The Coalition against Morbid Obesity has launched a social media campaign to combat morbid obesity in an attempt to break the isolation of people who are morbidly obese, raise their awareness of the dangerous effects of their disease on their health and quality of life, and inform them about the existence of an effective treatment. In two video testimonies that can be found on YouTube and the Coalition’s Website (www.lepoidsquitue. com) and Facebook page, women speak about their experiences with this condition. In the first video, a woman talks about becoming aware of the fact that she could not fight this disease alone and about the help she sought out. In the second video, another woman talks about her decision to undergo bariatric surgery in 2004, which was a choice she made to improve her quality of life. In the campaign’s second stage, people with morbid obesity will be invited to create their own video message. The Coalition hopes that this campaign will go viral to encourage people with morbid obesity to learn more about the disease, about the dangers it presents to their health and quality of life, and about bariatric surgery.
Canada’ s pioneer of integrative cancer care passes on
Dr. Roger Rogers, BA, BSW, MD, OBC, died peacefully on November 22, 2011 at the age of 83 years. As a co-founder of InspireHealth, Canada’s first integrative cancer care centre, Dr. Rogers is recognized as a pioneer and leader in integrative cancer care. “It is with sadness and the celebration of a life well lived that I share, with the extended InspireHealth family, that Dr. Rogers has peacefully passed away. Loved by his patients as much as he loved them, Roger was an inspiration to several generations of physicians,” commented Dr. Hal Gunn, co-founder and CEO of InspireHealth. Dr. Rogers was the grateful recipient of many awards and acknowledgements, culminating in the presentation of the Order of British Columbia in 2001. The Hecht Foundation created the Dr. Roger Rogers Prize in 2007, which awards a $250 000 prize biennially to a leader in complementary and alternative medicine. Dr Rogers leaves the legacy of an extraordinary career that will continue to shape and influence the future of cancer care not only in Canada but also worldwide.
Occupational therapists launch new guidelines to help stop elder abuse
TheCanadianAssociationofOccupational Therapists (CAOT) has launched “Strategies for occupational therapists to address elder abuse/mistreatment, “ a tool developed for occupational therapists to address and manage elder abuse. This resource provides a set of guidelines that outline what to look for (indicators), first steps to follow when suspecting abuse (e.g. What do I ask? Look for?), and strategies for discussing the situation with the older adult. “Occupational therapists are in a unique position to detect and manage elder abuse due to the strong working relationship they develop with an older adult through having an intimate knowledge of their daily life and routines,” said Rosemary Lester, Chair of the Elder Abuse Committee of Newfoundland and Labrador and External Member of the CAOT Board of Directors. “This tool will support this relationship and be an important resource in the ongoing effort to combat elder abuse.” Occupational therapists can download this tool off the CAOT website at www.caot.ca/elderabuse.
Gamma-Dynacare has been named one of Greater Toronto’s top employers
For the fourth consecutive year, Mediacorp Canada, Inc. has named Gamma-Dynacare Medical Laboratories one of Greater Toronto’s top employers. “We are once again delighted to receive this recognition,” said Naseem Somani, President and CEO, Gamma-Dynacare Medical Laboratories. “It is our belief that we have the very best employees in the industry – employees who, through their commitment and dedication to the Canadians we serve – live our mission of providing information, impacting lives and caring for people, each and every day.” The winners were selected from among 2700 workplaces with head offices or principal places of business in the Toronto area that are industry leaders at creating exceptional workplaces and attracting and retaining employees. Employers were evaluated based on a number of factors, including work atmosphere and communication, benefits, training and skills development, and community involvement.
WorkSafeBC has launched a new provider portal to accelerate referrals for injured workers
WorkSafeBC makes up to 35 000 referrals every year that affect injured workers, provider partners, and case management teams. TELUS Health Solutions and WorkSafeBC have recently launched a new provider portal to help injured workers receive faster referrals to the healthcare resources they need to facilitate their recovery. Open 24/7, the new provider portal supports electronic referrals, clinical reporting, online invoicing, and access to claim and payment information. “With the new portal open for business, we aim to reduce the process period from the time a rehab program is requested, to the provider’s receipt of the referral from one or two days to a matter of hours. This means injured workers in need of treatment will receive the services they require sooner,” said Andrew Montgomerie, director, Health Care Services, WorkSafeBC. With 85 clinics already using the portal, WorkSafeBC plans to expand to as many as 200 clinics this year including occupational rehabilitation and return- to-work providers.
The iDAPT Centre for Rehabilitation Research opens in downtown Toronto
The Toronto Rehabilitation Institute, part of the University Health Network, has opened its $36-million-dollar research centre, called iDAPT (Intelligent Design for Adaptation, Participation and Technology). iDAPT is located in downtown Toronto and scientists and research students from a broad range of disciplines work collaboratively to develop solutions that will help restore independence and quality of life for people recovering from injury or illness. The Institute Director, Dr. Geoff Fernie notes, “iDAPT research will produce new knowledge, more practical technologies and innovative treatments that will reduce accidents and illness and help people overcome disability. We can help people live healthier, more active and more independent lives.” “Much of the work Toronto Rehab is doing here will help people remain in their homes longer. The HomeLab, for instance, is a living environment where new assistive devices and adaptive technologies are being developed to help people stay safely in their home for longer,” said Nancy Lefebre, Senior Vice President, Knowledge and Practice, Saint Elizabeth. “Supporting people to stay in their homes will reduce the burden on the healthcare system.” For more information, visit www. torontorehab.com.
