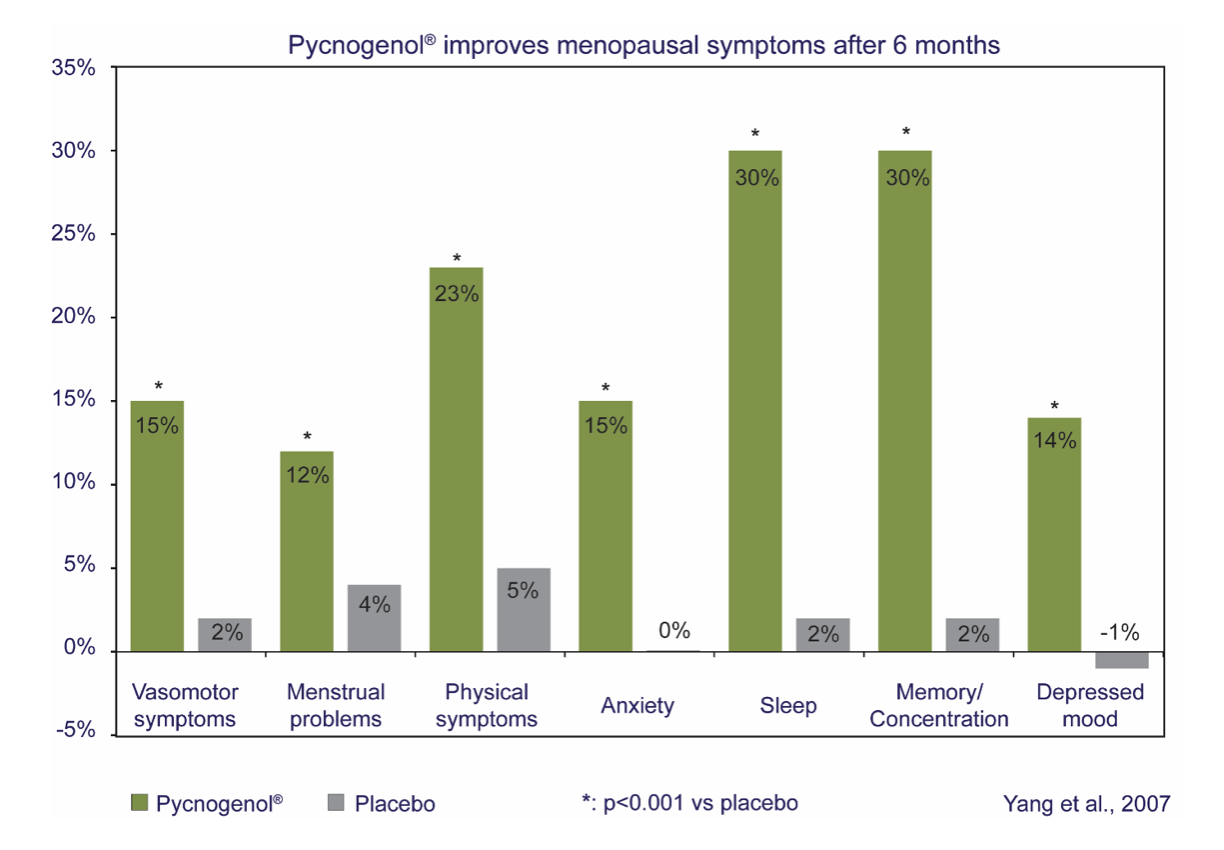
- In a 6-month randomized, double-blind, placebo-controlled trial in 155 peri-menopausal women, Pycnogenol® significantly improved all domains of the Women’s Health Questionnaire (WHQ)—including vasomotor, sleep, mood, cognition, attractiveness and sexual behaviour—versus placebo [10].
- In a study of 70 women, 100 mg/day for 8 weeks significantly improved 33 common menopausal complaints (hot flashes, night sweats, mood swings, libido, vaginal dryness) versus controls [11].
- A 3-month RCT (n=170) using 60 mg/day reduced the total menopause symptom score by 17% versus placebo. Importantly, measured sex-hormone levels did not change from baseline or versus placebo, supporting non-hormonal action [9].
