Introduction
Forty percent of Canadians develop cancer in their lifetime, and twenty-five percent of these cases are fatal (Canadian Cancer Society 2014). The standards of care for cancer are chemotherapy, radiation, and/or surgery, because these modalities have demonstrated the highest treatment efficacy in clinical practice (Canadian Cancer Society 2012, Canadian Cancer Society 2015). However, patients diagnosed with chemotherapy- and radiotherapy-resistant cancers face treatment challenges (Li 2014). Thus, treatments adjuvant to conventional therapy are being explored.
Turmeric (Curcuma longa) is a plant native to Asia (Shishodia 2005). This nutraceutical has garnered interest as a complementary cancer therapy, as observational evidence has demonstrated low rates of colorectal, prostate, and lung cancers in Asia, where high amounts of turmeric are consumed (Aggarwal 2003, Lao 2006, Sinha 2003).
Curcumin (diferuloylmethane) is the most thoroughly researched active component of turmeric (Chattopadhyay 2004, Lao 2006, Shishodia 2005). Research evidence demonstrates that curcumin is more effective when used in conjunction with chemotherapy than as a stand-alone therapy (Kusuhara 2012, Lin 2007). This is due to its ability to downregulate resistance proteins (Guo 2014, Rana 2015, Roy 2014) and modulate cancer stem cells, which are both integral mechanisms to cancer resistance, metastasis, and recurrence (Buhrmann 2014, Shakibaei 2014). Moreover, curcumin antagonizes many of chemotherapy’s negative side effects such as promotion of cell proliferation through NF-kB (Cabrespine-Faugeras 2010, Melisi 2007). This paper investigates the clinical potential for curcumin use with conventional therapy to improve treatment outcomes in a variety of cancers.
General Cancer Pathways
Figure 1: Cellular pathways involved in cancer pathology and the effects of curcumin and chemotherapy
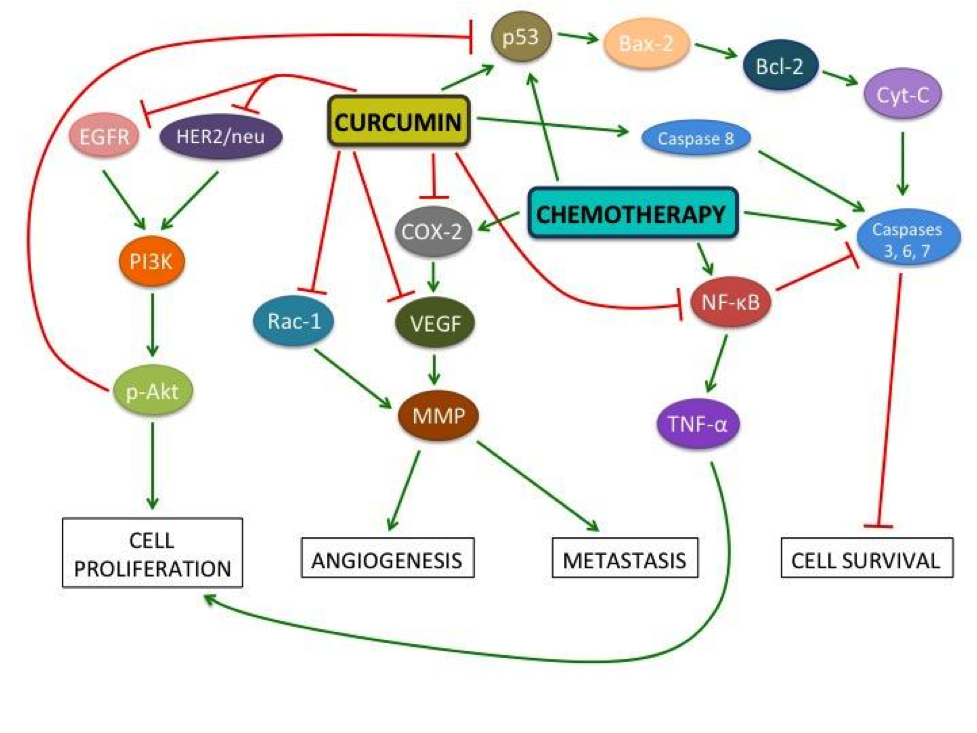
This diagram illustrates the effects of curcumin and chemotherapy on various molecules involved in cancer. Curcumin decreases angiogenesis, tumour cell proliferation, metastasis, and cancer cell survival. Nuclear factor-kappaB (NF-kB) activation facilitates tumour cell survival by downregulating caspase-3, -6, and -7, which are factors integral to cellular apoptosis (Cabrespine-Faugeras 2010, Melisi 2007). Paradoxically, chemotherapeutic drugs often upregulate NF-kB (Kamat 2009, Tharakan 2010), while curcumin inhibits it (Cabrespine-Faugeras 2010). NF-kB upregulates the inflammatory factor TNF-, which leads to increased cellular proliferation. Curcumin and chemotherapy drugs inhibit Bcl-2 – an apoptosis suppressing protein – by upregulating tumour suppressor protein p53. In turn, p53 upregulates Bcl-2-associated X protein (BAX), which directly antagonizes Bcl-2 (Cabrespine-Faugeras 2010). The ratio of BAX to Bcl-2 determines if a cell will survive or undergo apoptosis; curcumin increases this ratio, leading to apoptosis (Jiang 2015). Epidermal growth factor receptor (EGFR) and human epidermal growth factor receptor-2 (HER2)/neu signaling pathways lead to upregulation of Protein Kinase B (p-Akt) which causes angiogenesis, prevents cell cycle arrest, and thus increases cancer cell survival (Cabrespine-Faugeras 2010). Curcumin inhibits EGFR and HER2/neu thereby decreasing both angiogenesis and cell survival. Cyclooxygenase-2 (COX-2) is upregulated by chemotherapeutic drugs. It upregulates vascular endothelial growth factor (VEGF), which stimulates angiogenesis (Cabrespine-Faugeras 2010). This happens via upregulation of matrix metalloproteinases (MMPs), a critical step for metastasis (Cabrespine-Faugeras 2010, Chen 2014, Lin 2009). MMPs are also activated by Rac1 (Chen 2014). Curcumin inhibits both COX-2 and VEGF, thereby suppressing angiogenesis (Cabrespine-Faugeras 2010). Curcumin also downregulates the expression of multidrug-resist proteins, MRP1 and PGP1, which desensitize receptors to chemotherapy (Roy 2014).