Emergency rooms often skip tests for young kids with fever
A new study finds that babies and toddlers who arrive at the emergency room (ER) with an unexplained fever often receive no kind of diagnostic test to reveal the source of the high temperature. No diagnostic tests were ordered in 59% of the cases where a child aged three months to three years had a fever of unknown cause while urine tests and blood tests were conducted in 17% and 20% of cases, respectively. Prescribing antibiotics without testing to confirm the presence of a bacterial infection can potentially lead to antibiotic overuse. ER doctors prescribed antibiotics ~25% of the time, including for 20% of the fevers where no diagnostic test was performed. These results are consistent with past studies that have pointed to antibiotic overuse. Dr. Alan Simon, lead researcher, said that there is no universal advice to give parents on whether their feverish child should get a diagnostic test. “There are particulars of each case that would sway any one practitioner’s decision,” and the broader question is whether more testing, or less testing, would actually improve children’s care. “We don’t know yet if changes in current practice would lead to better outcomes.” Pediatrics. 2011 Nov 21. PMID: 22106081
Cytonet’s Liver Cell Therapy Trial for children with urea cycle disorders expands to Canada
Health Canada recently granted Cytonet approval to extend the SELICA (Safety and Efficacy of Liver Cell Application)-III clinical trial into Canada. This trial, which has been open in the U.S. since 2010, is designed to evaluate the safety and efficacy of liver cell therapy in infants to children up to five years of age with urea cycle disorders (UCD). Two centers in Canada will join the 12 medical centers in the U.S. taking part in the trial: the Alberta Children’s Hospital in Calgary and the Hospital for Sick Kids in Toronto. “Adding centers in Canada will help us achieve our goal of enrolling 20 children in the trial and more widely offers a much-needed new option for a very serious condition where the only current cure is liver transplantation,” says Dr. Wolfgang Rudinger, CEO and CSO of Cytonet Germany. For more information, please visit http://www.cytonetllc.com.
Effectiveness and safety of the homeopathic preparation Pasconal® Nerventropfen in patients with nervous disorders
Effectiveness and safety of the homeopathic preparation Pasconal® Nerventropfen in patients with nervous disorders
ABSTRACT
Objective:
We investigated the safety and effectiveness of a homoeopathic combination preparation in patients suffering from nervousness and sleep disorders.
Methods:
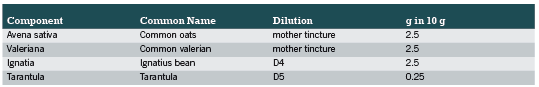
In this observational study, 325 patients suffering from nervous disturbances and sleep disorders were treated for about four weeks with a homeopathic combination preparation containing Avena sativa, Valeriana, Ignatia and Tarantula. The effectiveness and safety was assessed by a questionnaire filled out by the therapists.
Results:
Patients without concomitant medication and therapy duration over four weeks responded better to the therapy with the homeopathic preparation. A clear reduction in the symptom severity could be observed for all 12 analyzed symptoms. In patients who did not receive concomitant medication due to the inclusion diagnosis, a comparatively more positive and significant (p < 0.05) effect on the symptoms nervousness/restlessness and irritability/eccentricity was observed. The effect of PASCONAL® NERVENTROPFEN on five individual symptoms (nervousness/restlessness, irritability/eccentricity, sleep disorders, fitful sleep, and hyperactivity) was more pronounced in patients who were treated for four weeks or more than in patients with a shorter treatment period. All p-values for comparison between the two subgroups were < 0.05. The therapy was well-tolerated.
Conclusion:
This observational study indicates that the homoeopathic combination preparation is an effective and well-tolerated alternative to pharmaceuticals for the treatment of nervous disturbances and sleep disorders.
Keywords:
nervousness, sleep disorders, homeopathic combination preparation, Avena sativa, Valeriana, Ignatia, Tarantula
INTRODUCTION
Stress dominates the lives of many people, frequently leading to undesirable health consequences, including nervousness, tension and sleep disorders. Stress often arises as a response to current circumstances of life, such as job-related, school, or private stress; rising workload or academic demands; overstimulation; inability to relax; burdening living conditions; and personal disappointments or losses. Psychiatric disorders such as sleep disturbances, psychosomatic complaints, states of anxiety and depression occur far more often than is widely believed (Kraft 2007).
The spectrum of interventions for nervousness, restlessness, and sleep disturbances is broad, and treatments range from benzodiazepines and anxiolytics to relaxations techniques (Hankey 2006) and homoeopathic preparations (Kumar 2005). Traditional allopathic medicine is the most common mode of treatment for stress-related conditions but because side effects, interactions, and the potential for pharmaceutical dependence do not allow long-term treatment (Rosenberg 2006, Tan 2010), alternativetherapies are increasingly employed (Barnes 2004, Schneider 2004). Positive results have been reported for the treatment of sleep disorders (Coholic 2005, Waldschutz 2008), mild nervous disorders (Meerschaut 2007), and mental trauma (Ventegodt 2006) with complementary therapies. Given the superior tolerability of complementary and alternative therapy over conventional drug therapy, phytotherapeutic and homoeopathic remedies represent a potentially valuable treatment avenue in the management of sleep disorders, mild panic attacks, psychovegetative complaints, and depression. These alternative treatment methods can be used concomitantly with specific therapies addressing the underlying disease along with additional measures such as sleep hygiene, autogenic training, sports, identification of psychosocial burdens and appropriate psychotherapy (Kraft 2010).
The current observational study was undertaken to evaluate the safety and effectiveness of PASCONAL® NERVENTROPFEN on these nervous conditions in both adults and children.
METHODS
Study Design
This prospective, non-interventional, non-randomised, cohort study was conducted between 1st May and 31st August 2010 by 71 physicians. Male and female patients aged 1 to 89 years suffering from nervous disturbances and sleep disorders were treated with the homoeopathic combination product PASCONAL® NERVENTROPFEN (PASCOE pharmazeutische Praeparate GmbH, Giessen, Germany) in everyday practise. Treatment and dosage were decided by the therapist. No specific inclusion and exclusion criteria were defined due to the character of a non-interventional study. Data assessment included a physician-completed questionnaire on three occasions: at baseline (beginning of the observation period; visit one), approximately two weeks later (visit two) and approximately four weeks later (end of the observation period; visit three).
The study was registered with the Protocol Registration System ClinicalTrials.gov (NCT01125605). All participants and their parents gave written, informed consent.
Treatment
The patients were treated with the homoeopathic combination product PASCONAL® NERVENTROPFEN (ingredients listed in Table 1-Components of PASCONAL® NERVENTROPFEN and their dilutions). This product is indicated for the use in sleep disorders due to nervous restlessness according to its active ingredients (as listed in Felter´s Eclectic Materia Medica). Dose and duration of treatment were left to the respective physician’s discretion.