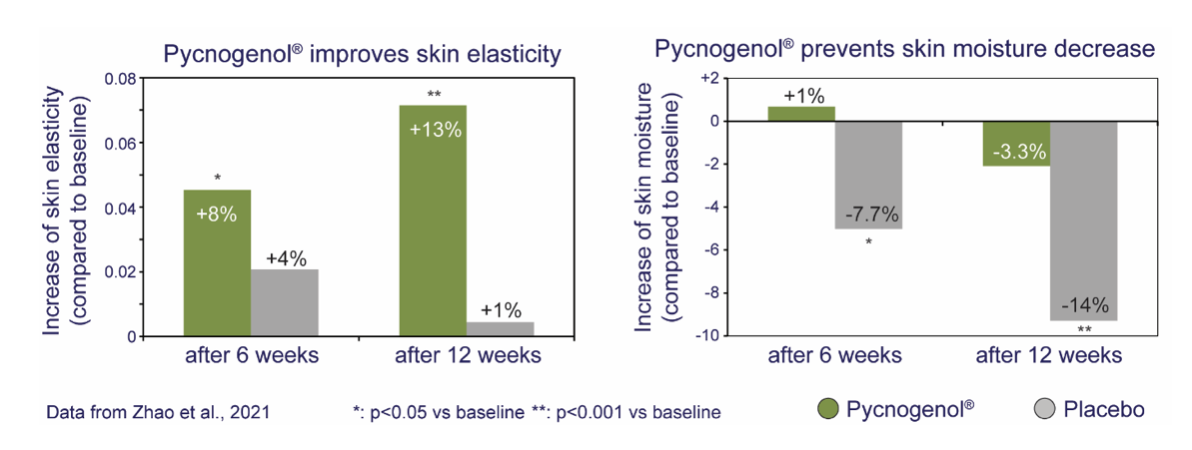
- Another 8-week study reported improvements in vasomotor symptoms, vaginal health, cognition and sleep versus baseline and control [12].
- A 90-day comparative study in 90 postmenopausal women found greater improvements in psychological, vasomotor and sexual symptoms with Pycnogenol® than with two other botanicals commonly used for menopause [13].
How it may work
Pycnogenol® supports antioxidant balance and inflammatory signalling, areas thought to modulate vasomotor symptoms and overall well-being during the transition [8–10].
Practical notes for retailers and clinicians
- Research ranges: 60–100 mg/day for 8–24 weeks are commonly studied doses [9–12].
- Positioning: For women seeking non-hormonal, evidence-based symptom support, including those not candidates for or declining hormone therapy.
- Counsel: Pair with sleep hygiene, balanced nutrition and physical activity for multi-modal support.
This article is for educational purposes only and is not a substitute for individual medical advice.
References: 1–13 as cited in source list.
References:
1. United Nations, Department of Economic and Social Affairs Population Division ,World
Population Prospects 2022 2023 [Available from:
https://population.un.org/wpp/
2. McKinlay SM, Brambilla DJ, Posner JG. The normal menopause transition. Maturitas.
1992;14(2):103-15.
3. Nappi RE, Siddiqui E, Todorova L, Rea C, Gemmen E, Schultz NM. Prevalence and quality-of-life burden of vasomotor symptoms associated with menopause: A European cross-sectional survey.
Maturitas. 2023;167:66-74.
4. Wu JM, Zelinski MB, Ingram DK, Ottinger MA. Ovarian Aging and Menopause: Current
Theories, Hypotheses, and Research Models. Experimental Biology and Medicine. 2005;230(11):818-28.
5. Woods NF, Mitchell ES. Symptoms during the perimenopause: prevalence, severity,
trajectory, and significance in women's lives. Am J Med. 2005;118 Suppl 12B:14-24.
6. Richters JMA. Menopause in Different Cultures. Journal of Psychosomatic Obstetrics & Gynecology. 1997;18(2):73-80.
7. Beral V, Peto R , Pirie K , Reeves G. Type and timing of menopausal hormone therapy and breast cancer risk: individual participant meta-analysis of the worldwide epidemiological evidence.
Lancet. 2019;394(10204):1159-68.