Breast Cancer
Bayet-Robert and colleagues (2010) administered a combined treatment of oral curcumin and Docetaxel to patients with advanced and metastatic breast cancer. One third of patients initially had inoperable cancers that became operable by the end of the study. Furthermore, none of the patients demonstrated disease progression. Of the nine patients evaluated for response, one patient had no residual tumour, six partially responded to treatment, and two remained in a stable disease state. Patients had lowered tumour marker and VEGF levels, indicating reduced cancer cell survival and angiogenesis (Bayet-Robert 2010). In healthy human subjects, breast cancer resistance protein (BCRP)-expressing cells treated with curcumin had increased uptake of sulphasalazine – an anti-inflammatory drug that inhibits NF-kB activation (Kusuhara 2012). BCRP plays a role in chemoresistance, thus, this study suggests that curcumin may act as a chemosensitizer in humans (Kusuhara 2012).
Curcumin increased breast cancer cell sensitivity to other chemotherapeutic drugs, including MMC, Doxorubicin, Tamoxifen, Paclitaxel, Trichostatin A and 5-Fluorouracil (5-FU), in in vitro and in vivo animal studies. In turn, curcumin can reduce the drug concentrations necessary to achieve equivalent drug efficacy (Aggarwal 2005, Chen 2013, De Gasperi 2009, Jiang 2013, Kang 2009, Vinod 2013, Yan 2013, Zhou 2009, Zhou 2011). Curcumin may also reduce side effects of chemotherapeutic drugs, such as weight loss, renal toxicity, and cytotoxicity (Zhou 2009, Zhou 2011).
Curcumin has demonstrated antiproliferative, anti-angiogenic, anti-metastatic, and pro-apoptotic effects in vivo and in vitro when used alone or in conjunction with chemotherapeutic drugs in hormone receptor-positive and -negative breast cancers (Bachmeier 2007, Chen 2013, De Gasperi 2009, Liu 2009, Shao 2002, Vinod 2013, Yan 2013, Zhou 2009). For instance, MMC with curcumin reduced tumor weight by 60.4% more than MMC alone in in vivo xenografts (Zhou 2011). A 21% reduction in VEGF levels was also observed after six cycles of combined Docetaxel and curcumin treatment (Bayet-Robert 2010). Curcumin suppressed the Paclitaxel-induced expression of antiapoptotic (e.g. Bcl-2), proliferative (e.g. COX-2), and metastatic proteins (e.g. VEGF and MMP-9) in vitro in chemotherapy-resistant cells, while significantly decreasing metastasis in in vivo xenografts (Aggarwal 2005).
Colorectal Cancer
When administered alone or alongside chemotherapy drugs, curcumin has effectively suppressed inflammatory factors such as TNF-α, thus upregulating tumour suppressors, inhibiting cancer cell proliferation, and causing cancer cell apoptosis in in vitro and in vivo models (Guo 2013, Guo 2015, Lim 2014, Wang 2015). Toden et al. (2015) found that curcumin sensitizes colorectal cancer cells to 5-FU in 5-FU-resistant cells and xenografts. Curcumin’s ability to enhance the apoptotic and anti-proliferative effects of chemotherapeutic drugs such as 5-FU and Oxaliplatin is significant in the treatment of both non-resistant and resistant cancers (Guo 2014, Rana 2015). Curcumin’s chemosensitizing effects modulate the activity of cancer stem cells, which are thought to cause cancer cell resistance (Buhrmann 2014, Shakibaei 2014). Furthermore, a human study found that curcumin promotes positive outcomes in colorectal cancer patients, such as weight gain and upregulation of the tumour suppressor, p53 (He 2011).
Pancreatic Cancer
Pancreatic cancer is aggressive and has an overall poor prognosis (Mayo Foundation 2015). It is often detected at advanced stages, and its 5-year observed survival rate is 15-20% (Canadian Cancer Society 2015). Animal studies demonstrate that curcumin can work synergistically with Gemcitabine (Kunnumakkara 2007); however, human studies have shown conflicting results.
In a Phase I/II trial, patients receiving curcumin and Gemcitabine reported severe gastrointestinal side effects such as diarrhea, nausea and intractable abdominal pain, as well as poor tolerability (Epelbaum 2010). It is unclear whether these side effects can be attributed to the progression of late-stage cancer and chemotherapy, or to curcumin alone, as there was no control group (Epelbaum 2010). A study conducted with Gemcitabine-resistant patients found that curcumin was well-tolerated (Kanai 2011). Median compliance was 100% and zero patients withdrew due to intolerability. The median survival time was 161 days. Several patients reported reduced chemotherapy-related side effects such as fatigue, pain, and constipation (Kanai 2011). Some patients exhibited partial response (Dhillon 2008) or stable disease (Kanai 2011) in some studies; however, these effects are not applicable to all patients (Dhillon 2008, Kanai 2011).