Outcome measures and Analyses
The data presented in this study are based on the data collected from the physician-completed questionnaires. The data were documented on at least two visits or on three visits at best. At the first visit, demographic data (sex, age, body height, body weight) were collected, as well as data on the duration of disease, previous therapies of the inclusion diagnosis, effectiveness and tolerability of previous therapies, relevant concomitant diseases/medication and the severity of symptoms. At visits two and three, dosage of the study medication and/or other medications, severity of 12 symptoms (nervousness/restlessness, irritability/eccentricity, sleep disorders, fitful sleep, hyperactivity, nocturnal activity, lack of concentration/forgetfulness, tiredness, discontent, listlessness, gastrointestinal problems, and headache/pressure), and effectiveness and tolerability of the study medication were recorded. If the study was completed on visit two, the effectiveness of PASCONAL® NERVENTROPFEN and adverse drug reactions were documented. Otherwise, these data were collected on visit three.
The severity of symptoms was graded on a four-point scale from 0 (no complaints) to 3 (strong complaints). The change from baseline to the end of the observation was calculated for the individual symptom scores and for the sum score of all 12 individual symptom scores (0 – 36).
Additionally, the improvement of the symptoms versus baseline was assessed at the end of the observation on a scale that ranged from improved, to no change, to deteriorated.
Statistics
Due to the character of an observational study, no hypotheses were specified and all conclusions were drawn from descriptive data analysis. The following values were computed, depending on the type of parameter: frequency data (absolute and relative frequencies), proportionally scaled measured values (median, 25% and 75% quartile, arithmetic mean, standard deviation, variance, minimum, maximum, number of valid data and number of missing data), and pre- / post-comparisons for data concerning symptoms.
Additionally, the main efficacy variables were analysed by explorative statistics. Parameters for analysis included the sum score of 12 clinical symptoms as well as the five individual symptoms nervousness/restlessness, irritability/eccentricity, sleep disorders, fitful sleep, and hyperactivity. Explorative statistical analysis was based on the change of the sum score between baseline (visit one) and the last documented visit (visit two or visit three), according to the LOCF (Last observation carried forward) principle. The change was calculated as “value pre minus value post”, which means that positive values indicate a decrease of the score (corresponding to an improvement). The sum score was also analysed with respect to age, gender, concomitant medication and treatment duration (< 4 weeks versus ≥ 4 weeks).
The one-sample t-test (two-sided) was used to test the null hypothesis that the changes from baseline were equal to zero. This analysis was performed for the total population and for each of the considered strata (age, gender, concomitant medication, treatment duration).
Analysis of covariance (ANCOVA) was applied to investigate the differences in the changes of the sum score between the various subgroups. The GLM (Generalized Linear Model) procedure of the SAS® system (with type III sums of squares) was used after including the baseline value of the sum score as a covariate in the underlying statistical model.
With respect to age, two types of analysis were carried out: 1) all four subgroups (1 to 6 years, 7 to 11 years, 12 to 17 years and ≥ 18 years); and 2) < 18 years and ≥ 18 years.
In order to show the results when baseline adjustment is omitted, the two-sample t-test (two-sided) was performed in addition to ANCOVA (only for pair-wise comparison).
RESULTS
A total of 325 patients were enrolled in the study and their treatment was documented during the observational study by 71 physicians. Baseline characteristics are summarised in Table 2. The inclusion diagnosis was made an average of two years ago and female patients suffered longer from nervousness than male patients (2.3 years versus 1.3 years). Two hundred forty five patients reported receiving previous therapies due to the inclusion diagnosis (11.1% only drug therapy; 24% only physical therapy/other therapy; 40.3% drug therapy and physical therapy/other therapy). The effectiveness of these previous therapies was mostly rated as “moderate” (58.4%) or “with no effect” (17.6%), while only 22.4% judged the effectiveness as “good” or “very good”. The tolerability of the previous therapies was rated as “very well” in 73.1% and only 4.5% stated poor tolerability. 70 patients continued their concomitant medication (92 medications in total) due to the inclusion diagnosis. It is shown that most of these continued drugs (43,5%) were all kinds of psychopharmaceuticals followed by all kinds of hypnotics/sedatives (40.2%)

The patients were treated with a mean daily dosage of three times a day 10 gtt PASCONAL® NERVENTROPFEN. The majority of patients were treated within the dosage recommendation as described in the SPC of PASCONAL® NERVENTROPFEN and the remaining patients were treated above the dosage recommendation.
In total, 12 different symptoms were observed during the time of the study, namely nervousness/restlessness, irritability/eccentricity, sleep disorders, hyperactivity, fitful sleep, nocturnal anxiety, lack of concentration/forgetfulness, tiredness, listlessness, discontent, gastrointestinal problems, and headache/head pressure. The change in the severity of each symptom between the start and end of the observation period is shown in Table 3. For the symptoms of nervousness/restlessness, irritability/eccentricity, sleep disorders, and fitful sleep, the majority of patients stated moderate or strong complaints (75.4%, 63.7%, 70.5% and 69.2%, respectively) at the start of the observation. These symptoms improved during the course of the study, with 87.7%, 88.9%, 84% and 87.4% of the patients, respectively, stating that they had no complaints anymore or only suffered from mild complaints.
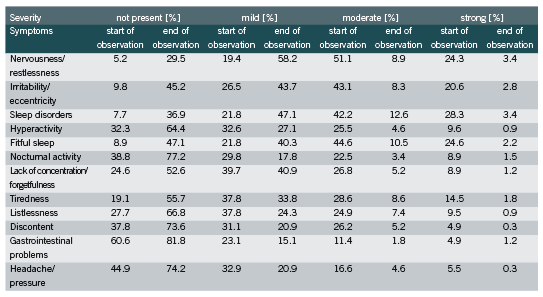
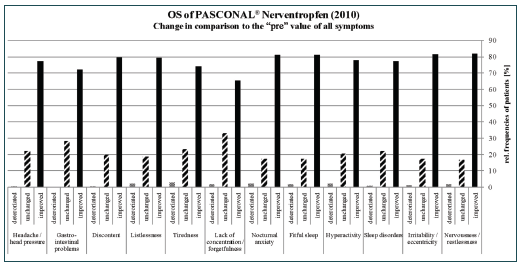
The other symptoms were less pronounced; for example, only 16.3% and 22.1% of the patients suffered from moderate and strong gastrointestinal problems and headache/pressure, respectively. Nevertheless, an improvement in at least 65% of the patients was observed for each symptom at the end of the observational study (Figure 1). In all symptoms less than 3% deterioration was seen.
