This study provided 198 healthy men 20 to 50 years of age with goserelin acetate (to suppress endogenous testosterone and estradiol) and randomly assigned them to receive a placebo gel or 1.25g, 2.5g, 5g, or 10g of testosterone gel daily for 16 weeks. Another 202 healthy men received goserelin acetate, placebo gel or testosterone gel, and anastrozole (to suppress the conversion of testosterone to estradiol). Changes in the percentage of body fat and in lean mass were the primary outcomes. Subcutaneous- and intraab- dominal-fat areas, thigh-muscle area and strength, and sexual function were also assessed. The results showed that the percentage of body fat increased in groups receiving placebo of 1.25g or 2.5g of testosterone daily without anastrozole (mean testosterone level, 44±13 ng per deciliter, 191±78 ng per deciliter, and 337±173 ng per deciliter, respectively). Lean mass and thigh-muscle area decreased in men receiving placebo and in those receiving 1.25g of testosterone daily without anastrozole. Leg-press strength fell only with placebo administration. In general, sexual desire declined as the testosterone dose was reduced. The authors conclude that the amount of testosterone required to maintain lean mass, fat mass, strength, and sexual function varied widely in men. N Engl J Med. 2013 September. PMID: 24024838
KLOX Technologies announces approval in Canada of acne vulgaris treatment
KLOX Technologies Inc. announced the approval in Canada of the Klox Biophotonic System (also known as the Lumigel Cleanse System) for the treatment of acne vulgaris after receiving this week a Medical Device License from the Therapeutic Products Directorate – Medical Devices Bureau of Health Canada. The Lumigel Cleanse System is a first-in-class innovative acne vulgaris treatment which has shown in clinical trials a superior profile in terms of combined safety and efficacy. “KLOX is proud to receive its first approval here in Canada. This important milestone fits perfectly into our regulatory strategy that we are pursuing around the world and to that end the company has also filed in Europe for a CE mark,” said Dr. Lise Hébert, President and CEO of KLOX. With this new, cutting edge topical product, of a different category than photodynamic therapy, KLOX is now actively pursuing its goals which are two-fold: to create important returns for its shareholders and to advance its large pipeline of products for unmet needs in dermatology. Acne vulgaris is a chronic disease that can have devastating physical, social and psychological effects on those who suffer from it, particularly in moderate to severe cases. Acne vulgaris affects over 5 million people in Canada, and approximately 150 million people worldwide. It accounts for over 30% of dermatology visits and over 40% of those suffering from this disease are looking for treatments with a better safety and efficacy profile than what is currently available. Many of the topical, oral and photodynamic treatments for acne vulgaris are characterized by inconsistent efficacy/persistence, systemic treatment schedules, lower patient tolerability and in most cases, side-effects. “Based on the new technology, the KLOX Lumigel Cleanse System is a first-in-line Class II medical device with an unmatched clinical profile in terms of efficacy and safety. Patients will appreciate its visible and long-lasting effects, as demonstrated clinically, with the benefit of being non-invasive in nature. On the other hand, practitioners will be able to offer an easy-to-administer and accessible treatment option,” said Dr. Steven Bernstein, Chief of Dermatology Surgery at the University of Montreal Hospital Center (CHUM).
Ottawa Life Launches 6-Part Year-Long Series on the Ottawa Integrative Cancer Centre
The Ottawa Integrative Cancer Centre (OICC) opened in 2011 as a research and new cancer care centre. The goal is prevention and improving the lives of those who are living with cancer. It works collaboratively with other health professionals and practices, integrating the use of naturopathic and other compatible whole-person treatments with standard cancer care to reach their goals. The OICC Founder and Executive Director, Dugald Seely, was selected as one of the Top 25 People in the Capital in 2013 by Ottawa Life for his vision and leadership in starting the first integrative cancer care and research centre in central and eastern Canada.
Vitamin B12
Vitamin B12
Update On Clinical Applications
Abstract
Vitamin B12 is an essential co-factor for methionine synthase and L-methyl-malonyl-CoA mutase, which are required in DNA synthesis and vital in hematopoietic pathways. There are several risk factors for B12 deficiency and associated anemia, including vegetarian or vegan diets and increasing age. Supplemental vitamin B12 is overwhelmingly safe and although there is insufficient evidence to establish the best long term route of administration for B12, the evidence suggests that oral, intramuscular, and parenteral administration are equally effective. Homocysteine and vitamin B12 levels should be routinely checked in individuals with cognitive decline and Alzheimer’s disease, as vitamin B12 is a viable treatment option. The totality of evidence is unclear as to the independent impact of vitamin B12 in relation to cardiovascular risk, but key studies will be discussed. Vitamin B12 is effective at ameliorating the symptoms of depression, as shown by improvements in various validated questionnaires. This article reviews the trials conducted using supplemental vitamin B12 in the treatment of these various health conditions and provides a clinical update.
Introduction
Vitamin B12, also known as cobalamin, can come in many forms including cyano-, methyl-, and hydroxy-cobalamin, among others (O’Leary 2010). It is involved in many physiological pathways in the body and a deficiency causes megaloblastic anemia, as well as a potential host of other damaging consequences. Serum B12 is currently the principle means of assessing B12 status (Carmel 2009), and as discussed below this test alone is insufficient for accurately establishing all stages of B12 deficiency. The addition of holotranscobalamin, methylmelonic acid, and homocysteine to the standard serum B12 measurement provides a more accurate assessment of functional B12 status (Herrmann 2003). Utilizing this expanded four test profile to assess B12 status can help to identify earlier stages of deficiency and initiate appropriate treatment before the manifestation of clinically evident deficiency symptoms (Herrmann 2003). Supplemental vitamin B12 is overwhelmingly safe and no toxic effects have been encountered from administration, indicating that B12 is essentially nontoxic (Food and Nutrition Board 1998). This is applicable when administering amounts at or below the recommended dietary allowance. However, there is limited data for high-dose B12 and there have been some reports of mild side effects such as rash, pruritus, and diarrhea. There is also not enough data to make any conclusions about the safety of large doses of vitamin B12 in pregnancy and breast feeding. In addition to anemia, vitamin B12 has been used as a treatment for numerous health issues, including for cardiovascular health, in depression, and for cognitive health. This article will review the evidence available in each of these areas and update practitioners on the clinical applications of vitamin B12.
Anemia Recent estimates suggest up to 14% of anemias may be associated with folate or vitamin B12 deficiency (Pang 2012). Vitamin B12 is an essential co-factor for methionine synthase and L-methyl-malonyl-CoA mutase, which are required in DNA synthesis and thus vital in hematopoietic pathways (O’Leary 2010). There are several risk factors for B12 deficiency and associated anemia, including vegetarian and vegan diets (Pawlak 2013), and increasing age (Chatthanawaree 2013, Park 2006). An important and often overlooked risk of vitamin B12 deficiency is depletion from drug interactions including proton pump inhibitors, metformin, anti-seizure medications, and nitric oxide (Mazokopakis 2008). Also of interest is a recent study indentifying an increased risk of vitamin B12 (as well as folate and iron) deficiency in patients following gastric bypass, with over 60% developing anemia within three years (Vargas-Ruiz 2008).
The most common anemia associated with vitamin B12 is pernicious anemia (PA), a macrocytic anemia caused by atrophic gastritis and an associated loss of intrinsic factor (IF), which is required to cleave vitamin B12 from protein sources thereby facilitating absorption (Stabler 2013). PA is most common in the seventh or eighth decade of life, with milder atrophic gastritis affecting an estimated 20% of older individuals (Stabler 2013). Long term risk factors of neurologic complications including peripheral neuropathy, and lateral spinal cord and cerebrum lesions, can be prevented or reversed if PA is recognized early in disease progression and supplemental vitamin B12 is provided (Annibale 2011). Since oral supplements provide vitamin B12 unbound to protein even in the absence of IF, absorption can still occur. PA has an insidious onset with non-descript signs and symptoms including fatigue, weakness, headaches, lack of concentration, paresthesias, and digestive disturbance, making it challenging, but important to diagnose early (O’Leary 2010). Patients with PA should be given supplemental doses of at least 500-1000mcg (O’Leary 2010).
Several studies investigating the merits of intramuscular, parenteral, and oral administration routes of vitamin B12 have concluded that in the short term, oral supplementation is equally as effective as other routes in cases of pernicious anemia and malabsoprtion (Seal 2002). Complicating the implications of study results is the common use of the cyanocobalamin form of vitamin B12 (hydroxy, and methyl are available commercially and used clinically), variety in dosing regimens, and unclear long-term comparisons of administration routes. Although cost of oral supplementation is significantly less than IM or parenteral, both IM and parenteral may be more effective in cases where compliance is an issue (Drug and Therapeutics Bulletin 2009).
Establishing parameters to measure B12 deficiency is not straightforward. Traditionally the lower limit of reference ranges for serum B12 reflected the level at which megaloblastic anemia normally presented (O’Leary 2010). While serum testing of total vitamin B12 is available and regularly used, it may misrepresent vitamin B12 status as it includes B12 in the form of hepatocorrin, which is biologically unavailable (Green 2011). More recently holotranscobolamin II, the available form of vitamin B12, has been recognized as a useful measure of B12 status (Green 2011). Measurements of other metabolic markers of vitamin B12 status are helpful in determining not only vitamin B12 status, but also differentiating vitamin B12 and folate deficiencies. Elevated levels of both homocysteine and methylmalonic acid are regularly noted in B12 deficiency, while homocysteine elevation with normal methylmalonic acid levels are more common in folate deficiency, although both markers may be elevated with normal B12 and folate status (Green 2011). Vitamin B12 deficiency is a risk factor for anemia and routine screening of haematological markers along with serum vitamin B12, homocysteine, and methylmalonic acid should be evaluated in individuals at risk of B12 deficiency, specifically elderly and vegetarian populations. Additionally, appropriate identification of PA is crucial in preventing serious long-term consequences. There is insufficient evidence to establish the best long term route of administration for B12, although oral, intramuscular, and parenteral administration appear to be equivocal in the short-term.
Cardiovascular Health Many prospective intervention studies have examined the role of vitamin B12 in cardiovascular disease. Unfortunately, there have been a number of limitations to these trials, including inadequate treatment with vitamin B12 and confounding effects from other simultaneous treatments (Righetti 2009). One recent systematic review of cohort studies included seven studies with varying populations, length of follow-up, study outcomes, and data analysis (Rafnsson 2011). Only one high-quality study reported that low B12 increased the risk of incident cerebral ischaemia. After controlling for homocysteine, the association persisted although weakened, suggesting that the effects of low B12 were only partly mediated by homocysteine. In two studies, higher B12 levels were associated with a greater risk of total mortality and combined fatal and non-fatal coronary events (Rafnsson 2011).
In examining other trials that were excluded from the aforementioned systematic review, there are two that stand out. The first is the Vitamin Intervention for Stroke Prevention Trial (Spence 2007). In the original interpretation of the trial, it was shown that vitamin therapy (B12, B6, folate) had no benefit for homocysteine lowering. However, there were problems with the trial, including the lack of a placebo control, the use of a multiple vitamin with low doses of vitamins, patients in both arms of the trial received injections of B12, and folate fortification of grain products in North America coincided with the initiation of the trial. Upon further interpretation of a subgroup from which patients who could not have responded to the study treatment were excluded, there was a significant reduction of stroke, coronary events, and death. When stratified by median entry level of B12, serum B12 status was the key determinant of response, with those with high serum B12 at entry who received high-dose vitamin (0.4mg cyano-cobalamin was considered the high-dose) had a 33% reduction of events compared to those with low B12 who received low-dose vitamins (Spence 2007). The second trial of interest is the Heart Outcomes Prevention Evaluation 2 Trial (Lonn 2006). In this trial, they evaluated whether long-term therapy with folic acid 2.5mg and vitamins B6 50mg and B12 1mg reduced the risk of major cardiovascular events in a high-risk population. The results showed a 25% significant reduction of stroke with vitamin therapy (p = 0.03), but the authors considered this a chance finding (Lonn 2006). Overall, the totality of evidence is unclear as to the independent impact of vitamin B12 in relation to cardiovascular risk. It is also difficult to make large generalizations based on studies with a range of doses, with limited experimental evidence available for supplemental doses above 1mg of B12.