Leukemia
Ghalaut et al. (2012) investigated the effect of oral consumption of curcumin in leukemia patients undergoing Imatinib chemotherapy in comparison to those undergoing chemotherapy alone. Treatment potentiated a favourable haematological response (i.e. lowered platelet, white blood cell, and immature granulocyte and basophil count) and decreased nitric oxide levels — an inducer of tumour growth, invasion, and metastasis (Ghalaut 2012). Curcumin has also been shown to decrease tumour growth rate and promote organism survival in xenograft mice models (William 2008, Yu 2013, Zunino 2013). In vitro studies have demonstrated the ability of curcumin to potentiate the effects of chemotherapeutic drugs including Tamoxifen (Pedroso 2013), L-asparaginase (Wang 2012), Methotrexate (Dhanasekaran 2013), Etoposide (Papiez 2014), Lonidamine (Sanchez 2010), and arsenic trioxide (Sanchez 2010), but not Silibinin (Pesakhov 2010) and Cisplatin (Sanchez 2010).
Bladder Cancer
Using an orthotopic mouse model, Tharakan and colleagues (2010) found that curcumin potentiates the apoptotic and anti-proliferative effects of Gemcitabine. Curcumin decreased biomarkers of proliferation and angiogenesis such as COX-2 and VEGF, respectively, with maximal effectiveness using combination therapy (Tharakan 2010). A study conducted in in vitro and xenograft models, using curcumin and Bacillus Calmette-Guerin (BCG), a standard drug for bladder cancer, demonstrated similar results with combination therapy being more effective than either treatment alone (Kamat 2009). Similar results have been achieved in an in vivo xenograft mouse model (Chadalapaka 2008). Tian and colleagues (2008) found that curcumin had a greater inhibitory effect on NF-kB than Cisplatin, but no synergistic effect was found between the two.
Liver cancer
Studies using Hepatocellular Carcinoma (HCC) xenografts have shown that intravenous injection or oral consumption of curcumin may produce anti-angiogenic and anti-proliferative effects on tumour development (Anand 2012, Cui 2006, Dai 2013, Ning 2009, Yoysungneon 2008). Curcumin has also demonstrated synergistic anti-cancer effects when administered in conjunction with chemotherapeutic drugs such as Doxorubicin (Zhao 2014) and Paclitaxel (Ganta 2010) in in vivo animal models, and Cisplatin (Notarbartolo 2005), 5-FU (Zhu 2013), and Adriamycin (Qian 2011) in vitro. Synergistic effects were also seen in combined treatment with anti-angiogenic agents such as Leflunomide and Perindopril in in vivo mice models (Nasr 2014).
Cancers of the Head and Neck
In in vivo xenografts, curcumin inhibited tumour development (LoTempio 2005, Odot 2004, Wang 2008, Zhu 2012) and increased organism survival (Clark 2010) when compared to control mice. Additionally, curcumin potentiated the effects of radiotherapy (Chiang 2014, Khalif 2009) and chemotherapeutic drugs 5-FU (Tian 2012) and Cisplatin (Duarte 2010) in mouse models.
Uterine and Cervical Cancer
Sreekanth et al. (2011) found that, in xenografts and chemically-induced mouse models, curcumin enhanced Paclitaxel’s antitumour effect by decreasing expression of anti-apoptotic factors NF-kB and p-Akt. Thus, combination therapy led to decreased tumour incidence and volume when compared to groups treated with either Paclitaxel or curcumin alone (Sreekanth 2011). Additionally, curcumin sensitized Cisplatin-resistant human cervical cancer cells to Cisplatin through modulation of multidrug-resistant proteins, such as MRP1 (Roy 2014). Furthermore, in a xenograft study using a hormone therapy called Letrozole, curcumin was shown to synergistically enhance inhibition of endometrial tumour growth (Liang 2009).
Prostate Cancer
A study on human prostate cancer cells found that curcumin alone inhibited 20% of the production of prostate-specific antigen – a biomarker of inflammation in the prostate (Ide 2010). These results have also been seen in xenografts injected with human prostate cancer cells (Dorai 2001). Additionally, studies examining xenografts and cancer cell lines found that curcumin sensitizes both hormone-resistant and hormone-sensitive prostate cancer cells to TNF-related apoptosis-inducing ligand (TRAIL), a cytokine that induces apoptosis (An 2014, Andrzejewski 2008). The combined therapy of TRAIL and curcumin induced apoptosis in cancer cells by inhibiting anti-apoptotic p-Akt and NF-kB (Andrzejewski 2008). Furthermore, curcumin and chemotherapeutic drugs, such as Paclitaxel, have demonstrated synergistic effects in reducing angiogenesis, proliferation, and metastasis (Cabrespine-Faugeras 2010).
Lung Cancer
In human lung cancer cell lines, curcumin reduced metastasis by inhibiting the Rac1 signaling pathway and MMP-2 and MMP-9 expression (Chen 2014a). In another in vitro study, curcumin was found to increase sensitivity of cells that were initially resistant to Cisplatin, leading to reduced cell proliferation (Chen 2014b). Similar effects were seen with Docetaxel in non-small cell lung cancers (Yin 2012).
Cancers with Inadequate Evidence
Clinical recommendations cannot be made for the use of curcumin as a complementary therapy for brain, gastric, skin, kidney, and bone cancers.
Brain cancer studies conducted on human glioma cell lines have shown that curcumin exerts an apoptotic and chemosensitizing effect by reducing the activity of transcription factors such as NF-kB (Dhandapani 2007). However, curcumin does not cross the blood-brain barrier unless delivered in a solubilized form, making it unsuitable as an adjuvant therapy for brain cancers (Purkayastha 2009).