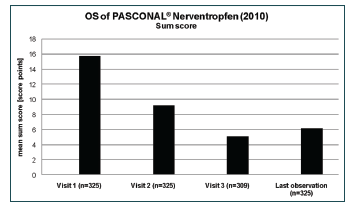
All symptoms were summarised to one “sum score”. The value decreased from and initial score of 15.8 to a score of 6.2 at the end of the study (Figure 2). The explorative analysis of the sum score was carried out with respect to age, gender, concomitant medication, and treatment duration. The difference between males and females in the mean decreases of the sum score (9.3 vs. 9.7) was not significant (p = 0.6115). The global test for differences between the four age groups did not show a significant result (p=0.0960). For a more robust analysis, all patients <18 years were combined into one age group. The mean baseline values of the sum score for the two resulting groups were 13.5 (<18 years) and 16.4 (≥18 years), respectively. Although the difference in the mean decreases was only 0.3 points, ANCOVA with baseline adjustment produced a significant result (p= 0.0238). The result of the analyses showed that the baseline value of the scan had a significant impact (p<0.0001) on the observed change.
Although the mean baseline values were comparable between patients who received concomitant medication due to the inclusion diagnosis and patients who did not (15.9 vs. 15.7), the decrease of the sum score was considerably more pronounced in the latter group (8.2 vs. 9.9). The difference between both subgroups of patients was significant (p = 0.0033).
Patients who received PASCONAL® NERVENTROPFEN for less than four weeks showed a considerably lower decrease of the sum score than patients who had been treated for four weeks or more (6.9 vs. 10.2). The difference between both subgroups of patients was significant (p = 0.0002).
The five individual symptoms, nervousness/restlessness, irritability/eccentricity, sleep disorders, fitful sleep, and hyperactivity, were analysed with respect to the direction of change between baseline and the last documented visit by means of the categories “improved”, “unchanged” and “worsened”. Differences between subgroups were analysed with Fisher’s exact test (two-sided) for stratification by age (<18 years vs. ≥ 18 years), concomitant medication (due to inclusion diagnosis), and duration of treatment with PASCONAL® NERVENTROPFEN. The age of the patients had no significant effect on the five selected symptoms. In patients who did not receive concomitant medication due to the inclusion diagnosis, a comparatively more positive and significant (p < 0.05) effect on the symptoms nervousness/restlessness and irritability/eccentricity was observed. The corresponding p-values for the remaining three symptoms were > 0.05. The effect of PASCONAL® NERVENTROPFEN on all five individual symptoms was more pronounced in patients who were treated for four weeks or more than in patients with a shorter treatment period. All p-values for comparison between the two subgroups were < 0.05.
Overall, PASCONAL® NERVENTROPFEN was well tolerated by 96.0% of the patients in the first observation interval and by 92.9% in the second observation interval. Fifteen patients reported moderate or poor tolerability of PASCONAL® NERVENTROPFEN. Seven adverse events were reported with possible relationship to study treatment (2x nausea, 2x headache, 2x tiredness and 1x tachycardia/acid reflux ), four with probable relationship (2x gastrointestinal disorders, 1 x severe tiredness and 1 x heartburn), two events with relationship unlikely (1 x aggressivity, 1 x stomach ache,) and only one with a certain relationship to the treatment (tongue and mucosa under tongue affected by alcohol concentration of the drops). In one case the relationship was not assessable. None of the adverse events required additional medical treatment.
DISCUSSION
The results of this observational study reveal that the homoeopathic combination preparation, PASCONAL® NERVENTROPFEN, is effective in the symptomatic treatment of sleep disorders due to nervous restlessness.
The baseline data revealed that the majority of patients suffered from nervousness/restlessness, irritability/eccentricity, sleep disorders, and fitful sleep. The majority of patients had already received therapies due to the inclusion diagnosis and half of those therapies were pharmaceuticals. Astonishingly, over 75% of the patients who used previous therapies reported either no effect or moderate effectiveness of these therapies.
After treatment with the homoeopathic combination preparation, a clear reduction in the symptom severity was observed for all analysed symptoms. Most patients experienced more than a 50% reduction in moderate and strong symptoms. Upon analysis of the sum score, it can be concluded that from start until the end of the observation, a reduction by 9.6 points could be observed, which corresponds to a decrease by 60%.
The explorative analysis showed a connection between the sum score and age groups, concomitant medication and therapy duration, respectively. Patients over 18 years of age had a significant reduction of the mean sum score compared with the group of patients less than 18 years. However, it is important to point out that both age groups differ in the number of patients (72 under 18 years versus 253 patients over 18 years).
Patients who did not take any concomitant medication due to the inclusion diagnosis showed a higher decrease in the mean value of the sum score at the end of the observation. Compared with the group of patients who received concomitant medication, this result was significant.
Therapy duration influenced the sum score value: patients who had been treated for four weeks and more showed a considerably higher decrease of the sum score than patients who received the homeopathic combination preparation less for than four weeks. The difference between both subgroups of patients was significant.
Age had no influence on the direction of change (improved, unchanged or worsened) between baseline (visit 1) and the last documented visit of the five symptoms (nervousness/restlessness, irritability/eccentricity, sleep disorders, fitful sleep and hyperactivity). However, patients who did not receive concomitant medication showed a significant effect on the direction of change in nervousness/restlessness and irritability/eccentricity. For the remaining three symptoms, no significant effect could be observed. Interestingly, an effect on all five symptoms could be shown in patients who were treated four weeks or more.
The therapy was well tolerated, which was to be expected based on the experience gained in pharmacovigilance surveillance of the product.
LIMITATIONS
Observational studies have several limitations (Black 1996), including the absence of a placebo group. This can create bias and can also mask cause and effect relationships or suggest correlations where there are none. Therefore, no definite conclusions can be extrapolated from these studies.
AUTHORS’ CONTRIBUTIONS
Financial support for the analysis was provided by PASCOE pharmazeutische Präparate GmbH, Germany. The sponsor had influence on the conduct of the analysis in so far as the management and evaluation of the data was conducted in the department of Clinical Research of PASCOE pharmazeutische Präparate GmbH. The explorative statistics were done by D. Schremmer, GKM Gesellschaft für Therapieforschung mbH, Germany.