Depression Low levels of vitamin B12 have been measured in patients with depressive disorders and in some studies, women with metabolically significant deficiencies have been found to have a two-fold higher risk of depression (Penninx 2000). Although many studies examine both folate and vitamin B12, one study has suggested B12 deficiency is independently related to depressive disorders (Tiemeier 2002). A separate study conducted with outpatients diagnosed with major depressive disorder looked at vitamin B12 levels and the consequent six-month treatment outcomes (Hintikka 2003). The authors examined haematological indices, including erythrocyte folate and serum vitamin B12 levels. The study included 115 outpatients with DSM-III-R major depressive disorder at baseline. The 17-item Hamilton Depression Rating Scale was compiled. The results showed that higher vitamin B12 levels were significantly associated with a better outcome at six-months (Hintikka 2003). More recently, a Finnish population-based study examined the association between vitamin B12 levels and melancholic depressive symptoms (Seppala 2013). Subjects were randomly selected from the National Population Register as part of the Finnish diabetes prevention program and the study consisted of 2806 participants. Health examinations were carried out and depressive symptoms were based on the Beck Depression Inventory. Groups were further divided by melancholic or non-melancholic subgroups based on the DSM-IV. The results showed that the mean vitamin B12 level was associated with melancholic depressive symptoms (Seppala 2013).
Many case studies have reported that supplemental vitamin B12 and folic acid can improve the symptoms of depression (Mitchell 2007). In this study, a sample of 12 outpatients with dysthymic disorder was supplemented with 800mcg of folic acid and 1000mcg of vitamin B12 for six weeks. Two instruments were used to measure depressive symptoms: the Beck Depression Inventory- II and the Symptom Checklist-90-Revised. At the end of the six weeks, the raw and t-scores of each of the participants indicated a reduction in the reported levels of symptoms of depression (Mitchell 2007). One large study examined the effects of dietary intake of vitamin B6, folate, and vitamin B12 on depressive symptoms (Skarupski 2010). The study sample consisted of 3503 adults aged over 65 years from the Chicago Health and Aging project. Dietary assessment was made by food-frequency questionnaire and incident depression was measured by the presence of at least four depressive symptoms from the 10-item version of the Center for Epidemiologic Studies Depression scale. The results showed that the higher total intakes of vitamin B6 and B12 were associated with a decreased likelihood of depression for up to 12 years of follow-up, after adjustments were made, including for antidepressant medication use. Each 10 additional milligrams of vitamin B6 and 10 additional micrograms of vitamin B12 were associated with 2% lower odds of depressive symptoms per year (Skarupski 2010). Finally, the B-VITAGE trial is a double-blind, placebo-controlled, randomized trial of homocysteine lowering treatment of depression in later life that is currently in progress (Ford 2010). Participants will receive daily citalopram (20-40mg), plus vitamin B12 (0.4mg), B6 (25mg), and folic acid (2mg) compared to citalopram plus placebo. Over the course of 52 weeks, depressive symptoms will be measured using validated questionnaires. This trial will clarify whether the systematic use of B-vitamins improve the response of older adults to standard antidepressant treatment. Although teasing out the specific effects of vitamin B12 is difficult, the evidence that is available suggests it is both a safe and effective option in the treatment of depressive symptoms.
Cognitive Decline and Alzheimer Disease Vitamin B12 deficiency and elevations in associated biomarkers of low vitamin B12 status, are associated with cognitive decline (CD) (Riggs 1996), including Alzheimer Disease (AD) (Wang 2001). The mechanism implicated in the relationship between Vitamin B12 status and CD is the homocysteine lowering effect of B12 (Werder 2010). Studies have shown that in individuals with elevated homocysteine levels and CD, reducing homocysteine through therapies including combinations of folate, vitamin B12, and vitamin B6, has a favourable benefit on cognitive function (Nilsson 2001).
Results of RCTs evaluating vitamin B12 supplementation as a treatment for CD and AD are mixed (Malouf 2003). Although a number of RCTs are available, due to small sample size and poor study design, a large number do not provide significant power with which to draw conclusions (Malouf 2003). Factors including dose and duration of treatment, extent and duration of cognitive decline, homocysteine levels, vitamin B12 status (and laboratory techniques used for evaluation), all confound results. A 2003 Cochrane review included only two RCTs evaluating vitamin B12 supplementation in the treatment of CD and AD, and concluded insufficient evidence of efficacy, primarily due to small trial size (Malouf 2003). Several newer studies demonstrate positive outcomes with supplemental B12 (0.5mg) in CD, with the largest effect in individuals with elevated homocysteine levels and with the shortest duration of cognitive decline (De Jager 2013). Additionally, studies have demonstrated a decrease in accelerated brain atrophy in both AD (Douaud 2013) and mild CD with homocysteine-lowering therapy by vitamin B12, folate and vitamin B6 (Smith 2010).
Supplemental B12 in combination with folate also shows benefit in as a preventative therapy in decreasing cognitive decline in older individuals with depressive disorders. A 2012 RCT showed significant improvement in cognitive measures after 24 months of supplementation of Vitamin B12 (100mcg) and folate in individuals over 60 (Walker 2012). Of note, in combination therapy with folate, one RCT showed no statistically significant improvement in CD with supplementation, which was likely due to the inclusion of individuals with low or normal homocysteine levels, suggesting lowering homocysteine below normal levels is of no benefit in CD (Pratico 2009). Although there is conflicting evidence represented in research regarding vitamin B12 supplementation in the treatment of CD and AD, there is significant evidence that supplementation in individuals with elevated homocysteine levels early in CD is effective (Walker 2012). Based on the relative safety of vitamin B12 administration and possible degree of benefit, homocysteine and vitamin B12 levels should be routinely checked in individuals with cognitive decline and vitamin B12 (along with folate and vitamin B6) considered as a viable treatment option to lower homocysteine and improve markers of mild CD and AD.
Conclusion
Supplemental vitamin B12 is overwhelmingly safe and no toxic effects have been encountered, especially at doses at or below 1000mcg. Regarding optimal route of administration, there is insufficient evidence to establish the best long term option, but oral, intramuscular, and parenteral administration are all adequate in the short-term. It is imperative to detect vitamin B12 deficiency in those at risk, especially the elderly. Vitamin B12 is an effective treatment for anemia in these cases. Despite some promising results, the totality of evidence is unclear as to the independent impact of vitamin B12 in relation to cardiovascular risk. Vitamin B12 is effective in treating the symptoms of depression, as shown by improvements in various validated questionnaires. Additional well-designed trials are currently in progress. Finally, earlier studies on CD and AD had small sample sizes and poor designs, but newer trials have shown that B12 supplementation in individuals with elevated homocysteine levels early in CD is effective, even in small doses.
References
Annibale B, Lahner E, Fave GD. Diagnosis and Management of Pernicious Anemia. Curr Gastroenterol Rep. 2011;13(6):518–24.
Carmel, R. Mild transcobalamin I (haptocorrin) deficiency and low serum cobalamin
concentrations. Clin Chem. 2003, 49(8):1367-74.
Chatthanawaree W. Biomarkers of cobalamin (vitamin B12) deficiency and its application. J Nutr Health Aging. 2011;15(3):227–31.
De Jager C a, Oulhaj A, Jacoby R, Refsum H, Smith AD. Cognitive and clinical outcomes of homocysteine-lowering B-vitamin treatment in mild cognitive impairment: a randomized controlled trial. Int J Geriatr Psychiatry. 2012;27(6):592–600.
Douaud G, Refsum H, De Jager C a, Jacoby R, Nichols TE, Smith SM, Smith AD. Preventing Alzheimer’s disease-related gray matter atrophy by B-vitamin treatment. Proceedings of the National Academy of Sciences of the United States of America. National Academy of Sciences. 20418; 2013;110(23):9523–8.
Drug and Therapeutics Bulletin. Oral or intramuscular vitamin B 12? DTB BMJ. 2009;47(2):19–21.
Food and Nutrition Board. Dietary reference intakes for thiamin, riboflavin, niacin, vitamin B6, folate, vitamin B12, pantothenic acid, biotin, and choline. Washington, DC: National Academy Press, 1998.
Ford AH, Flicker L, McCaul K, van Bockxmeer F, Hegarty S, Hirani V, Fenner S, Almeida OP. The B-VITAGE trial: a randomized trial of homocysteine lowering treatment of depression in later life. Trials. 2010;11:8
Green R. Indicators for assessing folate and vitamin B 12 status and for monitoring the efficacy of intervention strategies. Am J Clin Nutr. 2011;94(suppl):666S–72S.
Herrmann W, Schorr H, Obeid R, Geisel J. Vitamin B-12 status, particularly holotranscobalamin II and methylmalonic acid concentrations, and hyperhomocysteinemia in vegetarians. Am J clin Nutr 2003;78:131-6
Lonn E, Held C, Arnold JM, Probstfield J, McQueen M, Micks M, Pogue J, Sheridan P, Bosch J, Genest J, Yusuf S, HOPE-2 Investigators. Rationale, design and baseline characteristics of a large, simple, randomized trial of combined folic acid and vitamins B6 and B12 in high risk patients: the Heart Outcomes Prevention Evaluation (HOPE)-2 trial. Can J Cardiol. 2006; 22(1):47-53
Malouf R, Areosa Sastre A. Vitamin B12 for Cognition. Cochrane Database Syst Rev 2003;3.
Mazokopakis EE, Starakis IK. Recommendations for diagnosis and management of metformin-induced vitamin B12 (Cbl) deficiency. Diabetes Res Clin Pr. 2012; 97(3):359–67.
Mitchell JA. The Effect of Folic Acid and B12 on Depression: Twelve Case Studies. J Orthomolecular Med. 2007;22(4):183-192
O’Leary F, Samman S. Vitamin B12 in health and disease. Nutrients. 2010;2(3):299–316.
Nilsson K, Gustafson L, Hultberg B. Improvement of cognitive functions after cobalamin/folate supplementation in elderly patients with dementia and elevated plasma homocysteine. Int J Geriatr Psychiatry. 2001;16(6):609–14.
Pang WW, Schrier SL. Anemia in the elderly. Curr Opin Hematol. 2012;19(3):133–40.
Park S, Johnson MA. What is an Adequate Dose of Oral Vitamin B 12 in Older People with Poor Vitamin B 12 Status? Nutr Rev. 2006;64(8):373–8.
Pawlak R, Parrott SJ, Raj S, Cullum-Dugan D, Lucus D. How prevalent is vitamin B(12) deficiency among vegetarians? Nutr Rev. 2013;71(2):110–7.
Penninx BWJH, Guralink JM, Ferucci L, Fried LP, Allen RH, Stabler SP. Vitamin B12 deficiency and depression in physically disabled older women: Epidemiologic evidence from the women’s health and aging study. Am J Psychiatry. 2000; 157:715-721
Pratico D. High-dose vitamin supplements and Alzheimer disease. JAMA. 2009;301(10):1020–1.
Rafnsson SB, Saravanan P, Bhopal RS, Yajnik CS. Is a low blood level of vitamin B12 a cardiovascular and diabetes risk factor? A systematic review of cohort studies. Eur J Nutr 2011;50:97-106
Righetti M. Protective effect of vitamin B therapy on bone and cardiovascular disease. Recent Pat Cardiovasc Drug Discov. 2009;4(1):37-44
Riggs KM, Spiro A, Tucker K, Rush D. Relations of vitamin B-12 , vitamin B-6, folate, and homocysteine to cognitive performance in the Normative Aging Study. Am J Clin Nutr. 1996;63(3):306–14.
Seal E, Metz J, Flicker L, Melny J. Oral Vitamin B 12 Supplementation in Older Patients with Subnormal or Borderline Serum Vitamin B 12 Concentrations. J Am Geriatr Soc. 2002;50:146–51.
Seppala J, Koponen H, Kautiainen H, Eriksson JG, Kampman O, Leiviska J, Mannisto S, Mantyselka P, Oksa H, Ovaskainen Y, Viikki M, Vanhala M, Seppala J. Association between vitamin b 12 levels and melancholic depressive symptoms: a Finnish population-based study. BMC Psychiatry. 2013;13(1):145.