8. Oliff H. American Botanical Council – Scientific and clinical Monograph for Pycnogenol. ABC. 2019:1-46.
9. Kohama T, Negami M. Effect of low-dose French maritime pine bark extract on climacteric syndrome in 170 perimenopausal women: a randomized, double-blind, placebo-controlled trial. J Reprod Med. 2013;58(1-2):39-46.
10. Yang HM, Liao MF, Zhu SY, Liao MN, Rohdewald P. A randomised, double-blind, placebo-
controlled trial on the effect of Pycnogenol on the climacteric syndrome in peri-menopausal women. Acta Obstet Gynecol Scand. 2007;86(8):978-85.
11. Errichi S, Bottari A, Belcaro G, Cesarone MR, Hosoi M, Cornelli U, et al. Supplementation with Pycnogenol® improves signs and symptoms of menopausal transition. Panminerva Med. 2011;53(3 Suppl 1):65-70.
12. Luzzi R, Belcaro G, Hosoi M, Feragalli B, Cornelli U, Dugall M, et al. Normalization of
cardiovascular risk factors in peri-menopausal women with Pycnogenol(R). Minerva Ginecol.
2017;69(1):29-34.
13. Cesarone MR, Belcaro G, Cornelli U, Hosoi M, Scipione V, Scipione C, et al. Effects of
Pycnogenol® in women with postmenopausal symptoms: a 90-day comparative study. Panminerva Med. 2023.
14. Hildreth KL, Ozemek C, Kohrt WM, Blatchford PJ, Moreau KL. Vascular dysfunction across the stages of the menopausal transition is associated with menopausal symptoms and quality of life. Menopause. 2018;25(9):1011-9.
15. Moreau KL, Hildreth KL. Vascular Aging across the Menopause Transition in Healthy Women. Adv Vasc Med. 2014;2014.
16. Enseleit F, Sudano I, Periat D, Winnik S, Wolfrum M, Flammer AJ, et al. Effects of Pycnogenol on endothelial function in patients with stable coronary artery disease: a double-blind, randomized, placebo-controlled, cross-over study. Eur Heart J. 2012;33(13):1589-97.
17. Liu X, Wei J, Tan F, Zhou S, Wurthwein G, Rohdewald P. Pycnogenol, French maritime pine bark extract, improves endothelial function of hypertensive patients. Life Sci. 2004;74(7):855-62.
18. Zibadi S, Rohdewald PJ, Park D, Watson RR. Reduction of cardiovascular risk factors in
subjects with type 2 diabetes by Pycnogenol supplementation. Nutr Res. 2008;28(5):315-20.
19. Fitzpatrick DF, Bing B, Rohdewald P. Endothelium-dependent vascular effects of Pycnogenol.
J Cardiovasc Pharmacol. 1998;32(4):509-15.
20. Nishioka K, Hidaka T, Nakamura S, Umemura T, Jitsuiki D, Soga J, et al. Pycnogenol, French maritime pine bark extract, augments endothelium-dependent vasodilation in humans. Hypertens Res. 2007;30(9):775-80.
21. Conde DM, Verdade RC, Valadares ALR, Mella LFB, Pedro AO, Costa-Paiva L. Menopause and cognitive impairment: A narrative review of current knowledge. World J Psychiatry. 2021;11(8):412-28.
22. Hosoi M, Belcaro G, Saggino A, Luzzi R, Dugall M, Feragalli B. Pycnogenol(R) supplementation
in minimal cognitive dysfunction. J Neurosurg Sci. 2018;62(3):279-84.
23. Cesarone MR, Belcaro G, Hosoi M, Ledda A, Feragalli B, Maione C, et al. Supplementary
management with Pycnogenol® in Parkinson's disease to prevent cognitive impairment. J Neurosurg Sci. 2020;64(3):258-62.
24. Belcaro G DM, Ippolito E, Hu S, Saggino A, Feragalli B Improvement in cognitive function, attention, mental performance with Pycnogenol® in healthy subjects (55-70) with high oxidative stress. J Neurosurg Sci 2015;59:437-46.