LITERATURE
Barnes PM, Powell-Griner E, McFann K, Nahin RL. Complementary and alternative medicine use among adults: United States, 2002. Adv Data 2004; (343): 1-19.
Berger H, Fritz U. Medication in acute psychiatric treatment. Psychiatr Prax. 2004; 31 (2): 68-73.
Black N. Why we need observational studies to evaluate the effectiveness of health care. BMJ. 1996; 312(7040): 1215-8.
Coholic D. The Helpfulness of Spiritually Influenced Group Work in Developing Self-Awareness and Self-Esteem: A Preliminary Investigation.” The Scientific World Journal. 2005; 5: 789–802.
Hankey A. CAM and Post-Traumatic Stress Disorder. eCAM. 2006
Kraft K. Schlaf- und psychische Störungen im Kindesalter. Z Phytotherapie. 2007; 28: 235-7.
Kraft K. Depressionen und depressive Verstimmungszustände. MMW Fortschr Med. 2010;152(8): 20-21.
Kumar S, Anupam S. Anti-anxiety Activity Studies on Homoeopathic Formulations of Turnera phrodisiaca Ward. eCAM. 2005; 2 2(1): 117-119.
Meerschaut Lvd, Andrea S. The Homeopathic Preparation Nervoheel N can Offer an Alternative to Lorazepam Therapy for Mild Nervous Disorders. eCAM. 2007
Rosenberg RP. Sleep maintenance insomnia: strengths and weaknesses of current pharmacologic therapies. Ann. Clin. Psychiatry. 2006; 18: 49-56.
Schneider B, Hanisch J, Weiser M. Complementary medicine prescription patterns in Germany. Ann Pharmacother. 2004; 38: 502–507.
Tan KR, Brown M, Labouebe G, Yvon C, Creton C, Fritschy JM, Rudolph U, Luscher C. Neural bases for addictive properties of benzodiazepines. Nature. 2010; 463: 769-774.
Ventegodt S, Clausen B, Merrick J. Clinical Holistic Medicine: The Case Story of Anna. I. Long-Term Effect of Childhood Sexual Abuse and Incest with a Treatment Approach. The Scientific World Journal. 2006; 6: 1965-1976.
Waldschutz R, Klein P. The homeopathic preparation Neurexan vs. valerian for the treatment of insomnia: an observational study. Scientific World Journal. 2008; 8: 411-20.
Statin prescription for patients with diabetes
Statin prescription for patients with diabetes
Yeah or Nay?
Introduction
In 2011, the American Diabetes Association (ADA) published their view of the standard of medical care for diabetes (American Diabetes Association 2011). Central to prevention and management of cardiovascular complications is the focus on LDL cholesterol and the recommendation of statin use by a significant fraction of all individuals with diabetes. Is this position consistent with the evidence available?
LDL and Coronary Atherosclerosis. The Absence of an Association Among Individuals with Diabetes
A number of studies have been published recently regarding coronary artery calcium, a marker for atherosclerosis associated coronary plaque, in patients with type II diabetes free of symptomatic cardiovascular disease. All find LDL is not a factor in the prevalence of coronary artery calcium (CAC) (Elkeles 2004, Godsland 2006, Kobayashi 2008, Martin 2009, Mazzone 2007, Wolfe 2002). This conclusion was mostly based on multivariate risk factor analysis that failed to point to LDL as significant. Rather, age, systolic blood pressure or hypertension, gender and race were the important factors for prevalence. These studies, which all involved both genders and a wide range of age and ethnic background, raise a serious issue concerning the total lack of evidence for LDL as a target for therapy associated with coronary atherosclerosis in asymptomatic patients with diabetes. This is important because coronary atherosclerosis is almost always a prerequisite precursor for acute coronary events. These results are consistent with those found in a large number of studies of non-diabetic individuals free of symptomatic heart disease (Ware 2009).
Coronary Artery Calcification. Progression Studies Among Individuals with Diabetes
When coronary calcification is present, it almost always progresses and this is associated with increasing risk of acute cardiac events. LDL also does not appear associated with the risk of progression of CAC among individuals with type II diabetes. Consider the following studies involving subjects free of coronary heart disease (CHD).
• A substudy of the Veterans Affairs Diabetes Trial examined the progression of CAC in 189 individuals with type II diabetes (Saremi 2010). CAC progression was found in > 75% of subjects but was not influenced by standard risk factors and in particular, blood lipids. The albumin to creatinine ratio predicted progression independent of adjustment for age or other traditional risk factors.
• A recent study examined CAC progression in a group of 398 individuals with type II diabetes (Anand 2007). In the final multivariate analysis, odds ratios (OR) for independent factors for progression of CAC were; elevated baseline CAC (OR = 6.38), HbA1c > 7 (OR =1.95), and statin use (OR = 2.27), but not hyperlipidemia or smoking status. It is noteworthy that the researchers found statin therapy failed to inhibit progression of CAC but rather appeared to accelerate it. The increase in risk of progression was 212% for statin-treated patients (38-50%) with LDL < 2.6 mmol/L compared to those untreated. An earlier study from the same group found that 53% of a cohort of individuals with diabetes had subclinical CAC. One-third of these had progression during 2.5 years follow-up (Anand 2006).
• Progression of atherosclerosis over four years measured by CAC was also addressed as part of the PREDICT study (Elkeles 2008). The rate of change was strongly related to the baseline coronary artery calcium score (CACS) and also, independent of CACS, correlated positively with waist to hip ratio, male gender, the use of antihypertensive drugs or statins and the albumin to creatinine ratio. There was no relationship with traditional lipid risk factors.
• Compared to the above, somewhat different results have been reported (Budoff 2005). The study excluded individuals with cardiac symptoms or known CAD including revascularization, stroke or peripheral vascular disease. Two CT calcium scans were done with a mean of 27 months between. Hypercholesterolemia, defined as using cholesterol-lowering drugs or having total cholesterol (TC) ≥ 240 mg/dL, was not found to be a significant risk factor for progression. For the statin non-users, progression was at a median annual rate of 20% (4% to 44%) whereas for statin treated patients it was 10% (4% to 25%) No statistical analysis was included, but the reported ranges are very large.
• It is well known that depression is associated with diabetes and depressive symptoms are a risk factor for CHD (Campayo 2011). A recent report documented this in midlife women in the SWAN Heart Study, where depressive symptoms were independently associated with progression of CAC (Janssen 2011). In a multivariate analysis, the only other predictors of significance for progression were systolic blood pressure and a low level of education but not cholesterol levels.
