Mitochondrial Rescue
Turning cancer cells off
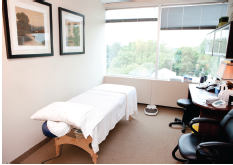
Professor Otto Warburg of the Max Planck Institute won a Nobel prize for his discoveries of how the human body uses sugars in health and in disease. In his seminal work ‘On the Origin of Cancer Cells” published in 1956 he described the “irreversible” damage to respiration as a metabolic process in malignant cells, resulting in a shift to anaerobic glycolysis. Observing well oxygenated cancer cells with abnormally high glycolysis and lactate production suggested to him that defects in mitochondrial (“grana”) functions are at the heart of the transformation of normal cells to malignant cells (Warburg 1956).
The “Grana” of Warburg are today called mitchondria. Of note is the fact that mitochondrial DNA comes from your mother’s egg, and contains no DNA from your father, whereas the nuclear DNA is a mix from both parents. The most important consequence of mitochondria incurring compromised oxidative capacity is loss of programmed cell death , or the apoptosis “off-switch” for old or mutated cells. This is a process that allows a cell to shut down and be recycled after 50 doublings, or earlier if the nuclear DNA becomes mutated and damaged. It prevents making two bad cells from an old or damaged cell. The cell membranes turn inside out, the cell is recycled, and a local stem cell wakes up out of dormancy to make a fresh new cell with the counter reset to zero. As we lose the ability to weed out bad cells, the potential for mutations that lead to cancer increase.
Cancer cells with mitochondrial failure become very hard to kill, and will continue to accrue mutations which make the cancer more invasive, hypermetabolic, and adaptable to metastatic environments. It is the restoration of this apoptotic switch by the oxidative stress of chemotherapy and radiation which make these the most widely applied and generally successful therapies for most cancers, which otherwise have quite different biology.
Warburg’s hypothesis is often misinterpreted to support the idea that oxygen is cytotoxic to cancer, and cancer cannot arise in a high-oxygen environment. It is also taken to imply that cancers originate only in an acid-environment, and that alkalizing is an effective cytoxic therapy for cancer. These myths have failed to unlock the potential for targeting mitochondria to cure cancer suggested by this seminal work (Wenzel 2004).
Mitochondria of tumour cells lose their ability to perform oxidative phosphorylation. Cellular energy metabolism shifts to anaerobic glycolysis.
Early on in the growth of a tumor, at about 2 mm in diameter, oxygen depletion begins to occur in the center of these cell clusters. Oxygen doesn’t freely diffuse this far into tissues, and the hyper-metabolic nature of cancer cells depletes what is there at an abnormal rate. The hypoxic cells send out distress signals, summoning immune and regulatory cells to reestablish blood inflow by neo-angiogenesis. A chronic inflammation repair process is created, but unable to fix the genetic problems of the transformed cells, it becomes “the wound that will not heal”. Growth factors and other immune cell products support continued tumor growth, and any immune-cell going into attack mode against cancer cells are shut off by cytokines such as transforming growth factor (TGF-B1) (Talmadge 2007). The immune system is now working for the cancer. They can even cannabilize fibroblasts and other cells to give spare parts to the suffering cancer cells (Martinez-Outschoorn 2010.
The new blood vessels in tumors do not remodel into efficient capillary beds, as is expected in normal tissue repair. As cancerous tumors mature, leakiness of the blood vessels creates a build-up of fluid pressure. Oncotic pressure eventually crushes the vessels, creating pockets of severely low oxygen, called hypoxia. If the oxygen tension falls to zero, called anoxia, like any human cell, the cancer cells will die. Sub-lethal hypoxia can cause mitochondria membranes to become hyper- polarized, and oxidative metabolism shuts off (Bonnet 2007). The cancer cell adapts by starting to process sugars into energy without oxygen, through the whole of the cell cytoplasm. This anaerobic glycolysis creates lactic acid as a waste product, and at this point the cancers become highly acidic (Warburg 1956).
This fermentation of sugars, very similar to how yeast generate energy, is not very efficient at creating ATP. This may on the surface appear to create a disadvantage for the cancer cell, but in fact it creates an abundance of building blocks for proteins, fatty acids and nucleic acids desperately needed by the cancer cells to maintain their overly-rapid growth rate (Bui 2006).