25. Ryan J, Croft, K., Wesnes, K and Stough, C. An examination of the effects of the antioxidant Pycnogenol® on cognitive performance, serum lipid profile, endocrinological and oxidative stress biomarkers in an elderly population. J Psychopharmacol. 2008;22(5):553-62.
26. Neogi T, Zhang Y. Epidemiology of osteoarthritis. Rheum Dis Clin North Am. 2013;39(1):1-19.
27. Jessberger S, Högger P, Genest F, Salter DM, Seefried L. Cellular pharmacodynamic effects of Pycnogenol(R) in patients with severe osteoarthritis: a randomized controlled pilot study. BMC Complement Altern Med. 2017;17(1):537.
28. Canali R, Comitato R, Schonlau F, Virgili F. The anti-inflammatory pharmacology of
Pycnogenol in humans involves COX-2 and 5-LOX mRNA expression in leukocytes. Int
Immunopharmacol. 2009;9(10):1145-9.
29. Grimm T, Chovanova Z, Muchova J, Sumegova K, Liptakova A, Durackova Z, et al. Inhibition of NF-kappaB activation and MMP-9 secretion by plasma of human volunteers after ingestion of maritime pine bark extract (Pycnogenol). J Inflamm (Lond). 2006;3:1.
30. Grimm T, Schäfer A, Högger P. Antioxidant activity and inhibition of matrix
metalloproteinases by metabolites of maritime pine bark extract (pycnogenol). Free Radic Biol Med. 2004;36(6):811-22.
31. Schäfer A, Chovanova Z, Muchova J, Sumegova K, Liptakova A, Durackova Z, et al. Inhibition of COX-1 and COX-2 activity by plasma of human volunteers after ingestion of French maritime pine bark extract (Pycnogenol). Biomed Pharmacother. 2005;60(1):5-9.
32. Belcaro G, Cesarone MR, Errichi S, Zulli C, Errichi BM, Vinciguerra G, et al. Variations in C-reactive protein, plasma free radicals and fibrinogen values in patients with osteoarthritis treated with Pycnogenol. Redox Rep. 2008;13(6):271-6.
33. Farid R, Mirfeizi Z, Mirheidari M, Rezaieyazdi Z, Mansouri H, Esmaelli H, et al. Pycnogenol supplementation reduces pain and stiffness and improves physical function in adults with knee osteoarthritis. Nutrition Research. 2007;27(11):692-7.
34. Cisar P, Jany R, Waczulikova I, Sumegova K, Muchova J, Vojtassak J, et al. Effect of pine bark extract (Pycnogenol) on symptoms of knee osteoarthritis. Phytother Res. 2008;22(8):1087-92.
35. Belcaro G, Cesarone MR, Errichi S, Zulli C, Errichi BM, Vinciguerra G, et al. Treatment of
osteoarthritis with Pycnogenol. The SVOS (San Valentino Osteo-arthrosis Study). Evaluation of signs,
symptoms, physical performance and vascular aspects. Phytother Res. 2008;22(4):518-23.
36. Mülek M, Seefried L, Genest F, Högger P. Distribution of Constituents and Metabolites of
Maritime Pine Bark Extract (Pycnogenol((R))) into Serum, Blood Cells, and Synovial Fluid of Patients with Severe Osteoarthritis: A Randomized Controlled Trial. Nutrients. 2017;9(5).
37. Zhao H, Wu J, Wang N, Grether-Beck S, Krutmann J, Wei L. Oral Pycnogenol(R) Intake Benefits the Skin in Urban Chinese Outdoor Workers: A Randomized, Placebo-Controlled, Double-Blind, and Crossover Intervention Study. Skin Pharmacol Physiol. 2021:1-11.
38. Marini A, Grether-Beck S, Jaenicke T, Weber M, Burki C, Formann P, et al. Pycnogenol(R) effects on skin elasticity and hydration coincide with increased gene expressions of collagen type I and hyaluronic acid synthase in women. Skin Pharmacol Physiol. 2012;25(2):86-92.
39. Belcaro G, Cesarone MR, Errichi BM, Ledda A, Di Renzo A, Stuard S, et al. Diabetic ulcers: microcirculatory improvement and faster healing with pycnogenol. Clin Appl Thromb Hemost. 2006;12(3):318-23.