• Insulin resistance has been independently associated with the progression of CAC. A CAC progression study based on the Kaiser Permanente of Northern California database addressed this issue (Lee 2009). In the univariate analysis TC, LDL, HDL and triglycerides (TGs) were not even close to being significantly associated with progression over two years of follow-up. In multivariate analysis, progression was associated with age, female gender, African American decent, diabetes, fasting insulin, dyslipidemia (presumably high TGs plus low HDL), hypertension, diastolic BP and pulse pressure but not with lipid lowering medication.
In some of these studies, 30-50% of the subjects were on statin therapy at baseline. It is thus curious that statin use also turns up in multivariate analysis in some of these studies to be a positive risk factor. It has been suggested that the positive associations may have been caused by these studies enrolling a high percentage of individuals who had been given statins because they were perceived to be at the greatest risk (Elkeles 2010).
Recently published observational studies, including one that looked at event-free survival, find no association between LDL and cardiovascular/coronary heart disease (CVD/CHD) events among individuals with diabetes (Anand 2006, van Dieren 2011). A recent review even suggested that type 2 diabetes should not be considered a true risk equivalent for CHD (Riche 2007). The consistent finding of no association between TC or LDL cholesterol and the extent or progression of subclinical coronary atherosclerosis is consistent with studies in cohorts of individuals free of diabetes (Ware 2009). Given this, why are LDL targets and lipid lowering with statins central to primary prevention of adverse cardiac events in patients with diabetes? Intervention trials are thus of interest.
Primary and Secondary Prevention of Diabetic Complications with Statins
If one examines the 2011 ADA standard of care (American Diabetes Association 2011), three primary prevention lipid lowering studies are cited where it is possible to stratify both by diabetes and the presence or absence of CHD and CVD, thus obtaining information on the role of statins in risk reduction in the context of primary prevention of acute CHD events in type 2 diabetes. Their Table 11 indicates endpoint restriction to fatal CHD and non-fatal heart attack, but it is extrapolated to 10-year risk. It is thus of interest to look at the individual primary prevention trials. The results were as follows for absolute percent risk reduction (ARR) the numbers needed to treat to prevent one event (NNT), the relative risk reduction (RRR) and the untreated event rate (UTER) for the endpoint of fatal and non-fatal heart attack over 4-5 years (calculated from reported events over the study period). See Table 1.
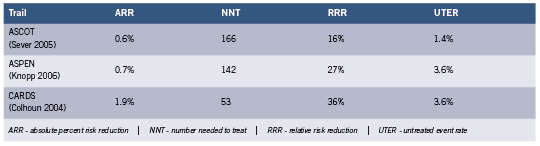
The HPS (MRC/BHF) trial (Collins 2003) is omitted because for the endpoint in question, the study included in the diabetic group individuals with arterial disease, including 33% with previous heart attack or other CHD and 18% with other occlusive artery disease. HPS only stratifies individuals with diabetes by prior CHD when the endpoint includes coronary events, all strokes, and coronary and non-coronary revascularization, which inflates the benefit.
The very small absolute risk reduction (average 1.1%) with large NNT seen in the above table was also found in studies of cohorts where diabetes is present in only a small fraction or absent. When meta-analyses were rigorously restricted to primary prevention statin trials (Wright 2010), absolute risk reduction for major CHD events was 1.0% (11 studies) and the NNT 100. There was no significant impact on mortality. The similarity to cohorts of individuals with diabetes is noteworthy.
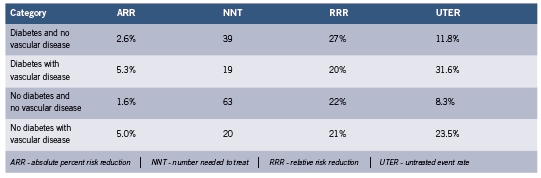
The largest meta-analysis of statin trials where both diabetes and primary vs. secondary prevention were stratified is the Cholesterol Treatment Trialists’ Collaborators (CCT) study. The results are summarized below for the composite endpoint of MI, coronary deaths, stroke, or coronary revascularization over 4-5 years, calculated from reported event numbers (Cholesterol Treatment Trialists’ (CTT) Collaborators 2008). See Table 2. Most participants in these trials were at elevated risk of adverse CVD events.
These results illustrate the greater impact of statins in secondary prevention. The result for individuals with diabetes with no vascular disease is somewhat higher than in CARDS, ASPEN and ASCOT discussed above, presumably because of the expanded endpoint which increases event rates. The results for no diabetes and no vascular disease are slightly higher than those reported above (Wright 2010), again for the same reason.
The CCT results tabulated above which include the NNT should be viewed with some caution since the combined studies average over somewhat different populations. The NNT as it is being used to illustrate clinical utility can only be applied for comparisons when the baseline absolute risks are similar. Also, the NNT decreases dramatically with the duration of studies (Breau 2009, Citrome 2011, McAlister 2008). In the CCT results, duration probably does not confuse the issue.
There is growing suspicion that a significant and perhaps major part of the action of statin drugs involves non lipid lowering (pleiotropic) effects, of which there is a long and impressive list containing actions that can influence acute events but have nothing to do with lower circulating cholesterol (Liao 2005, Mihos 2010, Sadowitz 2010). This significantly impacts the use of LDL lowering trials to prove that LDL is a causative factor. Some might argue that for secondary prevention it does not matter what the statin mechanism is as long as there is a modest (5%) absolute benefit.
Then there are the statin adverse effects. For liver dysfunction, cataracts and myopathy, the number needed to treat to harm one patient with statin therapy has been estimated for men at 142, 52 and 91 and for women 136, 33 and 259, respectively (Hippisley-Cox 2010). It is doubtful that cataracts disappear if statin therapy is terminated. It is also strongly suspected that adverse events are significantly under-reported, downplayed or prevalence data suppressed. The point at which the ARR is large enough and the adverse side effects small enough to justify treatment is of course debatable, and the attitudes of both patient and physician highly variable. Conservative clinicians might conclude that the treatment is not justified in general for very small ARRs, especially in the absence of symptomatic heart disease (Wright 2010)
Conclusions
Thus the obvious question. Does targeting LDL among individuals with diabetes free of cardiovascular disease make any sense when it rarely appears as significant if one examines correlations with the prevalence or progression of silent coronary atherosclerosis or coronary event free survival, or when one takes into account the very small absolute benefits seen in true primary prevention statin intervention trials? Furthermore, optimum LDL targets associated with guideline recommendations for diabetics have been described as not evidence based (Rutter 2011).