Several studies conducted using animal models and human gastric cancer cell lines have shown the benefits of curcumin in the treatment of gastric cancer (Cai 2013, Deshpande 1997, Huang 1994, Yu 2011). Yu and colleagues (2011) found that curcumin reversed chemoresistance by downregulating NF-kB in vitro. They found that curcumin, in conjunction with Etoposide and Doxorubicin, suppresses cancer cell growth more effectively than these chemotherapeutics alone (Yu 2011). Nonetheless, as there are no human studies and very few in vivo studies for curcumin in gastric cancer, it is difficult to make clinical recommendations.
There are very few studies investigating the effects of curcumin in conjunction with chemotherapy in skin, kidney, and bone cancers. Most available studies are in vitro, and there is a significant lack of human or animal studies. Thus, there is insufficient evidence to make clinical recommendations for these cancers.
Bioavailability, Administration Methods & Analogues
When free curcumin is administered, it exhibits low bioavailability due to its low water solubility, high rate of metabolism, and poor absorption in the human body, thus limiting its potential anti-cancer effects (Anand 2007, Heger 2013). In order to reach a therapeutic dose in cancer patients, curcumin analogues and alternative administration methods aside from oral delivery are being explored.
Various forms of nanoparticles have been tested in colorectal (Chuah 2014), pancreatic (Bisht 2010, Yallapu 2013), breast (Yallapu 2012), lung (Yin 2013), and liver cancers (Duan 2010, Yen 2010). Other delivery methods such as liposomes have been tested in pancreatic (Mach 2009) and lung cancers (Rahman 2012); microspheres in lung cancer (Cao 2011); micelles in colorectal cancer (Abouzeid 2013); polymers in prostate (Boztas 2013), breast (Bansal 2014, Liu 2013), and colorectal cancers (Chen 2012); and implants in breast cancer (Bansal 2014).
Other studies have combined these administration methods in forms such as solid lipid nanoparticles to further improve potency (Francis 2014, Mulik 2010, Wang 2013). Kanai et al. (2013) were the first to employ a novel curcumin administration method in humans through a nanoparticle called Theracurmin. Theracurmin has reduced particle size by over 100 times, addressed the issue of inadequate aqueous solubility, and employed a sustained drug release system. These strategies have increased bioavailability and reduced toxicity in animal as well as human subjects (Kanai 2013).
The use of synthetic analogues is being explored as a possible alternative to overcome the issue of limited bioavailability of curcumin. By making substitutions to various functional groups, analogues can exhibit enhanced therapeutic efficacy (Adams 2004). Examples of analogues that have been developed are HO-3867 (Dayton 2010) for lung, colon, liver, breast, and ovarian cancers, DM-1 and DM-2 (Faião-Flores 2012) for breast cancer, and D6 for melanoma (Rozzo 2013). Generally, these studies have demonstrated positive effects on bioavailability, such as increased uptake, higher serum levels, and increased accumulation in target organs (Adams 2004, Dayton 2010, Faião-Flores 2012, Rozzo 2013). Furthermore, this improved bioavailability results in reduced angiogenesis, cancer cell survival, tumour proliferation, and metastasis, and downregulation of multidrug resistance (Adams 2004, Dayton 2010, Faião-Flores 2012, Rozzo 2013). Human studies that test the clinical utility of these methods are required.
Dosing & Safety
Several human studies have investigated optimal dosing and dose-limiting toxicities of orally administered curcumin in patients undergoing chemotherapy and in healthy subjects. Some studies demonstrated that patients can tolerate up to 8g/day of orally delivered curcumin in capsule form (Cheng 2001, Dhillon 2008, Kanai 2011), while other studies suggested that the dose may lead to gastrointestinal side effects and lack of compliance due to bulkiness (Bayet-Robert 2010, Epelbaum 2010, Lao 2006). Regardless, when administered orally, the dose of 8g/day is insufficient to reach systemic bioavailability as measured via serum concentrations (Epelbaum 2010).
Limitations
The current literature describing the therapeutic effects of curcumin is thorough for certain cancers, while it is limited for others – particularly those with low prevalence. In general, animal studies demonstrate overwhelmingly positive results, but the low bioavailability of free curcumin limits its clinical utility as an anti-cancer therapy in humans. Although some novel methods have been studied to address bioavailability, further research is required.
Moreover, the literature on curcumin as a complementary chemotherapy is quite heterogeneous, making it difficult to compare and amalgamate study results and come to a general conclusion. Factors such as administration method, dosing, analogue type, follow-up period, and outcome measures should be standardized in future research.
Clinical Recommendation
A clinical recommendation for the use of curcumin cannot be made at this time due to inadequate evidence and the continued challenges associated with bioavailability.
References
Abouzeid AH, Patel NR, Rachman IM, Senn S, & Torchilin VP. Anti-cancer activity of anti-GLUT1 antibody-targeted polymeric micelles co-loaded with curcumin and doxorubicin. Journal of drug targeting. 2013;21(10):994-1000.
Adams BK, Ferstl EM, Davis MC, Herold M, Kurtkaya S, Camalier RF, Hollingshead MG, Kaur G, Sausville EA, Rickles FR, Snyder JP, Liotta DC, & Shoji M. Synthesis and biological evaluation of novel curcumin analogs as anti-cancer and anti-angiogenesis agents. Bioorg Med Chem. 2004 Jul 15;12(14):3871-83.
Aggarwal BB, Kumar A, & Bharti AC. Anticancer potential of curcumin: preclinical and
clinical studies. Anticancer res. 2003;23(1A):363-398.