Most of the anabolic processes required for accelerated growth rate are accomplished by increased glycolysis. Anaerobic glycolysis is supported by replenishing TCA cycle intermediates, such as acetyl-CoA and citrate, a process called anaplerosis (DeBerardinis 2008, Kroemer 2008, Ramos- Montoya 2006). Anaplerosis sustains TCA cycle function by either converting pyruvate to oxaloacetate or by breaking down glutamine into alphaketoglutarate by glutaminolysis (DeBerardinis 2007).
The critical shift between glycolysis and oxidative phosphorylation is controlled by the relative activities of two enzymes, pyruvate dehydrogenase (PDH) and lactate dehydrogenase (LDH), that determine the catabolic fate of pyruvate. Hypoxia inducible factor (HIF) can induce LDH activity while inhibiting PDH activity by stimulating its inhibitor PDK1 (Kim 2006, Wigfield 2008). Hypoxia results in hydrogen peroxide (H2O2) and nitric oxide (NO) generation, which in turn results in hydroxyl radicals and super oxides. Jurasunas postulated that this scenario results in depletion of the potent antioxidant manganese-super oxide dismutase MnSOD, which destroys the respiratory chain in hypoxic mitochondria (Jurasunas 2006).
As free radicals of oxygen accumulate there is an excessive uptake of lactate and pyruvate in the hypoxic mithochondria. This raises the mitochondrial membrane potential Delta -Psi-m (ΔΨm). Cancer cells thereby have hyperpolarized mitochondrial membranes compared to normal cells, preventing them from throwing the apoptotic off-switch no matter how old or mutated they become. Hexokinase II is highly expressed in cancer cells, induced by alterations to the Ras-P13-Akt signal transduction pathway. Hexokinase binds to mitochondrial porin, suppressing the calcium channels and the potassium channel K+v1.5 (Lemasters 2006, Wallace 2005).
Lactate dehydrogenase is upregulated in tumors, favouring cancer growth via VEGF and HIF-1α, increased angiogenesis, and metastasis via direct enhancement of cellular motility (Gottfried 2006, Koukourakis 2005, Koukourakis 2006, Kulawiec 2009, Ordys 2010, Pelicano 2006, Ralph 2010, Seth 2011, Walenta 2004, Walenta 2000). A build-up of the electron transport molecule NADH inactivates the vital tumor suppressor gene PTEN. This activates Akt protein kinase B survival pathway, resulting in immortalization of the cancer cell (Frezza 2009).
Strategies that successfully allow mitochondria in tumour cells to once again perform oxidative phosphorylation have proven to be potent anti tumour agents.
In 2007 Bonnet, Michelakis and group at the University of Alberta tested an old drug dichloroacetate or DCA on breast cancer infested rats, and made an astonishing discovery. Based on a hypothesis that inhibiting a mitochondrial enzyme pyruvate dehydrogenase kinase or PDK might repolarize mitochondrial membranes and spark up respiration in cancer cells, they found evidence DCA might do the job, and it did indeed reverse what Warburg deemed irreversible (Bonnet 2007).
An important line in the abstract states “DCA induces apoptosis, decreases proliferation and inhibits tumor growth, without apparent toxicity.” This may well apply to the rats in this study, but clear evidence of neurotoxicity and other risks to humans existed before this trial. In fact, human studies on DCA for diabetic acidosis had shown this drug was far from safe. DCA had long been listed as an environmental toxin and carcinogen by the USA Environmental Protection Agency. Release of DCA into the environment was a crime (Backshear 1975, Felitsyn 2007, Kaufmann 2006, Schaefer 2006). This impression of safety was compounded by an article that soon followed in the journal New Scientist claiming this drug was safe, and effective for most cancers (Coghlan 2007).
In the Globe& Mail June 2, 2007, Mikelakis warned of “severe nerve damage. People may lose their ability to walk, or speak” from impurities in DCA bought from sources such as the internet. Others reported nausea, drowsiness and peripheral neuropathy in DCA users (Kaufmann 2006).
There is no question DCA can reduce human tumors. DCA activates private dehydrogenase kinase, triggering an influx of acetyl-CoA into mitochondria. This drives more NADH into complex I. Superoxides that form are converted into hydrogen peroxide by manganese- super oxide dismutase. The H2O2 inhibits proton (H+) efflux, reducing mitochondrial membrane potential Δψm. This opens the mitochondrial transition pore (MTP), inhibiting calcium ion entry via voltage-dependent channels. Reduced intra-mitochondrial calcium (Ca++) suppresses a tonic activation of nuclear factor of activated T lymphocytes (NFAT). NFAT1 is a nuclear transcription activator similar in action to activator protein 1 (AP-1) and nuclear factor kappa B (NFκB). This reduces Kv1.5 expression, increasing potassium ion K+ efflux, reducing inhibition of caspases, and finally triggering cancer cell apoptosis (Bonnet 2007).