LDL is universally regarded as the “bad” cholesterol. If it is so bad, why is it consistently absent as a factor in studies of the risk of prevalence and progression of silent atherosclerosis, the prerequisite precursor of acute events? Statins are ineffective in this context for individuals free from diabetes (Henein 2010, Ware 2009), and individuals with diabetes as discussed above. Why does low or very low LDL characterize half of hospital admissions for heart attack (Al-Mallah 2009)? Why do the non-statin drugs that lower LDL not reduce the event risk, even in secondary prevention (Krumholz 2010)? The simple answer is that the association between heart disease and cholesterol is very much weaker than the conventional wisdom has led us to believe. A new paradigm is needed (de Lorgeril 2009, Ravnskov 2009, Rosch 2008).
The focus on relative rather than absolute risk reduction has resulted in widespread inflated perceptions of benefit, confused the risk/benefit analysis and produced a false sense of security among the millions of statin users. Note the disconnect between ARR and RRR in the above tables. A 20-30% relative risk reduction is impressive and strongly influences therapeutic decisions, but it can be associated with a 1%-2% or even smaller absolute event risk reduction. Patients need to be advised on the basis of absolute risk reductions. A strong case for this was made by Professor Peter Sawicki MD, PhD, from Köln in an invited lecture during a symposium on the cholesterol hypothesis (co-chaired by the author of this review) at the annual meeting of the European Association for the Study of Diabetes recently held in Lisbon, Portugal.
References
Al-Mallah,M.H., Hatahet,H., Cavalcante,J.L. and Khanal,S. Low admission LDLcholesterol is associated with increased 3-year all-cause mortality in patients with non ST segment elevation myocardial infarction. Cardiol. J 2009; 16(3): 227-233.
American Diabetes Association Standards of medical care in diabetes–2011. Diabetes Care 2011; 34 Suppl 1: S11-S61.
Anand,D.V., Lim,E., Darko,D., Bassett,P., Hopkins,D., Lipkin,D., Corder,R. and Lahiri,A. Determinants of progression of coronary artery calcification in type 2 diabetes role of glycemic control and inflammatory/vascular calcification markers. J Am Coll Cardiol. 2007; 50(23): 2218-2225.
Anand,D.V., Lim,E., Hopkins,D., Corder,R., Shaw,L.J., Sharp,P., Lipkin,D. and Lahiri,A. Risk stratification in uncomplicated type 2 diabetes: prospective evaluation of the combined use of coronary artery calcium imaging and selective myocardial perfusion scintigraphy. Eur Heart J 2006; 27(6): 713-721.
Breau,R.H., Fergusson,D. and Dahm,P. Evidence-based urology in practice: number needed to treat. BJU Int 2009; 104(1): 6-8.
Budoff,M.J., Yu,D., Nasir,K., Mehrotra,R., Chen,L., Takasu,J., Agrawal,N., Liu,S.T. and Blumenthal,R.S. Diabetes and progression of coronary calcium under the influence of statin therapy. American Heart Journal 2005; 149(4): 695-700.
Campayo,A., Gomez-Biel,C. and Lobo,A. Diabetes and Depression. Current Psychiatry Reports 2011; 13(1): 26-30.
Cholesterol Treatment Trialists’ (CTT) Collaborators Efficacy of cholesterol-lowering therapy in 18,686 people with diabetes in 14 randomised trials of statins: a meta-analysis. The Lancet 2008; 371(9607): 117-125.
Citrome,L. Number needed to treat: what it is and what it isn’t, and why every clinician should know how to calculate it. J Clin Psychiatry 2011; 72(3): 412-413.
Colhoun,H.M., Betteridge,D.J., Durrington,P.N., Hitman,G.A., Neil,H.A., Livingstone,S.J., Thomason,M.J., Mackness,M.I., Charlton-Menys,V. and Fuller,J.H. Primary prevention of cardiovascular disease with atorvastatin in type 2 diabetes in the Collaborative Atorvastatin Diabetes Study (CARDS): multicentre randomised placebocontrolled trial. Lancet 2004; 364(9435): 685-696.
Collins,R., Armitage,J., Parish,S., Sleigh,P. and Peto,R. MRC/BHF Heart Protection Study of cholesterol-lowering with simvastatin in 5963 people with diabetes: a randomised placebo-controlled trial. Lancet 2003; 361(9374): 2005-2016.
de Lorgeril,M. Disappointing recent cholesterol-lowering drug trials: is it not time for a full reappraisal of the cholesterol theory? http://michel. delorgeril. info/dwnl/wrnd/ DisappointingCholDrugTrials_wrnd2009. pdf). World Rev Nutr Diet 2009; 100: 80-89.
Elkeles,R.S. Coronary artery calcium and cardiovascular risk in diabetes. Atherosclerosis 2010; 210(2): 331-336. Elkeles,R.S., Feher,M.D., Flather,M.D., Godsland,I.F., Nugara,F., Richmond,W., Rubens,M.B. and Wang,D. The association of coronary calcium score and conventional cardiovascular risk factors in Type 2 diabetic subjects asymptomatic for coronary heart disease (The PREDICT Study). Diabet. Med 2004; 21(10): 1129-1134.
Elkeles,R.S., Godsland,I.F., Rubens,M.B., Feher,M.D., Nugara,F. and Flather,M.D. The progress of coronary heart disease in Type 2 diabetes as measured by coronary calcium score from electron beam computed tomography (EBCT): the PREDICT study. Atherosclerosis 2008; 197(2): 777-783.
Godsland,I.F., Elkeles,R.S., Feher,M.D., Nugara,F., Rubens,M.B., Richmond,W., Khan,M., Donovan,J., Anyaoku,V. and Flather,M.D. Coronary calcification, homocysteine, C-reactive protein and the metabolic syndrome in Type 2 diabetes: the Prospective Evaluation of Diabetic Ischaemic Heart Disease by Coronary Tomography (PREDICT) Study. Diabet. Med 2006; 23(11): 1192-1200.