Skarupski KA, Tagney C, Li H, Ouyang B, Evans DA, Morris MC. Longitudinal association of vitamin B-6, folate, and vitamin B-12 with depressive symptoms among older adults over time. Amer J Clin Nutr. 2010;92(2):330-5
Smith DA, Smith SM, de Jager CA, Whitbread P, Johnston C, Agacinski G, Oulhaj A, Bradley KM, Jacoby R, Refsum H. Homocysteine-lowering by B vitamins slows the rate of accelerated brain atrophy in mild cognitive impairment: a randomized controlled trial. PloS One. 2010;5(9):e12244.
Spence JD. Perspective on the efficacy analysis of the Vitamin Intervention for Stroke Prevention trial. Clin Chem Lab Med 2007;45(12):1582-5.
Stabler SP. Clinical practice. Vitamin B12 deficiency. New Engl J Med. 2013;368(2):149–60.
Tiemeier H, van Tuijl HR, Hofman A, Meijer J, Kilaan AJ, Breteler MM. Vitamin B12, folate, and homocysteine in depression: the Rotterdam study. Arch Gen Psychiatry. 2002; 159:2099-2101
Vargas-Ruiz AG, Hernández-Rivera G, Herrera MF. Prevalence of iron, folate, and vitamin B12 deficiency anemia after laparoscopic Roux-en-Y gastric bypass. Obes Surg. 2008;18(3):288–93.
Walker JG, Batterham PJ, Mackinnon AJ, Jorm AF, Hickie I, Fenech M, Kljakovic M, Crisp D, Christensen H. Oral folic acid and vitamin B-12 supplementation to prevent cognitive decline in community-dwelling older adults with depressive symptoms — the Beyond Ageing Project: a randomized controlled trial. Amer J Clin Nutr. 2012;95(1):194–203.
Wang H, Wahlin Å, Basun H, Fastbom J. Vitamin B 12 and folate in relation to the development of Alzheimer’s disease. Neurology. 2001; 56(9);1188-94.
Werder SF. Cobalamin deficiency, hyperhomocysteinemia, and dementia. Neuropsychiatr Dis Treat. 2010;6:159–95.
Diabetes Medications
Diabetes Medications
Reduced risk of disease-related complications?
Abstract
It is not debatable that the elevated blood glucose levels associated with diabetes represent a serious risk marker for macrovascular and microvascular adverse developments and events, and that the risk increases with the average of the daily fluctuations. This provides the principal rationale for maintaining both fasting blood glucose and the indicator of the long- term average, glycated hemoglobin, at levels close to normal. More than a dozen prescription drugs are currently used individually or in combination to treat type 2 diabetes. They act by decreasing liver glucose production, increasing insulin secretion, increasing insulin sensitivity, inhibiting glucagon release and slowing the absorption of carbohydrates. These drugs decrease serum glucose and glycated hemoglobin with the goal of glucose reduction to targets approaching normal, a result that is generally believed will significantly decrease the incidence of the complication associated with the disease. Failure to achieve blood glucose control generally results in additional drugs and even insulin, and is frequently termed intensive treatment. As will be discussed, there are not only new studies but recent meta-analyses that have examined the impact of intensive blood glucose lowering on complications in type 2 diabetes. Evidence will be presented that in fact glucose lowering, either intensive or not, is a failed therapy if judged by the impact on clinical complications, a failure termed by some medical scientists the Diabetes Paradox.
Introduction
Even before the beginning of the 20th century, carbohydrate restriction was the established medical response to diabetes and obesity. This changed dramatically in the early 1960s when the hypothesis that fat and in particular saturated fat increased the risk of both heart disease and cancer was accepted as true without adequate evidence and profoundly altered diets. The macronutrients that were most effective in driving up blood sugar, elevating insulin, and viewed as causing insulin resistance, beta cell dysfunction and diabetes became the main components of the so-called healthy, low fat diet in the developed world. These were mostly carbohydrates and as the trend accelerated, these were more refined and had higher glycemic activity. Then drugs became available which would lower blood sugar and its daily average and offered a logical solution. The number of anti-hyperglycaemic agents increased rapidly. If used intensively, the drugs were able to bring glucose levels down to somewhat above normal and this was viewed as a success. Thus the blood glucose elevations associated with diets rich in carbohydrates were counteracted, once diabetes had developed, by daily dosing with antihyperglycemic drugs. The impact on complications, the principal reason for glucose control, is however a developing story and the subject of this article. In what follows, the evidence will be presented that once one has diabetes, controlling both the daily fluctuations and lowering the long-term average of blood glucose with pharmaceutical intervention in fact has almost no impact on the incidence of complications. This is not good news for patients with diabetes who obviously have reason to be seriously concerned about the need to prevent complications.
Standard vs. Intensive glucose control
There has been great interest in intensive blood glucose control and the natural (or perhaps simplistic) assumption has been that lowering levels to the pre-diabetic range with multiple drugs, insulin and dose escalation would be beneficial. The wake-up call came in 2008. Two papers simultaneously published in the New England Journal of Medicine reported on studies designed to address this issue with large cohorts. The ACCORD study randomized 10,251 patients with type 2 diabetes to receive intensive therapy targeted with polypharmacy, increased doses and insulin if necessary, all directed at getting HbA1c down from a median level of 8.1% to below 6.0%, generally considered high-normal or pre-diabetic. The controls received standard drug therapy targeting a level of 7.0% to 7.9%. As compared with standard therapy, the use of intensive therapy for 3.5 years increased mortality resulting in early trial termination and did not significantly reduce major cardiovascular events (Gerstein 2008).
The ADVANCE study randomized 11,140 patients with type 2 diabetes to either standard glucose control using mostly oral glycemic drugs or an intensive intervention group mainly using the modified release sulfonylurea Gliclazide, plus other drugs including insulin if needed to achieve a HbA1c of 6.5%. For over a median five- year follow-up, there was a small (1.9%) absolute decrease in risk associated with the primary endpoint, combined micro- and macrovascular events, in the intensive treatment group. But this was due to new or worsening nephropathy. Among 14 secondary endpoints, only new-onset miroablinuria showed a decreased absolute risk of 2%. Thus there was no impact on the remaining common complications of type 2 diabetes including all cause mortality, major or all coronary events, all cerebrovascular events, peripheral vascular events, visual deteriation or new or worsening neuropathy (Patel 2008).
A smaller trial, the Veterans Affairs Diabetes Trial (VADT) that reported in 2009, involved a comparison between intensive and standard glucose control in patients with type II diabetes with suboptimal response to therapy. In this study 1791 men, mean age 60, were randomized to two groups with a median follow-up of 5.6 years. Median HbA1c dropped from 8.4% in the standard therapy group to 6.9% in the intensive group. The primary endpoint was the time from randomization to first occurrence of a major cardiovascular event, which was a composite of MI, stroke, deaths from cardiovascular causes, congestive heart failure, surgery for vascular disease, inoperable coronary disease and amputation for gangrene. The comparison was with standard drug therapy. It was found that intensive glucose control had no significant impact on the endpoints of major cardiovascular events, death or microvascular complications with the exception of evidence of the progression to kidney disease. Statistically significant increased incidence of nephropathy was seen in subgroup analysis when micro- and macroalbuminuria were combined (Duckworth 2009).
In 2011 a systematic review and meta-analysis of randomized trials concerning intensive glycemic control appeared in BMJ (Hemmingsen 2011). No impact on all cause mortality was found. With regard to other endpoints, the authors found that data available remained insufficient to prove or refute a relative risk reduction of CVD mortality, non-fatal MI, composite microvascular complications, or retinopathy at a relative risk reduction threshold they considered clinically significant, but identified a 30% increase in severe hypoglycemia.
Prompted by the ADVANCE and VADT results and earlier studies related to kidney complications, a study involving meta-analysis reporting in 2012 investigated if intensive vs. standard glucose control decreased significant renal clinical outcomes such as doubling of serum creatinine levels, end-stage renal disease or death from renal disease during the years of follow-up. No evidence was found when seven trials with follow-up from two to 15 years were evaluated (Coca 2012).
Metformin monotherapy
The standard care almost always involves either metformin alone or metformin plus another drug. Since the American Diabetes Association (ADA) recommends metformin as the first-line of standard care after diagnosis of type 2 diabetes (ADA 2012), to what extent does it reduce the macro- and microvascular complications of diabetes when compared with a placebo or non-drug treatment such as diet? This may seem like an odd issue bring up. Metformin has been used for decades. Metformin is also the most common standard against which other drugs are judged in clinical trials.
In 2012 a meta-analysis was published where the efficacy of metformin was examined. The trials were either metformin vs. diet or non-drug care or a placebo, metformin plus a sulphonylurea vs. the sulphonylurea alone, or metformin plus insulin vs. insulin plus a placebo or metformin vs. total withdrawal from the drug. For the above comparisons, no significant benefit was found for all-cause mortality, cardiovascular mortality, all MI, all strokes, congestive heart failure, peripheral vascular events, amputation, or microvascular events (Boussageon 2012).
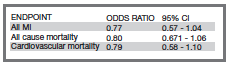
It is also important to look at just the studies in the above meta-analysis where metformin alone was compared with a placebo, diet or usual non-drug care and there were more than a few events. There are only two studies with longer than 12 months follow-up (Rachmani, 2002, UKPDS-34 1998), and only the endpoints of all cause mortality, cardiovascular mortality and all MI (heart attacks) were common to these studies. A third study lasting only 12 months was not judged relevant but was in fact negative (Cryer 2005). If the results from the two eligible studies are pooled and given equal weight (in the above meta-analysis they were given approximately equal weight), then as shown in the table below, for the three endpoints there was no statistically significant benefit found (95% confidence interval contains 1.0) These two studies together involved 537 subjects in the treated group and 609 in the control group.
Pooled analysis of two trials of metformin vs. diet (UKPDS-34 1998) or metformin withdrawal (Rachmani 2002).
The withdrawal study obtained only non-significant results indicating no benefit. The UKPDS-34 study is generally the only trial cited to justify the use of metformin because it claimed to have obtained evidence of benefit (UKPDS-34 1998). This study actually started in 1983 and reported finally in 1998. Over this period, the primary and main endpoints were changed a number of times and the announced termination date repeatedly extended. Furthermore, the study was not blinded for the investigators and the results of interim analysis were available. This study protocol is obviously open to bias. To quote one citric, “It seems that the authors continued the study until they obtained a result that was significant without adjusting for repeatedly looking at the data” (Ewart 2001). An investigator involved in the trial commented in 2008 that “UKPDS-34 broke almost all the rules of trial design. We are taught to believe that a study protocol should be predetermined and set in stone, but this study went to the other extreme, elevating the ad hoc into an art form” (Gale 2008). Thus the key study appears flawed.
In 2009 it was pointed out (Reaven 2009), based on the six-year report from UKPDS (UKPDS-16 1995), that following enrolment there was a progressive deteriation of glycemic control in all groups including those assigned to intensive control. In addition, beta cell function was estimated by HOMA-β and the investigators concluded that increasing hyperglycemia and decreasing beta cell function were significant features irrespective of the drug protocol used. Reaven also points out that the loss of secretory function does not appear to be inexorable and cites two studies where weight loss improved glycemic control. This view is supported by a study discussed below (Lim 2011).
The 2012 American Diabetes Association (ADA) Standards of Medical Care in Diabetes clearly indicates that metformin should be initiated as therapy for type 2 diabetes at the time of diagnosis. If the patient has markedly elevated blood glucose or HbA1c, then the advice is to consider insulin with or without oral agents. If metformin alone does not achieve or maintain satisfactory HbA1c levels over three to six months at maximal tolerated doses, the recommendation is to add a second oral agent (ADA 2012). This more intensive treatment is of course the subject of the several major studies discussed above which in general found no benefit. Furthermore, in the intensive vs. standard glucose lowering studies, metformin, the common reference treatment, itself does not appear to be effective. It is in fact quite remarkable that UKPDS-34 is the study used to justify metformin as the primary treatment and the drug used as a principal standard against which other drugs are tested.
Discussion and conclusions
There appears to be only very weak if any evidence regarding the ability of current drug treatments to significantly reduce the incidence of the many complications of type 2 diabetes, independent of the HbA1c level achieved. Among some diabetes researchers this is acknowledged and is termed the Diabetes Paradox, i.e. hyperglycemia is necessary for the development of late diabetic complications but average blood glucose (HbA1c) explains only a small percentage of complications (Bierhause 2011). Concerned researchers are now looking into the complex details of type 2 diabetes such as for example the potential for reactive glucose metabolites and their intermediates to act as active agents in the disease etiology and progression (Fleming 2012). There is obviously an urgent need for a new treatment paradigm rather than to merely reduce markers such as HbA1c.