40. Ayres EL, Silva JDS, Eberlin S, Facchini G, Vasconcellos C, Costa A. In-vitro effect of pine bark extract on melanin synthesis, tyrosinase activity, production of endothelin-1 and PPAR in cultured melanocytes exposed to Ultraviolet, Infrared, and Visible light radiation. J Cosmet Dermatol. 2021.
41. Grether-Beck S, Marini A, Jaenicke T, Krutmann J. French Maritime Pine Bark Extract
(Pycnogenol(R)) Effects on Human Skin: Clinical and Molecular Evidence. Skin Pharmacol Physiol.2016;29(1):13-7.
42. Pinto CAS, Delfes MFZ, Reis LMd, Garbers LE, Passos PCVdR, Torre DSd. The use of
pycnogenol in the treatment of melasma. Surgical & Cosmetic Dermatology.2015;7(3).
43. Ni Z, Mu Y, Gulati O. Treatment of melasma with Pycnogenol. Phytother Res. 2002;16(6):567-71.
44. Saliou C, Rimbach G, Moini H, McLaughlin L, Hosseini S, Lee J, et al. Solar ultraviolet-induced erythema in human skin and nuclear factor-kappa-B-dependent gene expression in keratinocytes are modulated by a French maritime pine bark extract. Free Radic Biol Med. 2001;30(2):154-60.
45. Kim YJ, Kang KS, Yokozawa T. The anti-melanogenic effect of pycnogenol by its anti-oxidative actions. Food Chem Toxicol. 2008;46(7):2466-71.
46. Kwon SH, Na JI, Choi JY, Park KC. Melasma: Updates and perspectives. Experimental
Dermatology. 2019;28(6):704-8.
47. Zouboulis CC, Blume-Peytavi U, Kosmadaki M, Roó E, Vexiau-Robert D, Kerob D, et al. Skin, hair and beyond: the impact of menopause. Climacteric. 2022;25(5):434-42.
48. Chaikittisilpa S, Rattanasirisin N, Panchaprateep R, Orprayoon N, Phutrakul P, Suwan A, et al. Prevalence of female pattern hair loss in postmenopausal women: a cross-sectional study. Menopause. 2022;29(4):415-20.
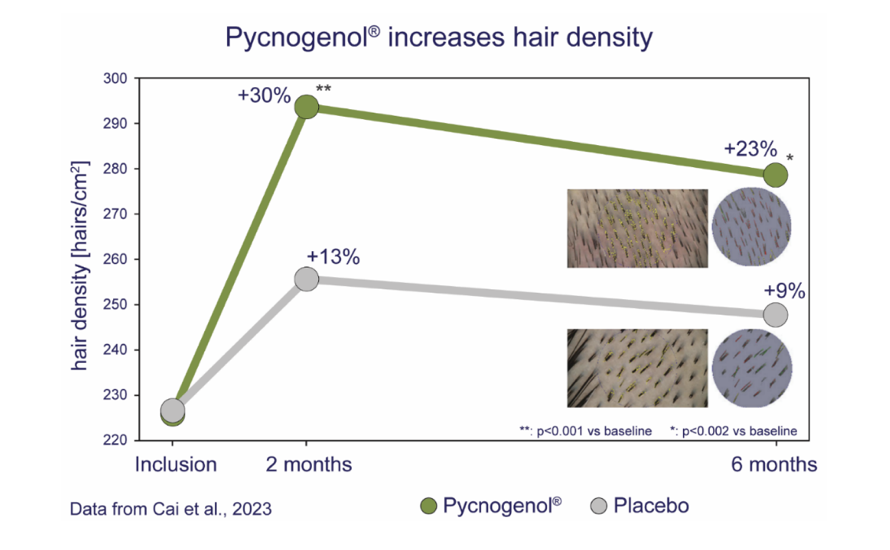
49. Cai C, Zeng B, Lin L, Zheng M, Burki C, Grether‐Beck S, et al. An oral French maritime pine bark extract improves hair density in menopausal women: A randomized, placebo‐controlled, double blind intervention study. Health Science Reports. 2023;6(1).