Henein,M.Y. and Owen,A. Statins moderate coronary stenoses but not coronary calcification: Results from meta-analyses. Int J Cardiol. 2010; Published ahead of print, September 4, 2010.
Hippisley-Cox,J. and Coupland,C. Individualising the risks of statins in men and women in England and Wales: population-based cohort study. Heart 2010; 96(12): 939-947.
Janssen,I., Powell,L.H., Matthews,K.A., Cursio,J.F., Hollenberg,S.M., Sutton-Tyrrell,K., Bromberger,J.T. and Everson-Rose,S.A. Depressive symptoms are related to progression of coronary calcium in midlife women: The Study of Women’s Health Across the Nation (SWAN) Heart Study. Am Heart J 2011; 161(6): 1186-1191.
Knopp,R.H., d’Emden,M., Smilde,J.G. and Pocock,S.J. Efficacy and safety of atorvastatin in the prevention of cardiovascular end points in subjects with type 2 diabetes: the Atorvastatin Study for Prevention of Coronary Heart Disease Endpoints in non-insulin-dependent diabetes mellitus (ASPEN). Diabetes Care 2006; 29(7): 1478-1485.
Kobayashi,S., Oka,M., Maesato,K., Ikee,R., Mano,T., Hidekazu,M. and Ohtake,T. Coronary Artery Calcification, ADMA, and Insulin Resistance in CKD Patients. Clinical Journal of the American Society of Nephrology 2008; 3(5): 1289-1295.
Krumholz,H.M. and Hayward,R.A. Shifting views on lipid lowering therapy. BMJ 2010; 341: c3531.
Lee,K.K., Fortmann,S.P., Fair,J.M., Iribarren,C., Rubin,G.D., Varady,A., Go,A.S., Quertermous,T. and Hlatky,M.A. Insulin resistance independently predicts the progression of coronary artery calcification. American Heart Journal 2009; 157(5): 939-945.
Liao,J.K. and Laufs,U. Pleiotropic effects of statins. Annu Rev Pharmacol Toxicol 2005; 45: 89-118.
Martin,S.S., Qasim,A.N., Mehta,N.N., Wolfe,M., Terembula,K., Schwartz,S., Iqbal,N., Schutta,M., Bagheri,R. and Reilly,M.P. Apolipoprotein B but not LDL cholesterol is associated with coronary artery calcification in type 2 diabetic whites. Diabetes 2009; 58(8): 1887-1892.
Mazzone,T., Meyer,P.M., Kondos,G.T., Davidson,M.H., Feinstein,S.B., D’Agostino,R.B., Sr., Perez,A. and Haffner,S.M. Relationship of traditional and nontraditional cardiovascular risk factors to coronary artery calcium in type 2 diabetes. Diabetes 2007; 56(3): 849-855.
McAlister,F.A. The “number needed to treat” turns 20–and continues to be used and misused. CMAJ 2008; 179(6): 549-553.
Mihos,C.G., Salas,M.J. and Santana,O. The pleiotropic effects of the hydroxy-methylglutaryl- CoA reductase inhibitors in cardiovascular disease: a comprehensive review. Cardiol. Rev 2010; 18(6): 298-304.
Ravnskov,U. and McCully,K.S. Review and Hypothesis: Vulnerable plaque formation from obstruction of Vasa vasorum by homocysteinylated and oxidized lipoprotein aggregates complexed with microbial remnants and LDL autoantibodies. Ann Clin Lab Sci 2009; 39(1): 3-16.
Riche,D.M. and McClendon,K.S. Role of statins for the primary prevention of cardiovascular disease in patients with type 2 diabetes mellitus. Am J Health Syst. Pharm. 2007; 64(15): 1603-1610.
Rosch,P.J. Cholesterol does not cause coronary heart disease in contrast to stress. Scand Cardiovasc J 2008; 42(4): 244-249.
Rutter,M.K. and Nesto,R.W. Blood pressure, lipids and glucose in type 2 diabetes: how low should we go? Re-discovering personalized care. Eur Heart J 2011. Sadowitz,B., Maier,K.G. and Gahtan,V. Basic science review: Statin therapy–Part I: The pleiotropic effects of statins in cardiovascular disease. Vasc Endovascular. Surg 2010; 44(4): 241-251.
Saremi,A., Moritz,T.E., Anderson,R.J., Abraira,C., Duckworth,W.C. and Reaven,P.D. Rates and determinants of coronary and abdominal aortic artery calcium progression in the Veterans Affairs Diabetes Trial (VADT). Diabetes Care 2010; 33(12): 2642-2647.
Sever,P.S., Poulter,N.R., Dahlof,B., Wedel,H., Collins,R., Beevers,G., Caulfield,M., Kjeldsen,S.E., Kristinsson,A., McInnes,G.T., Mehlsen,J., Nieminen,M., O’Brien,E. and Ostergren,J. Reduction in cardiovascular events with atorvastatin in 2,532 patients with type 2 diabetes: Anglo-Scandinavian Cardiac Outcomes Trial–lipid-lowering arm (ASCOT-LLA). Diabetes Care 2005; 28(5): 1151-1157.
van Dieren,S., Nothlings,U., Van der Schouw,Y.T., Spijkerman,A.M., Rutten,G.E., van der,A.D., Sluik,D., Weikert,C., Joost,H.G., Boeing,H. and Beulens,J.W. Non-fasting lipids and risk of cardiovascular disease in patients with diabetes mellitus. Diabetologia 2011; 54(1): 73-77.
Ware,W.R. The mainstream hypothesis that LDL cholesterol drives atherosclerosis may have been falsified by non-invasive imaging of coronary artery plaque burden and progression. Med Hypotheses 2009; 73(4): 596-600.
Wolfe,M.L., Iqbal,N., Gefter,W., Mohler,E.R., III, Rader,D.J. and Reilly,M.P. Coronary artery calcification at electron beam computed tomography is increased in asymptomatic type 2 diabetics independent of traditional risk factors. J Cardiovasc Risk 2002; 9(6): 369-376.
Wright,J. Do statins have a role in primary prevention? An update. Theraeutics Initative (Theraputics Letter) 2010; 77(March-April 2010, http://ti.ubc.ca/letter77).