In the past few years there have been calls (from the wilderness) for a return to the traditional approach used for decades for treating type 2 diabetes, the adoption of carbohydrate restriction in the context of both prevention and therapy (Accurso 2008, Leite 2009, Volek 2005a, Volek 2005b, Volek 2008, Volek 2009). However, such proposals face strong opposition from those who fear the substitution of fat for carbohydrates, even though there is compelling literature indicating that such fears are groundless (Ware 2012). The success achieved by Richard K. Bernstein using carbohydrate restriction and carefully selected carbohydrates matched to the individual’s metabolism is now documented in several editions of his book Dr. Bernstein’s Diabetes Solution (Bernstein 2011). The HbA1c levels he achieves are significantly better than anything conventional medicine appears able to accomplish, and this is generally only with diet. In fact, a recent study found complete reversal of type 2 diabetes achieved over a short period by a starvation diet, and
the benefits appear durable after return to higher calorie intake, strongly suggesting beta-cell regeneration and a permanent increase in insulin sensitivity (Lim 2011).
Are type 2 diabetes patients being given a false sense of security? They are told they need to get their blood glucose under control and many attempt to accomplish this in the belief that this reduces the risk of complications, which does not seem to be the case. The net result is a missed opportunity to restrict carbohydrates and calories and thus address the role of insulin in both insulin resistance, fat storage, and leptin resistance, deal with obesity and the metabolic syndrome, and also build muscle with resistance exercise to help increase insulin sensitivity. Another missed opportunity involves alternative medicines such as curcumin, which has produced better glucose control than prescription drugs and even appears to have reversed type 2 diabetes (Chuengsamarn 2012, Na 2012). Those who argue, probably with some justification, that severe carbohydrate restriction is not practical and adherence nearly impossible are also admitting that progress in reducing the complications of diabetes is at present nearly hopeless.
Reference List
Accurso,A., Bernstein,R.K., Dahlqvist,A., Draznin,B., Feinman,R.D., Fine,E.J., Gleed,A., Jacobs,D.B., Larson,G., Lustig,R.H., Manninen,A.H., McFarlane,S.I., Morrison,K., Nielsen,J.V., Ravnskov,U., Roth,K.S., Silvestre,R., Sowers,J.R., Sundberg,R., Volek,J.S., Westman,E.C., Wood,R.J., Wortman,J. and Vernon,M.C. Dietary carbohydrate restriction in type 2 diabetes mellitus and metabolic syndrome: time for a critical appraisal. Nutr Metab (Lond) 2008; 5: 9.
ADA. Standards of medical care in diabetes–2012. Diabetes Care 2012; 35 Suppl 1: S11-S63.
Bernstein,R.K., 2011. Dr. Bernstein’s Diabetes Solution. The complete guide to acheiving normal blood sugars. Little, Brown and Company, New York.
Bierhause,A., 2011. Failure of glucose lowering in clinical trials: The way to novel biochemical concedpts explaining diabetic late complications.The 26th Camillo Golgi Lecture, European Association for the Study of Diabetes. Annual meeting, Lisbon, September 2012.
Boussageon,R., Supper,I., Bejan-Angoulvant,T., Kellou,N., Cucherat,M., Boissel,J.P., Kassai,B., Moreau,A., Gueyffier,F. and Cornu,C. Reappraisal of metformin efficacy in the treatment of type 2 diabetes: a meta-analysis of randomised controlled trials. PLoS Med 2012; 9(4): e1001204.
Chuengsamarn,S., Rattanamongkolgul,S., Luechapudiporn,R., Phisalaphong,C. and Jirawatnotai,S.
Curcumin extract for prevention of type 2 diabetes. Diabetes Care 2012; 35(11): 2121-2127.
Coca,S.G., Ismail-Beigi,F., Haq,N., Krumholz,H.M. and Parikh,C.R. Role of intensive glucose control in development of renal end points in type 2 diabetes mellitus: systematic review and meta-analysis intensive glucose control in type 2 diabetes. Arch Intern Med 2012; 172(10): 761-769.
Cryer,D.R., Nicholas,S.P., Henry,D.H., Mills,D.J. and Stadel,B.V. Comparative outcomes study of metformin intervention versus conventional approach the COSMIC Approach Study. Diabetes Care 2005; 28(3): 539-543.
Duckworth,W., Abraira,C., Moritz,T., Reda,D., Emanuele,N., Reaven,P.D., Zieve,F.J., Marks,J., Davis,S.N., Hayward,R., Warren,S.R., Goldman,S., McCarren,M., Vitek,M.E., Henderson,W.G. and Huang,G.D. Glucose control and vascular complications in veterans with type 2 diabetes. N Engl J Med 2009; 360(2): 129-139.
Ewart,R.M. The case against aggressive treatment of type 2 diabetes: critique of the UK prospective diabetes study. BMJ 2001; 323(7317): 854-858.
Fleming,T., Cuny,J., Nawroth,G., Djuric,Z., Humpert,P.M., Zeier,M., Bierhaus,A. and Nawroth,P.P. Is diabetes an acquired disorder of reactive glucose metabolites and their intermediates? Diabetologia 2012; 55(4): 1151-1155.
Gale,E.A. Glucose control in the UKPDS: what did we learn? Diabet. Med 2008; 25 Suppl 2: 9-12.
Gerstein,H.C., Miller,M.E., Byington,R.P., Goff,D.C., Jr., Bigger,J.T., Buse,J.B., Cushman,W.C., Genuth,S., Ismail-Beigi,F., Grimm,R.H., Jr., Probstfield,J.L., Simons-Morton,D.G. and Friedewald,W.T. Effects of intensive glucose lowering in type 2 diabetes. N Engl J Med 2008; 358(24): 2545-2559.
Hemmingsen,B., Lund,S.S., Gluud,C., Vaag,A., Almdal,T., Hemmingsen,C. and Wetterslev,J. Intensive glycaemic control for patients with type 2 diabetes: systematic review with meta-analysis and trial sequential analysis of randomised clinical trials. BMJ 2011; 343: d6898.
Leite,J.O., DeOgburn,R., Ratliff,J.C., Su,R., Volek,J.S., McGrane,M.M., Dardik,A. and Fernandez,M.L. Low-carbohydrate diet disrupts the association between insulin resistance and weight gain. Metabolism 2009; 58(8): 1116-1122.
Lim,E.L., Hollingsworth,K.G., Aribisala,B.S., Chen,M.J., Mathers,J.C. and Taylor,R. Reversal of type 2 diabetes: normalisation of beta cell function in association with decreased pancreas and liver triacylglycerol. Diabetologia 2011; 54(10): 2506-2514.
Na,L.X., Li,Y., Pan,H.Z., Zhou,X.L., Sun,D.J., Meng,M., Li,X.X. and Sun,C.H. Curcuminoids exert glucose-lowering effect in type 2 diabetes by decreasing serum free fatty acids: a double-blind, placebo-controlled trial. Mol Nutr Food Res 2012.
Patel,A., MacMahon,S., Chalmers,J., Neal,B., Billot,L., Woodward,M., Marre,M., Cooper,M., Glasziou,P., Grobbee,D., Hamet,P., Harrap,S., Heller,S., Liu,L., Mancia,G., Mogensen,C.E., Pan,C., Poulter,N., Rodgers,A., Williams,B., Bompoint,S., de Galan,B.E., Joshi,R. and Travert,F. Intensive blood glucose control and vascular outcomes in patients with type 2 diabetes. N Engl J Med 2008; 358(24): 2560-2572.
Rachmani,R., Slavachevski,I., Levi,Z., Zadok,B., Kedar,Y. and Ravid,M. Metformin in patients with type 2 diabetes mellitus: reconsideration of traditional contraindications. Eur J Intern Med 2002; 13(7): 428.
Reaven,G.M. HOMA-beta in the UKPDS and ADOPT. Is the natural history of type 2 diabetes characterised by a progressive and inexorable loss of insulin secretory function? Maybe? Maybe not? Diab. Vasc Dis Res 2009; 6(2): 133-138.
UKPDS-16 U.K. prospective diabetes study 16. Overview of 6 years’ therapy of type II diabetes: a progressive disease. U.K. Prospective Diabetes Study Group. Diabetes 1995; 44(11): 1249-1258.
UKPDS-34. Effect of intensive blood-glucose control with metformin on complications in overweight patients with type 2 diabetes (UKPDS 34). UK Prospective Diabetes Study (UKPDS) Group. Lancet 1998; 352(9131): 854-865.
Volek,J.S. and Feinman,R.D. Carbohydrate restriction improves the features of Metabolic Syndrome. Metabolic Syndrome may be defined by the response to carbohydrate restriction. Nutr Metab (Lond) 2005a; 2: 31.
Volek,J.S., Fernandez,M.L., Feinman,R.D. and Phinney,S.D. Dietary carbohydrate restriction induces a unique metabolic state positively affecting atherogenic dyslipidemia, fatty acid partitioning, and metabolic syndrome. Prog. Lipid Res 2008; 47(5): 307-318.
Volek,J.S. and Forsythe,C.E. The case for not restricting saturated fat on a low carbohydrate diet. Nutr Metab (Lond) 2005b; 2: 21.
Volek,J.S., Phinney,S.D., Forsythe,C.E., Quann,E.E., Wood,R.J., Puglisi,M.J., Kraemer,W.J., Bibus,D.M., Fernandez,M.L. and Feinman,R.D. Carbohydrate restriction has a more favorable impact on the metabolic syndrome than a low fat diet. Lipids 2009; 44(4): 297-309.
Ware,W.R. Saturated fat. Friend, foe or simply neutral. Integrated Healthcare Practitioners 2012; 5(2): 68.
Chemotherapy-Related Cognitive Impairment
Chemotherapy-Related Cognitive Impairment
Strategies for intervention
Abstract
Chemotherapy-related cognitive impairment (CRCI) is characterized by memory loss, lack of concentration and attention, difficulty multitasking, organizing, planning, and thinking. CRCI can have a significant impact on quality of life but treatment options with strong supporting evidence of efficacy are scarce. This review highlights the current state of evidence regarding CRCI and therapies that may help improve cognitive function in this patient population.
Introduction
Chemobrain, also referred to as chemofog, or chemotherapy-related cognitive impairment, is a phenomenon that has been reported by patients receiving various forms of chemotherapy for the past three decades. Estimates of chemotherapy-related cognitive decline (CRCI) in breast cancer survivors are as high as 75% (Ahles 2002, Brezden 2000, Schagen 1999, Tchen 2003, Van Dam 1998). The cognitive impairment is typically characterized by memory loss, lack of concentration and attention, difficulty thinking, organizing, planning, and multi-tasking (Matsuda 2005, Reid-Arndt 2009).
While it was generally accepted by the 1990s that chemotherapeutic agents could be acutely neurotoxic when delivered directly to the nervous system, CRCI was still regarded as psychogenic (Hede 2008, Myers 2009). Given the inability of many chemotherapeutic agents to cross the blood-brain-barrier, it was believed that chemotherapy was an unlikely cause of neurotoxicity when administered systemically (O’Farrell 2013). Many confounding factors such as psychological distress associated with having cancer, fatigue, anxiety, depression, and effects of medication prescribed in conjunction with chemotherapeutic intervention, all can alter cognitive function as well.
Due to advancements in cancer treatment, there is also increased survival. As a result, there are a rising number of patients who are living with long-term side effects of these anti-cancer agents, which canhave significant negativeimpactson quality of life.
Impact on Quality of Life
In a 2010 survey conducted by the Canadian Breast Cancer Network, women who had received chemotherapy had the greatest reduction in house-hold income, took more time off work, were more likely to have had to quit their jobs, and had a greater perception that the financial burden imposed by their illness would impact their long-term health (Dunbrack 2010). In a study by Reid-Arndt et al., women with stages I-III breast cancer were evaluated one month after completing adjuvant chemotherapy. Data was collected regarding cognitive, emotional, social, and vocational functioning and an association between executive functioning deficits and decreased productivity, community involvement, and social role functioning was found (2009).