Aggarwal BB, Shishodia S, Takada Y, Banerjee S, Newman RA, Bueso-Ramos CE, & Price JE. Curcumin suppresses the paclitaxel-induced nuclear factor-κB pathway in breast cancer cells and inhibits lung metastasis of human breast cancer in nude mice. Clinical Cancer Research. 2005;11(20):7490-7498.
An D, Kim K, & Kim J. Microfluidic System Based High Throughput Drug Screening System for Curcumin/TRAIL Combinational Chemotherapy in Human Prostate Cancer PC3 Cells. Biomol Ther (Seoul). 2014;22(4):355-362. doi: 10.4062/biomolther.2014.078
Anand K, Sarkar A, Kumar A, Ambasta R, & Kumar P. Combinatorial Antitumor Effect of Naringenin and Curcumin Elicit Angioinhibitory Activities In Vivo. Nutrition and Cancer. 2012;64(5):714-724.
Anand P, Kunnumakkara A, Newman R, & Aggarwal B. Bioavailability of Curcumin: Problems and Promises. Molecular Pharmaceutics. 2007;4(6):807-818.
Andrzejewski T, Deeb D, Gao X, Danyluk A, Arbab AS, Dulchavsky SA, & Gautam SC. Therapeutic efficacy of curcumin/TRAIL combination regimen for hormone-refractory prostate cancer. Oncol Res. 2008;17(6):257-267.
Bachmeier BE, Nerlich AG, Iancu CM, Cilli M, Schleicher E, Vene R, Dell’Eva R, Jochum M, Albini A, & Pfeffer U. The chemopreventive polyphenol Curcumin prevents hematogenous breast cancer metastases in immunodeficient mice. Cellular Physiology and Biochemistry. 2007;19(1-4):137-152.
Bansal SS, Kausar H, Vadhanam MV, Ravoori S, Pan J, Rai SN, & Gupta RC. Curcumin Implants, Not Curcumin Diet, Inhibit Estrogen-Induced Mammary Carcinogenesis in ACI Rats. Cancer Prevention Research. 2014;7(4):456-465.
Bayet-Robert M, Kwiatowski F, Leheurteur M, Gachon F, Planchat E, Abrial C, Mouret-Reynier MA, Durando X, Barthomeuf C, & Chollet P. Phase I dose escalation trial of docetaxel plus curcumin in patients with advanced and metastatic breast cancer. Cancer biology & therapy. 2010;9(1):8-14.
Bisht S, Mizuma M, Feldmann G, Ottenhof NA, Hong SM, Pramanik D, Chenna V, Karikari C, Sharma R, Goggins MG, Rudek MA, Ravi R, Maitra A, & Maitra A. Systemic administration of polymeric nanoparticle-encapsulated curcumin (NanoCurc) blocks tumor growth and metastases in preclinical models of pancreatic cancer. Mol Cancer Ther. 2010 Aug;9(8):2255-64. doi: 10.1158/1535-7163.MCT-10-0172.
Boztas AO, Karakuzu O, Galante G, Ugur Z, Kocabas F, Altuntas CZ, & Yazaydin AO. Synergistic interaction of paclitaxel and curcumin with cyclodextrin polymer complexation in human cancer cells. Molecular pharmaceutics. 2013;10(7):2676-2683.
Buhrmann C, Kraehe P, Lueders C, Shayan P, Goel A, & Shakibaei M. Curcumin suppresses crosstalk between colon cancer stem cells and stromal fibroblasts in the tumor microenvironment: potential role of EMT. PLoS One. 2014 Sep 19;9(9):e107514. doi: 10.1371/journal.pone.0107514.
Cabrespine-Faugeras A, Bayet-Robert M, Bay JO, Chollet P, & Barthomeuf C. Possible benefits of curcumin regimen in combination with taxane chemotherapy for hormone-refractory prostate cancer treatment. Nutr Cancer. 2010;62(2):148-153. doi: 10.1080/01635580903305383
Cai XZ, Huang WY, Qiao Y, Du SY, Chen Y, Chen D, Yu S, Che RC, Liu N, & Jiang Y. Inhibitory effects of curcumin on gastric cancer cells: a proteomic study of molecular targets. Phytomedicine. 2013;20(6): 495-505.
Cao F, Ding B, Sun M, Guo C, Zhang L, & Zhai G. Lung-targeted delivery system of curcumin loaded gelatin microspheres. Drug delivery. 2011;18(8), 545-554.
Canadian Cancer Society’s Advisory Committee on Cancer Statistics. Canadian Cancer Statistics 2014. Toronto, ON: Canadian Cancer Society, 2014.
Canadian Cancer Society. Living with Cancer: A guide for people with cancer and their caregivers. Toronto, ON: Canadian Cancer Society, 2012.
Canadian Cancer Society. Treatment. Toronto, ON: Canadian Cancer Society, 2015. http://www.cancer.ca/en/cancer-information/diagnosis-and-treatment/treatment/?region=on
Chadalapaka G, Jutooru I, Chintharlapalli S, Papineni S, Smith R, Li X, & Safe S. Curcumin decreases specificity protein expression in bladder cancer cells. Cancer research. 2008;68(13):5345-5354.
Chattopadhyay I, Biswas K, Bandyopadhyay U, & Banerjee RK. Turmeric and curcumin: biological actions and medicinal applications. Current Science. 2004;87(1):44–53.
Chen MJ, Cheng YM, Lai PH, Wu JF, & Hsu YC. In vitro biocompatibility of thermally gelling liquid mucoadhesive loaded curcuminoids in colorectal cancer chemoprevention. Int J Colorectal Dis. 2012 Jul;27(7):869-78. doi: 10.1007/s00384-011-1393-3.