An interesting side-note is the fact that DCA converts into glyoxalate, an old remedy once promoted for cancer (Biswas 1997, Herbert 1979, Ray 1997). Victor Herbert quotes the leading DCA researcher Dr. Stacpoole, as saying “ the efficacy and safety of chronic dichloroacetate administration is unknown” (Stacpoole 1998).
I saw the toxicity of DCA on the nervous system for cancer patients who tried this experimental approach. I have seen cases completely disabled by the drug. Because the DCA was offpatent, it took Michelakis many months to fund a human trial. Two of my patients went to Edmonton and were enrolled, but returned to Victoria in about two weeks with severe neuropathy. As a result of this experience, the investigators wisely reduced the dose to 1/3 that level, and restricted its application to CNS tumors. The result was brain tumor responses with “acceptable toxicity” (Michelkakis 2010).
Does there exist a less toxic alternative for activating oxidative phosphorylation in mitochondria of tumour cells?
In 2007 I recognized the potential of this approach, awakening mitochondrial apoptosis switching through inhibition of PDK, but also saw the clinical limitations of the DCA drug. I began a search for a less harmful PDK inhibitor. I was able to find evidence that R+ alpha lipoid acid and thiamine (vitamin B-1) could do this (McKinney 2008). As we have come to expect with all natural and non-toxic approaches to cancer, single agents are rarely very potent. John Boik’s excellent texts have pointed out the need to find synergistic groupings of botanicals and nutraceuticals, if we expect to create significant impact on advanced cancers (Boik 1996, Boik 2001). I can say that the clinical application of my original mitochondrial rescue plan has produced responses in cancers, including rather difficult ones such as sarcoma and lung cancer. It is certainly in need of further refinement. I do not presently see it being useful in lymphomas and leukemias. These are the natural medicines I propose for mitochondrial rescue of cancer cells, to reverse the Warburg Effect:
R+ alpha lipoic acid is a potent natural PDK inhibitor (Hagen 1999, Hagen 2002, Korotchina 2004, Li 2009, Liu 2002, Moungjaroen 2006, Simbula 2007, Wenzel 2005a). It is used intravenously and orally for cancer, and has the added benefit of treating neuropathy.
The only other natural compound which is known to be a direct inhibitor of PDK is thiamin, or vitamin B1. Because it is fat soluble and thought to integrate better into mitochondrial membranes, we favor clinical use of benfotaimine (Babaei-Jadidi 2003, Parkhomenko 1987).
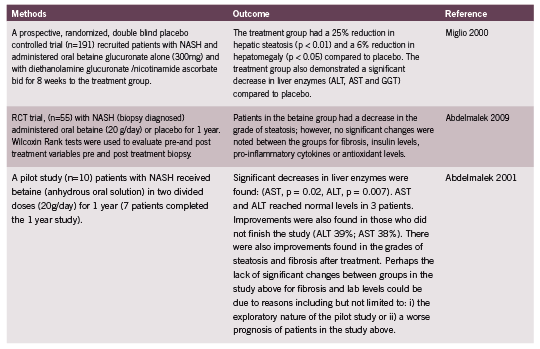
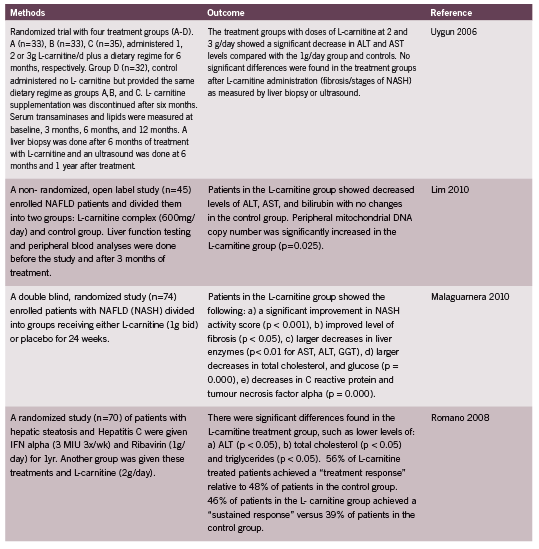
Some believe L-carnitine is key to mitochondrial restoration (Cruciani 2006, Hoang 2007, Wenzel 2005b), but it is not a PDK inhibitor, and will generate enough free radicals of oxygen to damage mitochondria further, unless accompanied by sufficient fat soluble antioxidants such as R+ ALA. In fact, the two supplements have an excellent synergy and should be given together (Hagen 2002, McMackin 2007). My clinical preference is prescription of acetyl-L-carnitine, which is fat soluble.