Advancements in Research
Studies on CRCI in the recent past reported poor, if any, correlation between subjective reports of cognitive decline and objective measures of cognitive performance (Biglia 2012, Hutchinson 2012, Jansen 2011). In order to establish appropriate guidelines for study design and methodology when performing research in this field, the International Cognition and Cancer Task Force (ICCTF) was created in 2006. This aided tremendously in promoting comparable findings among studies and reducing the number of confounding variables in the research (Wefel 2008).
Dr. Patricia Ganz conducted one of the first studies demonstrating that cognitive complaints after breast cancer treatment are associated with neuropsychological test performance. Her team enrolled 189 breast cancer patients one month post surgery, with or without radiation and/or chemotherapy, and prior to commencing hormone therapy—a common confounding factor. The control group consisted of a sample of age-matched healthy women and found that 23.3% of the breast cancer patients reported higher memory complaints and 19% had higher executive function complaints post-treatment; >1 SD above the mean for healthy control sample (Ganz 2013).
McDonald et al. performed the first controlled prospective MRI study in the field of neuroimaging whereby significant decreases in gray matter density were found in bilateral, frontal, temporal, and cerebellar regions as well as the right thalamus in breast cancer patients shortly following chemotherapy treatment. These changes were not evident in either a disease or healthy control group.
The cognitive changes could also not be accounted for by postsurgical effects, disease stage, psychiatric symptoms, psychotropic medication, or hormonal treatment status, suggesting the changes were due to chemotherapy treatment (2010). No other study had prospectively examined gray matter changes in patients both receiving and not receiving chemotherapy with a pre-treatment baseline with which to compare post-treatment changes. This groundbreaking study was the first to show decline in brain gray matter density shortly after chemotherapy and degree of long-term recovery via a prospective longitudinal approach.
Evidence-Based Interventions
Modafinil
Sixty-eight participants with memory problems persisting two years post-chemotherapy treatment for breast cancer were given 200mg daily of Modafinil (a psychostimulant used in the treatment of patients with narcolepsy) for four weeks, finding modest but statistically significant improvement in speed of memory and episodic memory, but not in working memory, compared with controls (Kohli 2009, Von 2011). Other groups have tried a one-time dose of 200mg, finding improvement in attention and psychomotor speed but not in working memory (Lundorff 2009).
In another study by Blackhall et al., initial doses of 100mg Modafinil for two weeks, increasing to 200mg daily in 27 patients with cancer of all stages, did not show improvement in cognitive functioning. Due to the mixed results and small sample sizes, effectiveness of this medication for the treatment of CRCI has not been established (2009).
Exercise
Korstjens evaluated the effects of a 12-week rehabilitation program combining exercise with a psychoeducational program on various aspects of quality of life, including cognition. Physical exercise sessions overseen by a physiotherapist occurred twice a week for two hours and included aqua aerobics, group sports, or individual endurance and strength training. The psychoeducational program consisted of seven two-hour sessions focused on coping with cancer. Improvements in global cognitive function were reported based on two items on the European Organization for Research and Treatment of Cancer Quality of Life Questionnaire Core-30 (2006). Although some improvements were reported in cognitive function in this and other studies (Schwartz 2002), it is difficult to determine the effect of an exercise intervention on cognitive impairment because of the difference in definition and delivery of the exercise intervention programs, small sample sizes, and study designs (combined multiple interventions). Further research is required.
Natural Environment Intervention
Attention-restoring theory identifies that the environment may influence one’s ability to concentrate and capacity to direct attention (Cimprich 2003); thus, natural restorative environmental interventions may replenish psychological reserves and improve cognitive functioning. Two studies were found that evaluated the impact of natural restorative environmental intervention on cognitive function in patients with cancer (Cimprich 1993, Cimprich 2003).
Cimprich examined the impact of a natural restorative environmental intervention (walking in nature or gardening for 20-30 minutes three times a week) in 32 patients with breast cancer and assessed their level of attention at three, 18, 60, and 90 days after surgery. Significant and sustained improvement in attentional fatigue scores were noted across all four time periods (1993). Similarly, in a follow-up randomized, controlled trial of 157 patients with breast cancer, Cimprich and Ronis demonstrated that those who were exposed to the natural environment for 120 minutes per week had improved capacity to direct attention from pretreatment as compared with the nonintervention group, even after controlling for age, education, attention scores prior to surgery, other health problems, distress, and extent of the surgery (2003). Additional longitudinal research is needed to understand its sustainability throughout the course of cancer treatment, as well as to test outcomes in other cancer populations.
Acetyl-L-Carnitine
Though not yet clinically tested for cognitive impairments due to chemotherapy, ALC improved memory, visuo-spatial capacity, and vocabulary recall when given at two grams per day over a test period of three months in cognitively impaired ex-alcoholics aged 30-60 years (Tempesta 1990).
In controlled trials, ALC improved depression as well as cooperation, sociability, and attention to personal appearance, though it did not consistently improve anxiety (Bonavita 1986). Dosage ranges from 1,500-3000mg per day with most trials using two grams or more (Kidd 1999).
Given that acetyl-L-carnitine aids in the treatment/prevention of chemotherapy-induced peripheral neuropathy (CIPN) and that CIPN can be caused by chemotherapeutic agents such as Paclitaxel and Cisplatin— known to also cause chemotherapy-related cognitive impairment—the use of ALC may help to target more than one of these side effects which may impact quality of life significantly (Pisano 2003).
Phosphatidylserine (PS)
PS is present in all cells of the human body and particularly in the membrane systems of nerve cells. While also not clinical studied for CRCI, phosphatidylserine consistently benefits memory, learning, concentration, word choice, mood, and relieves anxiety and depression, and the capacity to cope with stress (Crook 1992, Kidd 1998, Maggioni 1990). Effective intakes of PS range from 100mg per day (for smaller children and for maintenance in healthy adults) to 300mg/day for memory loss and up to 600mg/day for mood enhancement (Kidd 1998).
Conclusion
For the past several decades research on CRCI has focused on proving whether this side effect is psychogenic or neurological in nature. With studies now using advanced neuroimaging techniques, new research may attempt to elucidate mechanisms by which various chemotherapeutic agents may be causing cognitive decline in patients with other forms of cancer, not limited to breast cancer. Insight into mechanisms of action will in turn allow for more research regarding treatment and prevention of CRCI. Sample sizes of the majority of the present studies are too small to make appropriate inferences about any conclusions made. Patient populations in these studies are typically females with breast cancer and not representative of all other types of cancer.
References
Ahles TA, Saykin AJ, Furstenberg CT, et al. Neuropsychologic impact of standard dose chemotherapy in long-term survivors of breast cancer and lymphoma. J Clin Oncol. 2002;20:485-493.
Biglia N, Bounous VE, Malabaila A, et al. Objective and self-reported cognitive dysfunction in breast cancer women treated with chemotherapy: a prospective study. Eur J Cancer Care. 2012;21:485-92.
Blackhall, L., Petroni, G., Shu, J., Baum, L., & Farace, E. A pilot study evaluating the safety and efficacy of modafinil for cancer-related fatigue. Journal of Palliative Medicine. 2009;12:433-439.
Bonavita E. Study of the efficacy of tolerability of L-acetyl carnitine therapy in the senile brain. Intn J Clin Pharmacol Ther Toxicol. 1986;24:511-516.
Brezden CB, Phillips KA, Abdolell M, et al. Cognitive function in breast cancer patients receiving adjuvant chemotherapy. J Clin Oncology. 2000;(18):2695-2701.
Cimprich B. Development of an intervention to restore attention in cancer patients. Cancer Nursing. 1993;16:83-92.
Cimprich, B., & Ronis, D.L. An environmental intervention to restore attention in women with newly diagnosed breast cancer. Cancer Nursing. 2003;26:284-292.
Crook T, Petrie W, Wells C, Massari DC. Effects of phosphatidylserine in Alzheimer’s disease. Psychopharmacol Bull. 1992;28:61-66.
Dunbrack J. Breast cancer: Economic impact and labour force re-entry. Ottawa: Canadian Breast Cancer Network, 2010. Web. 27 Jul 2013 <http://www.cbcn.ca/documents/ Labour_Force_Re-Entry_Report_ENG_CBCN_2010.pdf> .
Ganz PA, Kwan L, Castellon SA, Oppenheim A, Bower JE, Silverman DH, Cole SW, Irwin MR, Ancoli-Israel S, Belin TR. Cognitive complaints after breast cancer treatments: examining the relationship with neuropsychological test performance. J Natl Cancer Inst. 2013 Jun 5;105(11):791-801.
Hede, K. Chemobrain is real but may need new name. J Natl Cancer Inst. 2008;100(3):162-169.
Hutchinson AD, Hosking JR, Kichenadasse G, et al. Objective and subjective cognitive impairment following chemotherapy for cancer: a systematic review. Cancer Treat Rev. 2012;38:926-34.
Jansen CE, Cooper BA, Dodd MJ et al. A prospective longitudinal study of chemotherapy-induced cognitive changes in breast cancer patients. Support Care Cancer. 2011;19:1647-1656.
Kidd PM. A review of nutrients and botanicals in the integrative management of cognitive dysfunction. Altern Med Rev. 1999 Jun;4(3):144-161.
Kidd PM. Phosphatidylserine. Number one Brain Booster. New Canaan, CT.: Keats Publishing; 1998.
Kohli S, Fisher SG, Tra Y, Adams MJ, Mapstone ME, Wesnes KA, Roscoe JA, Morrow GR. The effect of modafinil on cognitive function in breast cancer survivors. Cancer. 2009 Jun 15;115(12):2605-16.
Korstjens I, Mesters I, van der Peet E, Gijsen B, van den Borne B. Quality of life of cancer survivors after physical and psychosocial rehabilitation. European Journal of Cancer Prevention. 2006;15:541-547.
Lundorff LE, Jonsson BH, Sjogren P. Modafinil for attentional and psychomotor dysfunction in advanced cancer: A double-blind randomised, cross-over trial. Palliative Medicine. 2009;23:731-738.
Maggioni M, Picotti GB, Bondiolotti GP, Panerai A, Cenacchi T, Nobile P, Brambilla F. Effects of phosphatidylserine therapy in geriatric patients with depressive disorders. Acta Psychiatr Scand. 1990 Mar; 81(3):265-270.
Matsuda T, Takayama, T, Tashiro M. et al. Mild cognitive impairment after adjuvant chemotherapy in breast cancer patients –evaluation of appropriate research design and methodology to measure symptoms. Breast Cancer. 2005;12:279-287.
McDonald BC, Conroy SK, Ahles TA, West JD, Saykin AJ. Gray matter reduction associated with systemic chemotherapy for breast cancer: a prospective MRI study. Breast Cancer Res Treat. 2010 Oct;123(3):819-828.
Myers JS. Chemotherapy-related cognitive impairment. Clin J Oncol Nurs. 2009;13(4):413-421.
O’Farrell, E, Mackenzie J, Collins B. Clearing the Air: A Review of Our Current Understanding of “Chemo Fog.” Current Oncology Reports. 2013 Jun;15(3):260-9.
Pisano C, Pratesi G, Laccabue D, et al. Paclitaxel and Cisplatin-induced neurotoxicity: a protective role of acetyl- L-carnitine. Clin Cancer Res 2003;9(15)5756-67.
Reid-Arndt, SA. Breast cancer and “chemobrain”: the consequences of cognitive difficulties following chemotherapy and the potential for recovery. Mo Med. 2009;106(2):127-131.
Schagen SB, van Dam FS, Muller MJ, et al. Cognitive deficits after postoperative adjuvant chemotherapy for breast carcinoma. Cancer. 1999;85:640-650.
Schwartz AL, Thompson JA, Masood N. Interferon-induced fatigue in patients with melanoma: A pilot study of exercise and methylphenidate [Online exclusive]. Oncology Nursing Forum. 2002;29:E85-E90.
Tchen N, Juffs JG, Downie FP, et al. Cognitive function, fatigue, and menopausal symptoms in women receiving adjuvant chemotherapy for breast cancer. J Clin Oncology. 2003;21:4175-4183.
Tempesta E, Troncon R, Janiri L, et al. Role of acetyl-L-carnitine in the treatment of cognitive deficit in chronic alcoholism. Int J Clin Pharmacol Res. 1990;10:101-107.