50. Cesarone MR, Belcaro G, Agus GB, Ippolito E, Dugall M, Hosoi M, et al. Chronic venous
insufficiency and venous microangiopathy: management with compression and Pycnogenol(R). Minerva Cardioangiol. 2019;67(4):280-7.
51. Steigerwalt R, Belcaro G, Cesarone MR, Di Renzo A, Grossi MG, Ricci A, et al. Pycnogenol improves microcirculation, retinal edema, and visual acuity in early diabetic retinopathy. J Ocul Pharmacol Ther. 2009;25(6):537-40.
52. Wang S, Tan D, Zhao Y, Gao G, Gao X, Hu L. The effect of Pycnogenol® on the
microcirculation, platelet function and ischaemic myocardium in patients with coronary artery diseases. European Bulletin of Drug Research. 1999;7(2):19-25.
53. Cesarone MR, Belcaro G, Rohdewald P, Pellegrini L, Ledda A, Vinciguerra G, et al.
Improvement of diabetic microangiopathy with pycnogenol: A prospective, controlled study.
Angiology. 2006;57(4):431-6.
54. Luzzi R BG, Hu S, Dugall M, Hosoi M, Cacchio M, Ippolito E, Corsi M Improvement in
symptoms and cochlear flow with Pycnogenol in patients with Meniere’s disease and tinnitus. Minerva Med 2014;105:245-54.
55. Grossi MG, Belcaro G., Cesarone, M.R., Duggall, M., Hosoi, M Cacchio,. Improvement in
cochlear flow with Pycnogenol® in patients with tinnitus: a pilot evaluation. Panminerva Med. 2010;52(2):63-7.
56. Chovanova Z, Muchova J, Sivonova M, Dvorakova M, Zitnanova I, Waczulikova I, et al. Effect of polyphenolic extract, Pycnogenol, on the level of 8-oxoguanine in children suffering from attention deficit/hyperactivity disorder. Free Radic Res. 2006;40(9):1003-10.
57. D̆uračková Z, Trebatický B, Novotný V, Žitňanová I, Breza J. Lipid metabolism and erectile function improvement by pycnogenol®, extract from the bark of pinus pinaster in patients suffering from erectile dysfunction-a pilot study. Nutrition Research. 2003;23(9):1189-98.
58. Devaraj S V-LS, Kaul N, Schönlau F, Rohdewald P, Jialal I. Supplementation with a pine bark extract rich in polyphenols increases plasma antioxidant capacity and alters plasma lipoprotein profile. Lipids. 2002;37(10):931-4.
59. Kolacek M, Muchova J, Dvorakova M, Paduchova Z, Zitnanova I, Cierna I, et al. Effect of
natural polyphenols (Pycnogenol) on oxidative stress markers in children suffering from Crohn's disease–a pilot study. Free Radic Res. 2013;47(8):624-34.
60. Errichi S, Bottari, A., Belcaro, G., Cesarone, M.R., Hosoi, M., Cornelli, U., Dugall, M., Ledda, A., Feragalli, B. Supplementation with Pycnogenol® improves signs and symptoms of menopausal transition. Panminerva Med. 2011;53(3):65-70.
61. Al-Badr A, Al-Shaikh G. Recurrent Urinary Tract Infections Management in Women: A review. Sultan Qaboos Univ Med J. 2013;13(3):359-67.
62. Lüthje P, Brauner H, Ramos NL, Ovregaard A, Gläser R, Hirschberg AL, et al. Estrogen supports urothelial defense mechanisms. Sci Transl Med. 2013;5(190):190ra80.
63. Cotellese R, Hu S, Cesarone MR, Belcaro G, Dugall M, Feragalli B, et al. Pycnogenol®
supplementation prevents inflammation and symptoms in recurrent, non-severe urinary infections. Panminerva Med. 2021;63(3):343-8.
64. Ledda A, Hu S, Cesarone MR, Belcaro G, Dugall M, Feragalli B, et al. Pycnogenol®
Supplementation Prevents Recurrent Urinary Tract Infections/Inflammation and Interstitial Cystitis. Evidence-Based Complementary and Alternative Medicine. 2021;2021:9976299.