Co-enzyme Q-10 is widely recognized as a fat-soluble antioxidant that is clinically useful for mitochondrial disorders (Beal 1994, Berbel- Garcia 2004, Matthews 1998, Perumal 2005a, Rodriguez 2007). However, it can inhibit opening of the mitochondrial transition pore, which could counteract the DCA effect (Li 2005). My clinical experience suggests grapeseed extract is a better choice (Hu 2006).
B-vitamins are involved in energy metabolism as co-factors for many important enzymes. A B-complex seems a reasonable support for mitochondria dysfunction (Perumal 2005a, Perumal 2005b).
Indole-3-carbinol has been a most useful cancer therapy as a STAT-3 transcription factor inhibitor, and for its anti-estrogen effects. It has a potent effect on upregulation of bax in mitochondria, causing membrane depolarization and activation of apoptotic caspases (Rahman 2000, Rahman 2003).
Quercitin can increase or decrease mitochondrial membrane potential Delta Psim (Δψm) depending on concentration, inducing apoptosis (Kellner 2004, Kothan 2004, Yang 2006, Zhang 2005). This versatile anti-cancer agent interferes with glycolysis via reduced generation of glycolytic substrates adenosine diphosphate and inorganic phosphate (Suolinna 1975).
Gamma tocopherol is a dietary form of vitamin E that can stabilize mitochondrial membranes. It can be washed out of membranes by intake of excess alpha-tocopherol supplements. Always use the “mixed tocopherols” containing the gamma form, never just the “natural source” d-alpha tocopherol (Mahabir 2008).
Fish oils are an excellent omega 3 oil for mitochondrial membrane health, reducing mitochondria calcium levels (Hansford 1999).
Other adjuncts for mitochondrial regeneration include intense aerobic exercise (Lanza 2010), calorie restricted diets (Spindler 2010), and the diabetic drug Metformin (Suwa 2006)
References:
Babaei-Jadidi R, Karachalias N, Ahmed N, Battah S, Thornalley PJ. Prevention of incipient diabetic nephropathy by high-dose thiamine and benfotiamine. Diabetes. 2003 Aug;52(8):2110-20.
Backshear PJ, Holloway PA, Alberti KG. Metabolic interactions of dichloroacetate and insulin in experimental diabetic ketoacidosis. Biochem J. 1975 Feb;146(2):447-56.
Beal MF, Henshaw DR, Jenkins BG, Rosen BR, Schulz JB. Coenzyme Q10 and nicotinamide block striatal lesions produced by the mitochondrial toxin malonate. Ann Neurol. 1994 Dec;36(6):882-8.
Berbel-Garcia A, Barbera-Farre JR, Etessam JP, Salio AM, Cabello A, Gutierrez-Rivas E, Campos Y. Coenzyme Q 10 improves lactic acidosis, strokelike episodes, and epilepsy in a patient with MELAS (mitochondrial myopathy, encephalopathy, lactic acidosis, and strokelike episodes). Clin Neuropharmacol. 2004 Jul-Aug;27(4):187-91.
Biswas S, Ray M, Misra S, Dutta DP, Ray S. Selective inhibition of mitochondrial respiration and glycolysis in human leukaemic leucocytes by methylglyoxal. Biochem J. 1997 Apr 15;323 ( Pt 2):343-8.
Boik J. Cancer & Natural Medicine – a Textbook of Basic Science and Clinical Research. 1996. Oregon Medical Press.
Boik J. Natural Compounds in Cancer Therapy – Promising Non-toxic Anti-tumor Agents from Plants and Other Natural Sources. 2001. Oregon Medical Press.
Bonnet S, Archer SL, Allalunis-Turner J, Haromy A, Beaulieu C, Thompson R, Lee CT, Lopaschuk GD, Puttagunta L, Bonnet S, Harry G, Hashimoto K, Porter CJ, Andrade MA, Thebaud B, Michelakis ED. A mitochondria-K+ channel axis is suppressed in cancer and its normalization promotes apoptosis and inhibits cancer growth. Cancer Cell. 2007 Jan;11(1):37-51.