Van Dam FSAM, Schagen SB, Muller MJ, et al. Impairment of cognitive function in women receiving adjuvant treatment for high-risk breast cancer: high-dose versus standard-dose chemotherapy. J Natl Cancer Inst. 1998;90:210-218.
Von Ah D, Jansen C, Allen DH, Schiavone RM, Wulff J. Putting evidence into practice: evidence-based interventions for cancer and cancer treatment-related cognitive impairment. Clinical Journal of Oncology Nursing. 2011 Dec; 15(6):607-15.
Wefel JS, Vardy J, Ahles T, et al. Cancer and cancer-related cognitive dysfunction: an international perspective from the Venice cognitive workshop. Ann Oncol. 2008;19:623-9.
The metabolic fate of alpha linolenic acid (ALA)
The metabolic fate of alpha linolenic acid (ALA)
Extremely limited conversion efficiency
ABSTRACT:
EPA and DHA possess important physiological and biological properties in human health and development; however, it is alpha-linolenic acid (ALA) that is classified as the essential n-3 PUFA. While humans have the required enzymes to biosynthesize omega-3 long-chain polyunsaturated fatty acids (PUFA), studies demonstrate that the majority of ALA is β-oxidized and only ~5% of ALA is converted to eicosapentaenoic acid (EPA) and <0.5% of ALA is converted to docosahexaenoic acid (DHA). Even very high intakes of dietary ALA fail to effectively modulate plasma and tissue levels of DHA. Furthermore, the abundance of linoleic acid (LA) in our diet, as well as age, gender and genetics influence the conversion efficiency of ALA. Therefore, direct dietary consumption or supplementation of EPA and DHA from fish, fish oil, or algae is the only clinically effective way to increase blood and tissue levels of these longchain PUFA in humans for optimal health and disease prevention.
INTRODUCTION[spacer height=”20px”]
Considerable clinical interest has focused on the health benefits of omega-3 polyunsaturated fatty acids (ω-3 PUFA). While consumption of marine derived eicosapentaenoic acid (EPA; 20:5ω-3) and docosahexaenoic acid (DHA; 22:6ω-3) have been evidenced to prevent cardiovascular and neurological disease risk, the specific function of plant derived alpha-linolenic acid (ALA; 18:3ω-3) remains a matter of debate. EPA and DHA play a vital role in cellular membranes, maintaining fluidity, protein and cellular functions, as well as influencing eicosanoid metabolism, gene expression and cell signaling (Adkins 2010). However, the specific bioactive role of ALA is unclear (Sinclair 2002). It has been suggested that the major function of ALA is to serve as a precursor for EPA and DHA, a pathway that has received much attention and clinical investigation.
In 1929, Burr and Burr first identified the nutritional essentiality of LA, and later ALA (Burr 1929, Burr 1930). ALA is the parent ω-3 PUFA containing three double bonds with the first double bond located at the third carbon relative to the methyl end of the 18-carbon chain. ALA and ω-6 linoleic acid (LA, 18:2ω-6) are termed essential fatty acids because humans lack the delta (Δ)15- and Δ12-desaturase enzymes required for insertion of a double bond at the ω-3 or ω-6 position, respectively. Since EPA and DHA can be synthesized from ALA via a series of alternating desaturation and elongation steps, these long-chain PUFA are not considered essential. However, whether endogenous synthesis of EPA and DHA from ALA is adequate to support growth, physiological needs, and disease risk reduction is questioned (Harris 2009, Saldanha 2009). Considering negligible plasma and tissue levels of ALA, yet its classification of essential, this review will examine the biosynthesis of EPA and DHA from ALA, factors influencing ALA metabolism, and ω-3 PUFA in the current diet.
BIOSYNTHESIS OF LONG-CHAIN POLYUNSATURATED FATTY ACIDS
The predominate site of ALA desaturation and elongation occurs in the liver, however, also occurs to a lesser extent in other tissues, including the brain and heart (Cho 1999 A, Cho 1999 B). Dietary LA and ALA compete for the same desaturase and elongase enzymes for long-chain PUFA biosynthesis with the majority of this pathway occurring in the endoplasmic reticulum. Of interest, the desaturase enzymes have a higher affinity for ALA versus LA (Plourde 2007), however, high levels of dietary LA saturate Δ6-desaturation inhibiting the accumulation of ω-3 long-chain PUFA, namely EPA (Angela Liou 2009, Liou 2007).
The first reaction in the conversion pathway is the desaturation of ALA to stearidonic acid (SDA; 18:4ω-3) or LA to gamma-linolenic acid (GLA; 18:3ω-6) via the rate-limiting enzyme Δ6-desaturase (Sprecher 2000) (Figure 1). Next, elongation and Δ5-desaturation converts SDA to EPA and GLA to arachidonic acid (AA; 20:4ω-6), the major bioactive n-6 PUFA in tissue membranes and precursor for proinflammatory eicosanoids (Adkins 2010). Compared to AA, EPA is quantitatively a minor fatty acid in tissue membranes and undergoes further elongation to docosapentaenoic acid (DPA; 22:5ω- 3), or is metabolized by cyclooxygenase (COX) and lipoxygenase (LOX) enzymes in the synthesis of anti-inflammatory eicosanoids. In humans, DPA undergoes another elongation and Δ6-desaturation step, and then partial β-oxidation in the peroxisome to form DHA (Sprecher 2000). Structurally, DHA is the predominant ω-3 PUFA esterified into tissue membrane phospholipids. DHA can also be metabolized to produce resolvins and protectins in the resolution of inflammation (Adkins 2010). It has been hypothesized that multiple use of the rate-limiting Δ6-desaturase enzyme for the conversion of ALA to SDA and 24:5n-3 to 24:6n-3 may lead to a “bottle-neck” in the metabolic pathway and an associated decrease in the synthesis of DHA (D’andrea 2002, Kitson 2010). Another possible rate-limiting step may be related to the compartmental translocation of 24:6n-3 from the endoplasmic reticulum to the peroxisome. Both hypotheses demand further investigation.
RESULTS FROM HUMAN STUDIES:
The conversion of ALA to long-chain ω-3 PUFA is very inefficient. Dietary supplementation and isotope tracer studies in humans demonstrate a direct linear relationship between dietary ALA and plasma and tissue concentrations of EPA, at approximately 5% conversion efficiency (Plourde 2007). However, biosynthesis of DHA is negligible with typically less than 0.5% conversion of ALA to DHA. Human studies supplementing ALA ranging from 1.5 to 40 g/day for a minimum of three weeks report a relative increase in phospholipid concentration of EPA ranging from trace levels to 250% (Plourde 2007). Studies have also observed an increase in plasma and tissue concentrations of DPA, although to a lesser extent than EPA concentrations. However, the majority of dietary ALA intervention studies fail to effectively modulate plasma and tissue levels of DHA. Gillingham et al demonstrated that consuming 20 g/day of ALA from a flaxseed oil supplemented diet (~2.5 tbsp flaxseed oil daily) resulted in a relative 222% increase in plasma EPA concentration (1.74% total fatty acids) compared with control (0.54% total fatty acids) (2011). However, DHA concentration did not change after supplementing with flaxseed oil. Similarly, supplementing lactating women with 20 g/day of flaxseed oil (10.7 g/day ALA) for four weeks increased breast milk EPA and DPA, but not DHA concentrations (Francois 2003).
Additionally, very high levels of dietary ALA, as well as LA, may saturate the Δ6-desaturase enzyme further inhibiting the final steps of Δ6-desaturation of 24:5n-3 to DHA (Gibson 2011). Even with vegetarian and vegan diets, ALA conversion to DHA is not up-regulated (Fokkema 2000, Li 1999). James et al revealed that compared to dietary ALA, supplementing with SDA-enriched vegetable oil (therefore, bypassing the first Δ6-desaturase rate-limiting step) more effectively increased plasma and tissue concentrations of EPA, however, did not raise DHA concentrations (2003). In addition, EPA supplementation does not significantly elevate DHA concentrations (Brenna 2009). Therefore, the only effective way to increase tissue DHA concentrations is through its direct consumption.
FACTORS AFFECTING EPA AND DHA BIOSYNTHESIS FROM ALA
DIETARY Ω-6 LINOLEIC ACID CONTENT: The abundance of LA and resulting high ω-6 to ω-3 PUFA ratio in current Western diets significantly impedes the conversion efficiency of ALA. Liou and collegues demonstrated that while maintaining dietary ALA at 1% energy, increasing levels of dietary LA from 4% to 10% energy, thus varying the ω-6 to ω-3 PUFA ratio from 4:1 to 10:1, resulted in a decrease in plasma EPA concentrations in healthy men (Angela Liou 2009, Liou 2007). In another human dietary intervention study, lowering the ω-6 to ω-3 PUFA ratio resulted in a significant decrease in platelet aggregation (Freese 1994). Furthermore, trans fats rich in hydrogenated vegetable oils inhibit the synthesis of ω-3 long-chain PUFA (Kummerow 2004).
MICRONUTRIENTS: Vitamins B3, B6, and C, magnesium and zinc are important cofactors to the Δ5- and Δ6-desaturase enzymes. Therefore, low intakes of these essential micronutrients may lead to a reduction in the synthesis of ω-3 long-chain PUFA (Cunnane 1988, de Lorgeril 2005, Harris 2009).
GENDER: Gender may affect biosynthesis of long-chain PUFA, as DHA concentration of plasma phospholipids have been shown to be higher in women than in men (Decsi 2011). Giltay et al observed a 15% increase in DHA status in women compared with men (2004). Furthermore, administration of oral estradiol increased DHA status by 42%, while testosterone decreased DHA status by 22%. It is proposed that estrogen may upregulate ALA metabolism to DHA, and thus, increase maternal DHA status particularly during pregnancy due to the greater demand of DHA for fetal neurological development (Otto 2001, Innis 2000).
AGE: Studies suggest that infants exhibit increased conversion of ALA to long-chain PUFA, including DHA (Clark 1992, Jensen 1996). A study tracing ALA metabolism in preterm infants fed long-chain PUFA enriched formula reported that in one-month old infants about 42% of ALA was converted to DHA, whereas only 11% was converted at three-months of age, and 7% at seven months of age (Carnielli 2007). Typically, conversion of ALA to DHA is still limited in healthy infants at ~1% (Brenna 2009). However, differences in adult age (18–29 versus 45–69 years) have not been shown to influence metabolism of ALA to EPA or DHA (Patenaude 2009).
GENETICS: Fatty acid desaturase genes FADS1 and FADS2 encode for Δ5-desaturase and Δ6-desaturase, respectively (Cho 1999, Cho 1999). Schaeffer and colleagues published the first study reporting that single nucleotide polymorhpisms (SNP) of the FADS1/FADS2 gene cluster modulate ALA and LA metabolism leading to differences in phospholipid PUFA concentrations (2006). More specifically, minor allele carriers for SNPs located in the FADS1/ FADS2 gene cluster have reduced ability to convert LA to AA (Tanaka 2009) or ALA to EPA (Gillingham 2013a). Of interest, genetic variation explains ~40% or more of the interindividual variability in all fatty acid concentrations (Lemaitre 2008). Martinelli et al reported that individuals carrying FADS polymorphisms, associated with higher conversion of LA to AA, have increased proinflammatory CRP concentrations and risk for coronary artery disease (2008).
OTHER METABOLIC PATHWAYS
β-oxidation accounts for the major metabolic fate of dietary ALA (McCloy 2004). McCloy et al. demonstrated that ~71% of dietary ALA was oxidized over a seven day period, the highest oxidation rate of all fatty acids (2004). During mitochondrial β-oxidation of ALA, carbon units generated in the form of acetyl-CoA can be recycled to synthesize SFA, MUFA, cholesterol and ketone bodies (Burdge 2003). Second to β-oxidation, storage of ALA in adipose tissues accounts for a main metabolic route of dietary ALA (McCloy 2004), however, ALA is readily mobilized from fatty acid tissue during increased energy demands of the body.