Chen Q, Zheng Y, Jiao D, Chen F, Hu H, Wu Y, & Lv G. Curcumin inhibits lung cancer cell migration and invasion through Rac1-dependent signaling pathway. The Journal of Nutritional Biochemistry. 2014a;25(2):177–185. doi:10.1016/j.jnutbio.2013.10.004
Chen P, Li J, Jiang H-G, Lan T, & Chen Y-C. Curcumin reverses cisplatin resistance in cisplatin-resistant lung cancer cells by inhibiting FA/BRCA pathway. Tumor Biology. 2014b. doi:10.1007/s13277-014-2996-4
Chen WC, Lai YA, Lin YC, Ma JW, Huang LF, Yang NS, Ho CT, Kuo SC, & Way TD. Curcumin Suppresses Doxorubicin-Induced Epithelial–Mesenchymal Transition via the Inhibition of TGF-β and PI3K/AKT Signaling Pathways in Triple-Negative Breast Cancer Cells. Journal of agricultural and food chemistry. 2013;61(48):11817-11824.
Cheng AL, Hsu CH, Lin JK, Hsu MM, Ho YF, Shen TS, Ko JY, Lin JT, Lin BR, Ming-Shiang W, Yu HS, Jee SH, Chen GS, Chen TM, Chen CA, Lai MK, Pu YS, Pan MH, Wang YJ, Tsai CC, & Hsieh CY. Phase I clinical trial of curcumin, a chemopreventive agent, in patients with high-risk or pre-malignant lesions. Anticancer research, 2001;21(4B), 2895-2900.
Chiang I, Liu YC, Hsu FT, Chien YC, Kao CHK, Lin WJ, Chung JG, & Hwang JJ. Curcumin synergistically enhances the radiosensitivity of human oral squamous cell carcinoma via suppression of radiation-induced NF-κB activity. Oncology reports. 2014;31(4):1729-1737.
Chuah LH, Roberts CJ, Billa N, Abdullah S, & Rosli R. Cellular uptake and anticancer effects of mucoadhesive curcumin-containing chitosan particles. Colloids Surf B Biointerfaces. 2014;116:228-236. doi:10.1016/j.colsurfb.2014.01.007.
Clark C, McEachern M, Shah S, Rong Y, Rong X, & Smelley C. Curcumin Inhibits Carcinogen and Nicotine-Induced Mammalian Target of Rapamycin Pathway Activation in Head and Neck Squamous Cell Carcinoma. Cancer Prevention Research. 2010;3(12):1586-1595.
Cui S, Qu X, Xie Y, Zhou L, Nakata M, Makuuchi M, & Tang W. Curcumin inhibits telomerase activity in human cancer cell lines. Int J Mol Med. 2006;18(2):227-31
Dai X, Yin H, Sun L, Hu X, Zhou C, Zhou Y, Zhang W, Huang XE, & Li XC. Potential Therapeutic Efficacy of Curcumin in Liver Cancer. Asian Pacific Journal of Cancer Prevention. 2013;14(6):3855-3859.
Dayton A, Selvendiran K, Kuppusamy M, Rivera B, Meduru S, & Kálai T. Cellular Uptake, Retention and Bioabsorption of HO-3867, a Fluorinated Curcumin analog with Potential Antitumor Properties. Free Radical Biology and Medicine. 2010;49:S179.
De Gasperi M, & Cavazos D. Curcumin Modulates Tamoxifen Response in Resistant Breast Cancer Cells. Cancer Research. 2009;69(24 Supplement):3098-3098.
Deshpande SS, Ingle AD, & Maru GB. Inhibitory effects of curcumin-free aqueous turmeric extract on benzo [a] pyrene-induced forestomach papillomas in mice. Cancer letters. 1997;118(1):79-85.
Dhanasekaran S, Biswal B, Sumantran V, & Verma R. Augmented sensitivity to methotrexate by curcumin induced overexpression of folate receptor in KG-1 cells. Biochimie. 2013;95(8):1567-1573.
Dhandapani KM, Mahesh VB, & Brann DW. Curcumin suppresses growth and chemoresistance of human glioblastoma cells via AP-1 and NFκB transcription factors. Journal of Neurochemistry. 2007;102(2):522–538. doi:10.1111/j.1471-4159.2007.04633.x
Dhillon N, Aggarwal BB, Newman RA, Wolff RA, Kunnumakkara AB, Abbruzzese JL, Ng CS, Badmaev V, & Kurzrock R. Phase II trial of curcumin in patients with advanced pancreatic cancer. Clinical Cancer Research. 2008;14(14):4491-4499.
Dorai T, Cao YC, Dorai B, Buttyan R, & Katz AE. Therapeutic potential of curcumin in human
prostate cancer. III. Curcumin inhibits proliferation, induces apoptosis, and inhibits angiogenesis of LNCaP prostate cancer cells in vivo. Prostate. 2001;47(4):293-303. doi: 10.1002/pros.1074
Duan J, Zhang Y, Han S, Chen Y, Li B, & Liao M. Synthesis and in vitro/in vivo anti-cancer evaluation of curcumin-loaded chitosan/poly(butyl cyanoacrylate) nanoparticles. International Journal of Pharmaceutics. 2010;400(1-2):211-220.
Duarte V, Han E, Veena M, Salvado A, Suh J, & Liang L. Curcumin Enhances the Effect of Cisplatin in Suppression of Head and Neck Squamous Cell Carcinoma via Inhibition of IKK Protein of the NF B Pathway. Molecular Cancer Therapeutics. 2010;9(10):2665-2675.
Epelbaum R, Schaffer M, Vizel B, Badmaev V, & Bar-Sela G. Curcumin and gemcitabine in patients with advanced pancreatic cancer. Nutrition and cancer. 2010;62(8):1137-1141.