Bui T, Thompson CB. Cancer’s sweet tooth. Cancer Cell. 2006 Jun;9(6):419-20.
Coghlan. Cheap, Safe Drug Kills Most Cancers. New Scientist; January 2007.
Cruciani RA, Dvorkin E, Homel P, Malamud S, Culliney B, Lapin J, Portenoy RK, Esteban-Cruciani N. Safety, tolerability and symptom outcomes associated with L-carnitine supplementation in patients with cancer, fatigue, and carnitine deficiency: a phase I/II study. J Pain Symptom Manage. 2006 Dec;32(6):551-9.
DeBerardinis RJ, Lum JJ, Hatzivassiliou G, Thompson CB. The biology of cancer: metabolic reprogramming fuels cell growth and proliferation. Cell Metab. 2008 Jan;7(1):11-20.
DeBerardinis RJ, Mancuso A, Daikhin E, Nissim I, Yudkoff M, Wehrli S, Thompson CB. Beyond aerobic glycolysis: transformed cells can engage in glutamine metabolism that exceeds the requirement for protein and nucleotide synthesis. Proc Natl Acad Sci U S A. 2007 Dec 4;104(49):19345-50.
Felitsyn N, Stacpoole PW, Notterpek L. Dichloroacetate causes reversible demyelination in vitro: potential mechanism for its neuropathic effect. J Neurochem. 2007 Jan;100(2):429-36.
Frezza C., Gottlieb E., Mitochondria in cancer: Not just innocent bystanders, Seminars in Cancer Biology 2009; 19: 4-11.
Gottfried E, Kunz-Schughart LA, Ebner S, Mueller-Klieser W, Hoves S, Andreesen R, Mackensen A, Kreutz M. Tumor-derived lactic acid modulates dendritic cell activation and antigen expression. Blood. 2006 Mar 1;107(5):2013-21.
Hagen TM, Ingersoll RT, Lykkesfeldt J, Liu J, Wehr CM, Vinarsky V, Bartholomew JC, Ames AB. (R)-alpha-lipoic acid-supplemented old rats have improved mitochondrial function, decreased oxidative damage, and increased metabolic rate. FASEB J. 1999 Feb;13(2):411-8.
Hagen TM, Liu J, Lykkesfeldt J, Wehr CM, Ingersoll RT, Vinarsky V, Bartholomew JC, Ames BN. Feeding acetyl-L-carnitine and lipoic acid to old rats significantly improves metabolic function while decreasing oxidative stress. Proc Natl Acad Sci U S A. 2002 Feb 19;99(4):1870-5.
Hagen TM, Moreau R, Suh JH, Visioli F. Mitochondrial decay in the aging rat heart: evidence for improvement by dietary supplementation with acetyl-L-carnitine and/or lipoic acid. Ann N Y Acad Sci. 2002 Apr;959:491-507.
Hansford RG, Tsuchiya N, Pepe S. Mitochondria in heart ischaemia and aging. Biochem Soc Symp. 1999;66:141-7.
Herbert V. Pangamic acid (“vitamin B15”). Am J Clin Nutr. 1979 Jul;32(7):1534-40.
Hoang BX, Graeme Shaw D, Pham P, Levine S. Restoration of cellular energetic balance with L-carnitine in the neuro-bioenergetic approach for cancer prevention and treatment. Med Hypotheses. 2007;69(2):262-72.
Hu H, Qin YM. Grape seed proanthocyanidin extract induced mitochondria-associated apoptosis in human acute myeloid leukaemia 14.3D10 cells. Chin Med J (Engl). 2006 Mar 5;119(5):417-21.
Jurasunas. Mitochondria and Cancer. Townsend Letter. 2006; 83-86: 146-148.
Kaufmann P, Engelstad K, Wei Y, Jhung S, Sano MC, Shungu DC, Millar WS, Hong X, Gooch CL, Mao X, Pascual JM, Hirano M, Stacpoole PW, DiMauro S, De Vivo DC. Dichloroacetate causes toxic neuropathy in MELAS: a randomized, controlled clinical trial. Neurology. 2006 Feb 14;66(3):324-30.
Kellner C, Zunino SJ. Nitric oxide is synthesized in acute leukemia cells after exposure to phenolic antioxidants and initially protects against mitochondrial membrane depolarization. Cancer Lett. 2004 Nov 8;215(1):43-52.
Kim JW, Tchernyshyov I, Semenza GL, Dang CV. HIF-1-mediated expression of pyruvate dehydrogenase kinase: a metabolic switch required for cellular adaptation to hypoxia. Cell Metab. 2006 Mar;3(3):177-85.