DIETARY OMEGA-3 POLYUNSATURATED FAT INTAKE AND RECOMMENDATIONS
Since ALA and LA are the only fatty acids classified as essential, the US Institute of Medicine’s (IOM) Food and Nutrition Board, together with Health Canada, has established an Adequate Intake (AI, an intake level necessary to achieve nutritional adequacy and prevent deficiency symptoms) for ALA as 1.1-1.6 g/day and for LA as 12-17 g/day for adults (Institute of Medicine 2002). Symptoms of ALA and LA deficiency include scaly dry skin, reproductive failure, numbness, weakness, pain, and blurred vision (Collins 1971, Holman 1982). However, current Western intake of ALA at 1.4-1.8 g/day and LA at 13-18 g/day by adults (Rhodes 2012) exceed the outlined AI by the IOM. The clinical concern is that current intake of combined EPA and DHA at 90–120 mg/day fail to meet recommendations outlined by professional health organization ranging from 500–4000 mg/ day (Table 1). Due to the importance of DHA in fetal retina and brain growth and development, the minimum recommended intake of DHA during pregnancy and lactation is 200 mg/day (Koletzko 2007). However, pregnant and lactating women in Canada and the United States only consume approximately 80 and 50 mg/day, respectively (Denomme 2005, Rhodes 2012). In addition, biosynthesis of long-chain PUFA, namely DHA from dietary ALA is insufficient to meet target recommended intakes of EPA and DHA for optimal health and disease prevention.
The FAO/WHO joint committee recommends an ω-6 to ω-3 ratio between 5:1 and 10:1 (WHO/FAO 1995). Of interest, the Paleolithic diet contained an ω-6 to ω-3 ratio of <1:1 (Kuipers 2010), while current intakes range from 10-25:1 in the North America diet.
SAFETY OF EPA AND DHA SUPPLEMENTATION
In 1997, the US Food and Drug Administration (FDA) granted Generally Recognized As Safe (GRAS) status to refined fish oils and indicated that the consumption of up to 3g/day of EPA+DHA is considered safe for the adults population, including patients with diabetes, bleeding tendencies, and elevated LDL-cholesterol (1997). Intake exceeding 3g/day should only be recommended under the guidance of a healthcare practitioner.
DIETARY SOURCES:
ω-6 LA is abundant in our diet, rich in corn, safflower, soybean, and sunflower oils. However, ω-3 ALA is found in only a limited amount of foods, namely flaxseed, walnuts, soybean and their oils, as well as canola oil, butternuts, and chia seeds. Although flaxseed oil represents the richest source of ALA (7.3 g ALA/tbsp), it is not commonly consumed compared with soybean oil (0.9 g ALA/tbsp) and canola oil (1.3 g ALA/ tbsp) (USDA 2011). Purslane, a wild leafy vegetable common in the Eastern Mediterranean diet, contains 300–400mg ALA/100g serving (Simopoulos 1996).
EPA and DHA are primarily found in fish, fish oil, krill oil or algae. However, the concentration of EPA and DHA substantially varies in fish species (Table 2). For example, fatty fish such as farmed Atlantic salmon provides 1.83 g EPA+DHA per 3 oz serving, whereas the same amount of lean fish such as cod provides only 0.14 g EPA+DHA (USDA 2011). Therefore, one would have to consume approximately four servings of farmed Atlantic salmon or 52 servings of cod per week to achieve a recommended intake of 1g/day (Table 2). Where fatty fish intake exceeds two servings per day, such as Greenland and Japan (Stark 2002, Iso H 1989), plasma EPA/DHA levels are in what is widely considered a therapeutic range (Harris 2010). However, in most parts of the world, theselevels are not being achieved. In addition, depending on the species of fish, excessive fish intake may lead to concerns surrounding mercury exposure, particularly in pregnancy (Koletzko 2007). Taken together, fish oil supplements may provide a more feasible option to target clinical recommendations for EPA+DHA. Minor amounts of EPA can also be found in seaweed, such as kelp, laver, and wakame ranging in EPA levels from 0.004 to 0.186 g/100g serving (USDA 2011). Additionally, microalgae-based supplements offer an alternative for DHA intakes for vegetarians/vegans.
CONCLUSION
The consensus of human clinical trials substantiate that the biosynthesis of EPA and DHA from dietary ALA is extremely limited and insufficient to meet protective tissue levels for disease prevention. In addition, factors including diet, gender, age and genetics affect individual’s capacity for biosynthesis and resulting EPA and DHA tissue concentration. Considering the majority of dietary ALA is β-oxidized, negligible conversion rates, and low intakes of EPA and DHA in the current diet, professional organizations emphasize direct supplementation of EPA and DHA in the diet for optimal health and disease risk reduction.
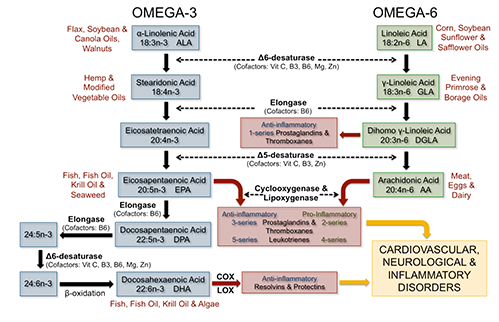
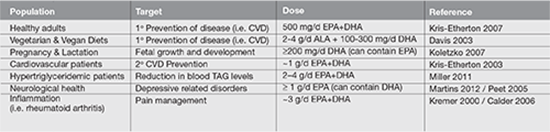
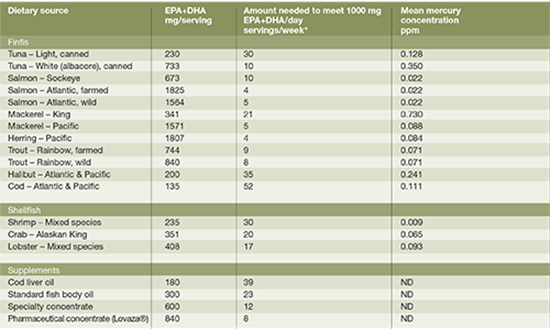
week to provide 1000 mg of EPA+DHA per day.
REFERENCES
Adkins Y, Kelley DS. Mechanisms underlying the cardioprotective effects of omega-3 polyunsaturated fatty acids. J Nutr Biochem. 2010 Sep;21(9):781-92.
Angela Liou Y, Innis SM. Dietary linoleic acid has no effect on arachidonic acid, but increases n-6 eicosadienoic acid, and lowers dihomo-gamma-linolenic and eicosapentaenoic acid in plasma of adult men. Prostaglandins Leukot Essent Fatty Acids. 2009 Apr;80(4):201-6.
Brenna JT, Salem N,Jr, Sinclair AJ, Cunnane SC, International Society for the Study of Fatty Acids and Lipids, ISSFAL. alpha-Linolenic acid supplementation and conversion to n-3 long-chain polyunsaturated fatty acids in humans. Prostaglandins Leukot Essent Fatty Acids. 2009 Feb-Mar;80(2-3):85-91.
Burdge GC, Wootton SA. Conversion of alpha-linolenic acid to palmitic, palmitoleic, stearic and oleic acids in men and women. Prostaglandins Leukot Essent Fatty Acids. 2003 Oct;69(4):283-90.
Burr GO, Burr MM. A new deficiency disease produced by the rigid exclusion of fat from the diet. J Biol Chem. 1929(2);82:345-67.
Burr GO, Burr MM. On the nature and role of the fatty acids esential in nutrition. J Biol Chem. 1930(2);86:587-621.
Calder PC. n-3 polyunsaturated fatty acids, inflammation, and inflammatory diseases. Am J Clin Nutr. 2006 Jun;83(6 Suppl):1505S-1519S.
Carnielli VP, Simonato M, Verlato G, Luijendijk I, De Curtis M, Sauer PJ, Cogo PE. Synthesis of long-chain polyunsaturated fatty acids in preterm newborns fed formula with long-chain polyunsaturated fatty acids. Am J Clin Nutr. 2007 Nov;86(5):1323-30.
Clark KJ, Makrides M, Neumann MA, Gibson RA. Determination of the optimal ratio of linoleic acid to alpha-linolenic acid in infant formulas. J Pediatr. 1992 Apr;120(4 Pt 2):S151-8.
Cho HP, Nakamura M, Clarke SD. Cloning, expression, and fatty acid regulation of the human delta-5 desaturase. J Biol Chem. 1999A Dec;274(52):37335-9.
Cho HP, Nakamura MT, Clarke SD. Cloning, expression, and nutritional regulation of the mammalian delta-6 desaturase. J Biol Chem. 1999B Jan;274(1):471-7.
Collins FD, Sinclair AJ, Royle JP, Coats DA, Maynard AT, Leonard RF. Plasma lipids in human linoleic acid deficiency. Nutr Metab. 1971;13(3):150-67.
Cunnane SC. Role of zinc in lipid and fatty acid metabolism and in membranes. Prog Food Nutr Sci. 1988;12(2):151- 88.
D’andrea S, Guillou H, Jan S, Catheline D, Thibault JN, Bouriel M, Rioux V, Legrand P. The same rat delta 6-desaturase not only acts on 18- but also on 24-carbon fatty acids in very-long-chain polyunsaturated fatty acid biosynthesis. Biochem J. 2002 May;364(Pt 1):49-55.
Davis BC, Kris-Etherton PM. Achieving optimal essential fatty acid status in vegetarians: current knowledge and practical implications. Am J Clin Nutr. 2003 Sep;78(3 Suppl):640S-646S.
de Lorgeril M, Salen PDefaye P. Importance of nutrition in chronic heart failure patients. Eur Heart J. 2005 Nov;26(21):2215-7.
Decsi T, Kennedy K. Sex-specific differences in essential fatty acid metabolism. Am J Clin Nutr. 2011 Dec;94(6 Suppl):1914S-1919S.
Food and Drug Administration. Mercury levels in commercial fish and shellfish (1990-2010). Washington, DC, 2011. Internet: http://www.fda.gov/Food/FoodSafety/Product-SpecificInformation/Seafood/Foodborne Pathogens Contaminants/Methylmercury/ucm115644.htm.
Fokkema MR, Brouwer DA, Hasperhoven MB, Martini IA, Muskiet FA. Short-term supplementation of low-dose gamma-linolenic acid (GLA), alpha-linolenic acid (ALA), or GLA plus ALA does not augment LCP omega 3 status of Dutch vegans to an appreciable extent. Prostaglandins Leukot Essent Fatty Acids. 2000 Nov;63(5):287-92.
Francois CA, Connor SL, Bolewicz LC, Connor WE. Supplementing lactating women with flaxseed oil does not increase docosahexaenoic acid in their milk. Am J Clin Nutr. 2003 Jan;77(1):226-33.
Freese R, Mutanen M, Valsta LM, Salminen I. Comparison of the effects of two diets rich in monounsaturated fatty acids differing in their linoleic/alpha-linolenic acid ratio on platelet aggregation. Thromb Haemost. 1994 Jan;71(1):73-7.
Gibson RA, Muhlhausler B, Makrides M. Conversion of linoleic acid and alpha-linolenic acid to long-chain polyunsaturated fatty acids (LCPUFAs), with a focus on pregnancy, lactation and the first 2 years of life. Matern Child Nutr. 2011 Apr;7 Suppl 2:17-26.
Gillingham LG, Gustafson JA, Han SY, Jassal DS, Jones PJ. High-oleic rapeseed (canola) and flaxseed oils modulate serum lipids and inflammatory biomarkers in hypercholesterolaemic subjects. Br J Nutr. 2011 Feb;105(3):417-27.
Gillingham LG, Harding SV, Rideout TC, Yurkova N, Cunnane SC, Eck PK, Jones PJ. Dietary oils and FADS1-FADS2 genetic variants modulate [13C]α-linolenic acid metabolism and plasma fatty acid composition. Am J Clin Nutr. 2013 Jan;97(1):195-207.(a)
Gillingham LG, Jones PJH. Chapter 4. The Evolution of Omega-3 Fatty Acids in the Human Diet, in The Omega-3 Fatty Acid Deficiency Syndrome: Opportunities for Disease Prevention. Editor: Robert K. McNamara; Nova Science Publishers, Hauppauge, NY, 2013.(b)
Giltay EJ, Gooren LJ, Toorians AW, Katan MB, Zock PL. Docosahexaenoic acid concentrations are higher in women than in men because of estrogenic effects. Am J Clin Nutr. 2004 Nov;80(5):1167-74.