Faião-Flores F, Suarez JAQ, Pardi PC, & Maria DA. DM-1, sodium 4-[5-(4-hydroxy-3-methoxyphenyl)-3-oxo-penta-1, 4-dienyl]-2-methoxy-phenolate: a curcumin analog with a synergic effect in combination with paclitaxel in breast cancer treatment. Tumor Biology. 2012;33(3):775-785.
Francis AP, Murthy PB, & Devasena T. Bis-Demethoxy Curcumin Analog Nanoparticles: Synthesis, Characterization, and Anticancer Activity In Vitro. Journal of nanoscience and nanotechnology. 2014;14(7):4865-4873.
Ganta S, Devalapally H, Amiji M. Curcumin enhances oral bioavailability and anti-tumor therapeutic efficacy of paclitaxel upon administration in nanoemulsion formulation. Journal of Pharmaceutical Sciences. 2010;99(11):4630-4641.
Ghalaut V, Sangwan L, Dahiya K, Ghalaut P, Dhankhar R, & Saharan R. Effect of imatinib therapy with and without turmeric powder on nitric oxide levels in chronic myeloid leukemia. Journal of Oncology Pharmacy Practice. 2012;18(2):186-190
Guo LD, Chen XJ, Hu YH, Yu ZJ, Wang D, Liu JZ. Curcumin inhibits proliferation and induces apoptosis of human colorectal cancer cells by activating the mitochondria apoptotic pathway. Phytother Res. 2013 Mar;27(3):422-30. doi: 10.1002/ptr.4731. Epub 2012 May 25.
Guo LD, Shen YQ, Zhao XH, Guo LJ, Yu ZJ, Wang D, Liu LM, Liu JZ. Curcumin Combined with Oxaliplatin Effectively Suppress Colorectal Carcinoma in vivo Through Inducing Apoptosis. Phytother Res. 2014 Nov 24. doi: 10.1002/ptr.5257. [Epub ahead of print]
Guo Y, Shu L, Zhang C, Su ZY, Kong AN. Curcumin inhibits anchorage-independent growth of HT29 human colon cancer cells by targeting epigenetic restoration of the tumor suppressor gene DLEC1. Biochem Pharmacol. 2015 Jan 29. pii: S0006-2952(15)00078-7. doi: 10.1016/j.bcp.2015.01.009. [Epub ahead of print]
He ZY, Shi CB, Wen H, Li FL, Wang BL, Wang J. Upregulation of p53 expression in patients with colorectal cancer by administration of curcumin. Cancer Invest. 2011 Mar;29(3):208-13. doi: 10.3109/07357907.2010.550592
Heger M, van Golen R, Broekgaarden M, & Michel M. The Molecular Basis for the Pharmacokinetics and Pharmacodynamics of Curcumin and Its Metabolites in Relation to Cancer. Pharmacological Reviews. 2013;66(1):222-307.
Huang MT, Lou YR, Ma W, Newmark HL, Reuhl KR, & Conney AH. Inhibitory effects of dietary curcumin on forestomach, duodenal, and colon carcinogenesis in mice. Cancer research. 1994;54(22):5841-5847.
Ide H, Tokiwa S, Sakamaki K, Nishio K, Isotani S, Muto S, Hama T, Masuda H, & Horie S. Combined inhibitory effects of soy isoflavones and curcumin on the production of prostate-specific antigen. Prostate. 2010;70(10):1127-1133. doi: 10.1002/pros.21147
Jiang AJ, Jiang G, Li LT, & Zheng JN. Curcumin induces apoptosis through mitochondrial pathway and caspases activation in human melanoma cells. Mol Biol Rep. 2015;42(1):267-275. doi: 10.1007/s11033-014-3769-2
Jiang M, Huang O, Zhang X, Xie Z, Shen A, Liu H, Geng M & Shen, K. (2013). Curcumin induces cell death and restores tamoxifen sensitivity in the antiestrogen-resistant breast cancer cell lines MCF-7/LCC2 and MCF-7/LCC9. Molecules. 2013;18(1):701-720.
Kamat AM, Tharakan ST, Sung B, & Aggarwal BB. Curcumin potentiates the antitumor effects of Bacillus Calmette-Guerin against bladder cancer through the downregulation of NF-κB and upregulation of TRAIL receptors. Cancer research. 2009;69(23):8958-8966. doi: 10.1158/0008-5472.CAN-09-2045
Kanai M, Yoshimura K, Asada M, Imaizumi A, Suzuki C, Matsumoto S, Nishimura T, Mori Y, Masui T, Kawaguchi Y, Yanagihara K, Yazumi S, Chiba T, Guha S, & Aggarwal BB. A phase I/II study of gemcitabine-based chemotherapy plus curcumin for patients with gemcitabine-resistant pancreatic cancer. Cancer chemotherapy and pharmacology. 2011;68(1):157-164.
Kanai M, Otsuka Y, Otsuka K, Sato M, Nishimura T, & Mori Y. A phase I study investigating the safety and pharmacokinetics of highly bioavailable curcumin (Theracurmin®) in cancer patients. Cancer Chemotherapy and Pharmacology. 2013;71(6):1521-1530.
Kang HJ, Lee SH, Price JE, & Kim LS. Curcumin Suppresses the Paclitaxel‐Induced Nuclear Factor‐κB in Breast Cancer Cells and Potentiates the Growth Inhibitory Effect of Paclitaxel in a Breast Cancer Nude Mice Model. The breast journal. 2009;15(3): 223-229.
Khalif A, Lev-Ari S, Vexler A, Barnea I, Starr A, & Karaush V. Curcumin: A potential radio-enhancer in head and neck cancer. The Laryngoscope. 2009;119(10):2019-2026.