Korotchkina LG, Sidhu S, Patel MS. R-lipoic acid inhibits mammalian pyruvate dehydrogenase kinase. Free Radic Res. 2004 Oct;38(10):1083-92.
Kothan S, Dechsupa S, Leger G, Moretti JL, Vergote J, Mankhetkorn S. Spontaneous mitochondrial membrane potential change during apoptotic induction by quercetin in K562 and K562/adr cells. Can J Physiol Pharmacol. 2004 Dec;82(12):1084-90.
Koukourakis MI, Giatromanolaki A, Simopoulos C, Polychronidis A, Sivridis E. Lactate dehydrogenase 5 (LDH5) relates to up-regulated hypoxia inducible factor pathway and metastasis in colorectal cancer. Clin Exp Metastasis. 2005;22(1):25-30.
Koukourakis MI, Giatromanolaki A, Sivridis E, Gatter KC, Harris AL; Tumour Angiogenesis Research Group. Lactate dehydrogenase 5 expression in operable colorectal cancer: strong association with survival and activated vascular endothelial growth factor pathway–a report of the Tumour Angiogenesis Research Group. J Clin Oncol. 2006 Sep 10;24(26):4301-8.
Kroemer G, Pouyssegur J. Tumor cell metabolism: cancer’s Achilles’ heel. Cancer Cell. 2008 Jun;13(6):472-82.
Kulawiec M, Owens KM, Singh KK. Cancer cell mitochondria confer apoptosis resistance and promote metastasis. Cancer Biol Ther. 2009 Jul;8(14):1378-85.
Lanza IR, Sreekumaran Nair K. Regulation of skeletal muscle mitochondrial function: genes to proteins. Acta Physiol (Oxf). 2010 Aug;199(4):529-47.
Lemasters JJ, Holmuhamedov E. Voltage-dependent anion channel (VDAC) as mitochondrial governator—thinking outside the box. Biochim Biophys Acta. 2006 Feb;1762(2):181-90.
Li CJ, Zhang QM, Li MZ, Zhang JY, Yu P, Yu DM. Attenuation of myocardial apoptosis by alpha-lipoic acid through suppression of mitochondrial oxidative stress to reduce diabetic cardiomyopathy. Chin Med J (Engl). 2009 Nov 5;122(21):2580-6.
Li G, Zou LY, Cao CM, Yang ES. Coenzyme Q10 protects SHSY5Y neuronal cells from beta amyloid toxicity and oxygen-glucose deprivation by inhibiting the opening of the mitochondrial permeability transition pore. Biofactors. 2005;25(1-4):97-107.
Liu J, Head E, Gharib AM, Yuan W, Ingersoll RT, Hagen TM, Cotman CW, Ames BN. Memory loss in old rats is associated with brain mitochondrial decay and RNA/DNA oxidation: partial reversal by feeding acetyl-L-carnitine and/or R-alpha -lipoic acid. Proc Natl Acad Sci U S A. 2002 Feb 19;99(4):2356-61.
Mahabir S, Schendel K, Dong YQ, Barrera SL, Spitz MR, Forman MR. Dietary alpha-, beta-, gamma- and delta-tocopherols in lung cancer risk. Int J Cancer. 2008 Sep 1;123(5):1173-80.
Martinez-Outschoorn UE, Trimmer C, Lin Z, Whitaker-Menezes D, Chiavarina B, Zhou J, Wang C, Pavlides S, Martinez-Cantarin MP, Capozza F, Witkiewicz AK, Flomenberg N, Howell A, Pestell RG, Caro J, Lisanti MP, Sotgia F. Autophagy in cancer associated fibroblasts promotes tumor cell survival: Role of hypoxia, HIF1 induction and NFOE∫B activation in the tumor stromal microenvironment. Cell Cycle. 2010 Sep 1;9(17):3515-33.
Matthews RT, Yang L, Browne S, Baik M, Beal MF. Coenzyme Q10 administration increases brain mitochondrial concentrations and exerts neuroprotective effects. Proc Natl Acad Sci U S A. 1998 Jul 21;95(15):8892-7.
McCarty MF, Barroso-Aranda J, Contreras F. The “rejuvenatory” impact of lipoic acid on mitochondrial function in aging rats may reflect induction and activation of PPARgamma coactivator-1alpha. Med Hypotheses. 2009 Jan;72(1):29-33.