Harris WS, Mozaffarian D, Lefevre M, Toner CD, Colombo J, Cunnane SC, Holden JM, Klurfeld DM, Morris MC, Whelan J. Towards establishing dietary reference intakes for eicosapentaenoic and docosahexaenoic acids. J Nutr. 2009 Apr;139(4):804S-19S.
Harris WS. The omega-3 index: clinical utility for therapeutic intervention. Curr Cardiol Rep. 2010 Nov;12(6):503-8.
Holman RT, Johnson SB, Hatch TF. A case of human linolenic acid deficiency involving neurological abnormalities. Am J Clin Nutr. 1982 Mar;35(3):617-23.
Innis SM. The role of dietary n-6 and n-3 fatty acids in the developing brain. Dev Neurosci. 2000 Sep-Dec;22(5-6):474-80.
Institute of Medicine. Dietary reference intakes: Energy, carbohydrates, fiber, fat, fatty acids, cholesterol, protein, and amino acids. Washington, DC: National Academies Press, 2002.
Iso H, Sato S, Folsom AR, Shimamoto T, Terao A, Munger RG, Kitamura A, Konishi M, Iida M, Komachi Y. Serum fatty acids and fish intake in rural Japanese, urban Japanese, Japanese American and Caucasian American men. Int J Epidemiol. 1989 Jun;18(2):374-81.
James MJ, Ursin VM, Cleland LG. Metabolism of stearidonic acid in human subjects: comparison with the metabolism of other n-3 fatty acids. Am J Clin Nutr. 2003 May;77(5):1140-5.
Jensen CL, Chen H, Fraley JK, Anderson RE, Heird WC. Biochemical effects of dietary linoleic/alpha-linolenic acid ratio in term infants. Lipids. 1996 Jan;31(1):107-13.
Kitson AP, Stroud CK, Stark KD. Elevated production of docosahexaenoic acid in females: potential molecular mechanisms. Lipids. 2010 Mar;45(3):209-24.
Koletzko B, Cetin I, Brenna JT. Dietary fat intakes for pregnant and lactating women. Br J Nutr. 2007 Nov;98(5):873-7.
Kremer JM. n-3 fatty acid supplements in rheumatoid arthritis. Am J Clin Nutr. 2000 Jan;71(1 Suppl):349S-51S.
Kris-Etherton PM, Harris WS, Appel LJ; AHA Nutrition Committee. American Heart Association. Omega-3 fatty acids and cardiovascular disease: new recommendations from the American Heart Association. Arterioscler Thromb Vasc Biol. 2003 Feb;23(2):151-2.
Kris-Etherton PM, Innis S, Ammerican Dietetic Assocition, Dietitians of Canada. Position of the American Dietetic
Association and Dietitians of Canada: dietary fatty acids. J Am Diet Assoc. 2007 Sep;107(9):1599-611.
Kuipers RS, Luxwolda MF, Dijck-Brouwer DA, Eaton SB, Crawford MA, Cordain L, Muskiet FA. Estimated macronutrient and fatty acid intakes from an East African Paleolithic diet. Br J Nutr. 2010 Dec;104(11):1666-87.
Kummerow FA, Zhou Q, Mahfouz MM, Smiricky MR, Grieshop CM, Schaeffer DJ. Trans fatty acids in hydrogenated fat inhibited the synthesis of the polyunsaturated fatty acids in the phospholipid of arterial cells. Life Sci. 2004 Apr;74(22):2707-23.
Lemaitre RN, Siscovick DS, Berry EM, Kark JD, Friedlander Y. Familial aggregation of red blood cell membrane fatty acid composition: the Kibbutzim Family Study. Metabolism. 2008 May;57(5):662-8.
Li D, Sinclair A, Wilson A, Nakkote S, Kelly F, Abedin L, Mann N, Turner A. Effect of dietary alpha-linolenic acid on thrombotic risk factors in vegetarian men. Am J Clin Nutr. 1999 May;69(5):872-82.
Liou YA, King DJ, Zibrik D, Innis SM. Decreasing linoleic acid with constant alpha-linolenic acid in dietary fats increases (n-3) eicosapentaenoic acid in plasma phospholipids in healthy men. J Nutr. 2007 Apr;137(4):945-52.
Martinelli N, Girelli D, Malerba G, Guarini P, Illig T, Trabetti E, Sandri M, Friso S, Pizzolo F, Schaeffer L, et al. FADS genotypes and desaturase activity estimated by the ratio of arachidonic acid to linoleic acid are associated with inflammation and coronary artery disease. Am J Clin Nutr. 2008 Oct;88(4):941-9.
Martins JG, Bentsen H, Puri BK. Eicosapentaenoic acid appears to be the key omega-3 fatty acid component associated with efficacy in major depressive disorder: a critique of Bloch and Hannestad and updated meta-analysis. Mol Psychiatry. 2012 Dec;17(12):1144-9; discussion 1163-7.
McCloy U, Ryan MA, Pencharz PB, Ross RJ, Cunnane SC. A comparison of the metabolism of eighteen-carbon 13C-unsaturated fatty acids in healthy women. J Lipid Res. 2004 Mar;45(3):474-85.
Miller M, Stone NJ, Ballantyne C, Bittner V, Criqui MH, Ginsberg HN, Goldberg AC, Howard WJ, Jacobson MS, Kris- Etherton PM, et al. Triglycerides and cardiovascular disease: a scientific statement from the American Heart Association. Circulation. 2011 May 24;123(20):2292-333.
Otto SJ, van Houwelingen AC, Badart-Smook A, Hornstra G. Changes in the maternal essential fatty acid profile during early pregnancy and the relation of the profile to diet. Am J Clin Nutr. 2001 Feb;73(2):302-7.
Patenaude A, Rodriguez-Leyva D, Edel AL, Dibrov E, Dupasquier CM, Austria JA, Richard MN, Chahine MN, Malcolmson LJ, Pierce GN. Bioavailability of alpha-linolenic acid from flaxseed diets as a function of the age of the subject. Eur J Clin Nutr. 2009 Sep;63(9):1123-9.
Peet M, Stokes C. Omega-3 fatty acids in the treatment of psychiatric disorders. Drugs. 2005;65(8):1051-9.
Plourde M, Cunnane SC. Extremely limited synthesis of long chain polyunsaturates in adults: implications for their dietary essentiality and use as supplements. Appl Physiol Nutr Metab. 2007 Aug;32(4):619-34.
Simopoulos AP, Salem N,Jr. Purslane: a terrestrial source of omega-3 fatty acids. N Engl J Med. 1986 Sep;315(13):83.
Stark KD, Mulvad G, Pedersen HS, Park EJ, Dewailly E, Holub BJ. Fatty acid compositions of serum phospholipids of postmenopausal women: a comparison between Greenland Inuit and Canadians before and after supplementation with fish oil. Nutr. 2002 Jul-Aug;18(7-8):627-30.
Rhodes, D.G., Clemens, J.C., Goldman, J.D., LaComb, R.P., Moshfegh, A.J. 2012. 2009-2010 What We Eat In America, NHANES Tables 1-36. Worldwide Web Site: Food Surveys Research Group. Retrieved from http://www.ars.usda.gov/ Services/docs.htm?docid=18349
Saldanha LG, Salem N,Jr, Brenna JT. Workshop on DHA as a required nutrient: overview. Prostaglandins Leukot Essent Fatty Acids. 2009 Aug-Sep;81(2-3):233-6.
Schaeffer L, Gohlke H, Müller M, Heid IM, Palmer LJ, Kompauer I, Demmelmair H, Illig T, Koletzko B, Heinrich J. Common genetic variants of the FADS1 FADS2 gene cluster and their reconstructed haplotypes are associated with the fatty acid composition in phospholipids. Hum Mol Genet. 2006 Jun;15(11):1745-56.
Sinclair AJ, Attar–Bashi NM, Li D. What is the role of α–linolenic acid for mammals? Lipids. 2002 Dec;37(12):1113-23.
Sprecher H. Metabolism of highly unsaturated n-3 and n-6 fatty acids. Biochim Biophys Acta. 2000 Jul;1486(2-3):219-31.
Tanaka T, Shen J, Abecasis GR, Kisialiou A, Ordovas JM, Guralnik JM, Singleton A, Bandinelli S, Cherubini A, Arnett D, et al. Genome-wide association study of plasma polyunsaturated fatty acids in the InCHIANTI Study. PLoS Genet. 2009 Jan;5(1):e1000338.
US Food and Drug Administration, Department of Health and Human Services. Substances affirmed as generally recognized as safe: menhaden oil. 1997. Retrieved from http://www.gpo.gov/fdsys/ pkg/FR-1997-06-05/html/97-14683.htm
USDA (US Department of Agriculture). National nutrient database 2011. Retrieved from http://www.nal.usda.gov/fnic/ foodcomp/search/ WHO/FAO joint consultation.
WHO and FAO joint consultation: fats and oils in human nutrition. Nutr Rev. 1995 Jul;53(7):202-5.
Brain health and response to cocoa in older people
This study investigated the relationship between neurovascular coupling and cognitive function in elderly individuals with vascular risk factors to determine whether neurovascular coupling could be modified by cocoa consumption. Sixty older people were studied in a parallel- arm, double-blind clinical trial of neurovascular coupling and cognition in response to 24 hours and 30 days of cocoa consumption. Measures included Mini- Mental State Examination and Trail Making Test A and B. Neurovascular coupling was measured from the beat- to-beat blood flow velocity responses in the middle cerebral arteries to the N-Back Task. The results showed that neurovascular coupling was associated with Trails B scores and performance on the 2-Back Task. Higher coupling was also associated with significantly higher anisotropy in cerebral white matter hyperintensities. 30 days of cocoa consumption was associated with increased coupling and improved Trails B times in those with impaired neurovascular coupling at baseline. The authors conclude that there is a strong correlation between neurovascular coupling and cognitive function and that both can be improved by regular cocoa consumption in individuals with baseline impairments, and that better coupling is also associated with greater white matter structural integrity. Neurology. 2013 August. PMID: 23925758
Associations between active travel to work and overweight, hypertension, and diabetes
Increasing active travel through walking, biking, and public transport, is promoted as a key strategy to increase physical activity and reduce the growing burden of noncommunicable diseases globally. However, not much is known about any associated cardiovascular health benefits in low-
and middle-income countries. This was a cross-sectional study of 3902 participants in the Indian Migration Study. Associations between mode
and duration of active travel and cardiovascular risk factors were assessed using random-effect logistic regression models adjusting for age, sex, caste, standard of living, occupation, factory location, leisure time physical activity, daily fat intake, smoking status, and alcohol use. The prevalence of overweight or obesity was 50.0%, 37.6%, 24.2%, 24.9%; hypertension was 17.7%, 11.8%, 6.5%, 9.8%; and diabetes was 10.8%, 7.4%, 3.8%, 7.3% in participants who travelled to work by private transport, public transport, bicycling, and walking respectively. Those walking or bicycling to work were significantly less likely to be overweight or obese and those travelling by private transport. Those bicycling to work were less likely to have hypertension or diabetes. There was a dose-response relationship between duration of bicycling to work and these health factors. PLoS Med. 2013 June. PMID: 23776412
High caloric intake at breakfast vs. dinner and impact on weight loss
This study examined time-of-day nutrient intake and its association with metabolic syndrome. The goal of the study was to compare a weight loss diet with high caloric intake during breakfast to an isocaloric diet with high caloric intake at dinner. Overweight and obese women (mean BMI 32.4) with metabolic syndrome were randomized into two isocaloric (1400 kcal) weight loss groups, a breakfast (700 kcal breakfast, 500 kcal lunch, 200 kcal dinner) or a dinner group (200 kcal breakfast, 500 kcal lunch, 700 kcal dinner) for 12 weeks. The results showed that the breakfast group showed greater weight loss and waist circumference reduction. Although fasting glucose, insulin, and ghrelin were reduced in both groups, fasting glucose, insulin, and HOMA- IR decreased significantly to a greater extent in the breakfast group. Mean triglyceride levels decreased by 33.6% in the breakfast group, but increased by 14.6% in the dinner group. Oral glucose tolerance test led to a greater decrease of glucose and insulin in the breakfast group. In response to meal challenges, the overall daily glucose, insulin, ghrelin, and mean hunger scores were significantly lower, whereas mean satiety scores were significantly higher in the breakfast group. Obesity (Silver Spring). 2013 March. PMID: 23512957.