McKinney N. Mitochondria Rescue (Possibly) Heals Cancer? Naturopathic Doctor News & Review (NDNR) 2008; 4 (5): 10-11.
McMackin CJ, Widlansky ME, Hamburg NM, Huang AL, Weller S, Holbrook M, Gokce N, Hagen TM, Keaney JF Jr, Vita JA. Effect of combined treatment with alpha- Lipoic acid and acetyl-L-carnitine on vascular function and blood pressure in patients with coronary artery disease. J Clin Hypertens (Greenwich). 2007 Apr;9(4):249-55.
Michelakis ED, Sutendra G, Dromparis P, Webster L, Haromy A, Niven E, Maguire C, Gammer TL, Mackey JR, Fulton D, Abdulkarim B, McMurtry MS, Petruk KC. Metabolic modulation of glioblastoma with dichloroacetate. Sci Transl Med. 2010 May 12;2(31):31ra34.
Moungjaroen J, Nimmannit U, Callery PS, Wang L, Azad N, Lipipun V, Chanvorachote P, Rojanasakul Y. Reactive oxygen species mediate caspase activation and apoptosis induced by lipoic acid in human lung epithelial cancer cells through Bcl-2 downregulation. J Pharmacol Exp Ther. 2006 Dec;319(3):1062-9.
Ordys BB, Launay S, Deighton RF, McCulloch J, Whittle IR. The role of mitochondria in glioma pathophysiology. Mol Neurobiol. 2010 Aug;42(1):64-75.
Parkhomenko IuM, Chernysh IIu, Churilova TIa, Khalmuradov AG. [Effect of thiamine phosphates on the activity of regulatory enzymes of the pyruvate dehydrogenase complex]. Ukr Biokhim Zh. 1987 Sep-Oct;59(5):49-54.
Pelicano H, Xu RH, Du M, Feng L, Sasaki R, Carew JS, Hu Y, Ramdas L, Hu L, Keating MJ, Zhang W, Plunkett W, Huang P. Mitochondrial respiration defects in cancer cells cause activation of Akt survival pathway through a redox-mediated mechanism. J Cell Biol. 2006 Dec 18;175(6):913-23.
Perumal SS, Shanthi P, Sachdanandam P. Augmented efficacy of tamoxifen in rat breast tumorigenesis when gavaged along with riboflavin, niacin, and CoQ10: effects on lipid peroxidation and antioxidants in mitochondria. Chem Biol Interact. 2005a Feb 28;152(1):49-58.
Perumal SS, Shanthi P, Sachdanandam P. Energy-modulating vitamins–a new combinatorial therapy prevents cancer cachexia in rat mammary carcinoma. Br J Nutr. 2005b Jun;93(6):901-9.
Rahman KM, Aranha O, Glazyrin A, Chinni SR, Sarkar FH. Translocation of Bax to mitochondria induces apoptotic cell death in indole-3-carbinol (I3C) treated breast cancer cells. Oncogene. 2000 Nov 23;19(50):5764-71.
Rahman KM, Aranha O, Sarkar FH. Indole-3-carbinol (I3C) induces apoptosis in tumorigenic but not in nontumorigenic breast epithelial cells. Nutr Cancer. 2003;45(1):101-12.
Ralph SJ, Rodriguez-Enriquez S, Neuzil J, Saavedra E, Moreno-Sanchez R. The causes of cancer revisited: “mitochondrial malignancy” and ROS-induced oncogenic transformation – why mitochondria are targets for cancer therapy. Mol Aspects Med. 2010 Apr;31(2):145-70.
Ramos-Montoya A, Lee WN, Bassilian S, Lim S, Trebukhina RV, Kazhyna MV, Ciudad CJ, Noé V, Centelles JJ, Cascante M. Pentose phosphate cycle oxidative and Nonoxidative balance: A new vulnerable target for overcoming drug resistance in cancer. Int J Cancer. 2006 Dec 15;119(12):2733-41.
Ray S, Biswas S, Ray M. Similar nature of inhibition of mitochondrial respiration of heart tissue and malignant cells by methylglyoxal. A vital clue to understand the biochemical basis of malignancy. Mol Cell Biochem. 1997 Jun;171(1-2):95-103.
Rodriguez MC, MacDonald JR, Mahoney DJ, Parise G, Beal MF, Tarnopolsky MA. Beneficial effects of creatine, CoQ10, and lipoic acid in mitochondrial disorders. Muscle Nerve. 2007 Feb;35(2):235-42.
Schaefer AM. Trial of dichloroacetate in MELAS: toxicity overshadows the assessment of potential benefit. Neurology. 2006 Feb 14;66(3):302-3.
Seth P, Grant A, Tang J, Vinogradov E, Wang X, Lenkinski R, Sukhatme VP. On-target inhibition of tumor fermentative glycolysis as visualized by hyperpolarized pyruvate. Neoplasia. 2011 Jan;13(1):60-71.
Simbula G, Columbano A, Ledda-Columbano GM, Sanna L, Deidda M, Diana A, Pibiri M. Increased ROS generation and p53 activation in alpha-lipoic acid-induced apoptosis of hepatoma cells. Apoptosis. 2007 Jan;12(1):113-23.
Spindler SR. Caloric restriction: from soup to nuts. Ageing Res Rev. 2010 Jul;9(3):324-53.
Stacpoole PW, Henderson GN, Yan Z, James MO. Clinical pharmacology and toxicology of dichloroacetate. Environ Health Perspect. 1998 Aug;106 Suppl 4:989-94.
Suolinna EM, Buchsbaum RN, Racker E. The effect of flavonoids on aerobic glycolysis and growth of tumor cells. Cancer Res. 1975 Jul;35(7):1865-72.
Suwa M, Egashira T, Nakano H, Sasaki H, Kumagai S. Metformin increases the PGC- 1alpha protein and oxidative enzyme activities possibly via AMPK phosphorylation in skeletal muscle in vivo. J Appl Physiol. 2006 Dec;101(6):1685-92.
Talmadge JE, Donkor M, Scholar E. Inflammatory cell infiltration of tumors: Jekyll or Hyde. Cancer Metastasis Rev. 2007 Dec;26(3-4):373-400.
Walenta S, Mueller-Klieser WF. Lactate: mirror and motor of tumor malignancy. Semin Radiat Oncol. 2004 Jul;14(3):267-74.
Walenta S, Wetterling M, Lehrke M, Schwickert G, Sundfor K, Rofstad EK, Mueller- Klieser W. High lactate levels predict likelihood of metastases, tumor recurrence, and restricted patient survival in human cervical cancers. Cancer Res. 2000 Feb 15;60(4):916-21.
Wallace DC. Mitochondria and cancer: Warburg addressed. Cold Spring Harb Symp Quant Biol. 2005;70:363-74.
Warburg O. On the origin of cancer cells. Science. 1956 Feb 24;123(3191):309-14.
Wenzel U, Daniel H. Early and late apoptosis events in human transformed and nontransformed colonocytes are independent on intracellular acidification. Cell Physiol Biochem. 2004;14(1-2):65-76.
Wenzel U, Nickel A, Daniel H. alpha-Lipoic acid induces apoptosis in human colon cancer cells by increasing mitochondrial respiration with a concomitant O2-*- generation. Apoptosis. 2005a Mar;10(2):359-68.
Wenzel U, Nickel A, Daniel H. Increased carnitine-dependent fatty acid uptake into mitochondria of human colon cancer cells induces apoptosis. J Nutr. 2005b Jun;135(6):1510-4.
Wigfield SM, Winter SC, Giatromanolaki A, Taylor J, Koukourakis ML, Harris AL. PDK-1 regulates lactate production in hypoxia and is associated with poor prognosis in head and neck squamous cancer. Br J Cancer. 2008 Jun 17;98(12):1975-84.
Yang JH, Hsia TC, Kuo HM, Chao PD, Chou CC, Wei YH, Chung JG. Inhibition of lung cancer cell growth by quercetin glucuronides via G2/M arrest and induction of apoptosis. Drug Metab Dispos. 2006 Feb;34(2):296-304.
Zhang XM, Chen J, Xia YG, Xu Q. Apoptosis of murine melanoma B16-BL6 cells induced by quercetin targeting mitochondria, inhibiting expression of PKC-alpha and translocating PKC-delta. Cancer Chemother Pharmacol. 2005 Mar;55(3):251-62.
Ziegler D, Hanefeld M, Ruhnau KJ, Hasche H, Lobisch M, Schutte K, Kerum G, Malessa R. Treatment of symptomatic diabetic polyneuropathy with the antioxidant alpha-lipoic acid: a 7-month multicenter randomized controlled trial (ALADIN III Study). ALADIN III Study Group. Alpha-Lipoic Acid in Diabetic Neuropathy. Diabetes Care. 1999 Aug;22(8):1296-301.