Kunnumakkara AB, Guha S, Krishnan S, Diagaradjane P, Gelovani J, Aggarwal BB. Curcumin potentiates antitumor activity of gemcitabine in an orthotopic model of pancreatic cancer through suppression of proliferation, angiogenesis, and inhibition of nuclear factor-kappaB-regulated gene products. Cancer Res. 2007 Apr 15;67(8):3853-61.
Kusuhara H, Furuie H, Inano A, Sunagawa A, Yamada S, Wu C, Fukizawa S, Morimoto N, Ieiri I, Morishita M, Sumita K, Mayahara H, Fujita T, Maeda K, & Sugiyama Y. Pharmacokinetic interaction study of sulphasalazine in healthy subjects and the impact of curcumin as an in vivo inhibitor of BCRP. British journal of pharmacology. 2012;166(6):1793-1803.
Lao CD, Ruffin MT, Normolle D, Heath DD, Murray SI, Bailey JM, Boggs ME, Crowell J, Rick CL, & Brenner DE. Dose escalation of a curcuminoid formulation. BMC complementary and alternative medicine. 2006;6(1):10. doi:10.1186/1472-6882-6-10
Li Y, Zhang T. Targeting cancer stem cells by curcumin and clinical applications. Cancer Lett. 2014;346(2):197-205. doi: 10.1016/j.canlet.2014.01.012
Liang YJ, Hao Q, Wu YZ, Wang QL, Wang JD, Hu YL. Aromatase inhibitor letrozole in synergy with curcumin in the inhibition of xenografted endometrial carcinoma growth. Int J Gynecol Cancer. 2009 Oct;19(7):1248-52. doi: 10.1111/IGC.0b013e3181b33d76
Lim TG, Lee SY, Huang Z, Lim do Y, Chen H, Jung SK, Bode AM, Lee KW, Dong Z. Curcumin suppresses proliferation of colon cancer cells by targeting CDK2. Cancer Prev Res (Phila). 2014 Apr;7(4):466-74. doi: 10.1158/1940-6207.CAPR-13-0387. Epub 2014 Feb 18.
Lin YG, Kunnumakkara AB, Nair A, Merritt WM, Han LY, Armaiz-Pena GN, Kamat AA, Spannuth WA, Gershenson DM, Lutgendorf SK, Aggarwal BB, Sood AK. Curcumin inhibits tumor growth and angiogenesis in ovarian carcinoma by targeting the nuclear factor-kappaB pathway. Clin Cancer Res. 2007;13(11):3423-30.
Lin S-S, Lai K-C, Hsu S-C, Yang J-S, Kuo C-L, Lin J-P, Ma Y-S, W C-C, & Chung J-G. Curcumin inhibits the migration and invasion of human A549 lung cancer cells through the inhibition of matrix metalloproteinase-2 and -9 and Vascular Endothelial Growth Factor (VEGF). Cancer Letters. 2009;285(2):127-133. doi:10.1016/j.canlet.2009.04.037
Liu L, Sun L, Wu Q, Guo W, Li L, Chen Y, Li Y, Gong C, Qian Z, & Wei Y. Curcumin loaded polymeric micelles inhibit breast tumor growth and spontaneous pulmonary metastasis. International journal of pharmaceutics. 2013;443(1):175-182.
Liu Q, Loo WT, Sze SCW, & Tong Y. Curcumin inhibits cell proliferation of MDA-MB-231 and BT-483 breast cancer cells mediated by down-regulation of NFκB, cyclinD and MMP-1 transcription. Phytomedicine. 2009;16(10):916-922.
LoTempio M. Curcumin Suppresses Growth of Head and Neck Squamous Cell Carcinoma. Clinical Cancer Research. 2005;11(19):6994-7002.
Mach CM, Mathew L, Mosley SA, Kurzrock R, & Smith JA. Determination of minimum effective dose and optimal dosing schedule for liposomal curcumin in a xenograft human pancreatic cancer model. Anticancer Res. 2009 Jun;29(6):1895-9.
Mayo Foundation for Medical Education and Research. Pancreatic Cancer. Scottsdale, AZ: Mayo Clinic, 2015.
Melisi D, & Chiao PJ. NF-κB as a target for cancer therapy. Expert Opinion on Therapeutic Targets. 2007;11(2):133–144. doi:10.1517/14728222.11.2.133
Mulik RS, Mönkkönen J, Juvonen RO, Mahadik KR, & Paradkar AR. Transferrin mediated solid lipid nanoparticles containing curcumin: enhanced in vitro anticancer activity by induction of apoptosis. International journal of pharmaceutics. 2010;398(1):190-203.
Nasr M, Selima E, Hamed O, & Kazem A. Targeting different angiogenic pathways with combination of curcumin, leflunomide and perindopril inhibits diethylnitrosamine-induced hepatocellular carcinoma in mice. European Journal of Pharmacology. 2014;723:267-275.
Ning L, Wentworth L, Chen H, & Weber S. Down-regulation of Notch1 signaling inhibits tumor growth in human hepatocellular carcinoma. American Journal of Translational Research. 2009;1(4): 358-366
Notarbartolo M, Poma P, Perri D, Dusonchet L, Cervello M, & D’Alessandro N. Antitumor effects of curcumin, alone or in combination with cisplatin or doxorubicin, on human hepatic cancer cells. Analysis of their possible relationship to changes in NF-kB activation levels and in IAP gene expression. Cancer Lett. 2005;224(1):53-55.
Odot J, Albert P, Carlier A, Tarpin M, Devy J, & Madoulet C. In vitro andin vivo anti-tumoral effect of curcumin against melanoma cells. International Journal of Cancer. 2004;111(3):381-387.
Papiez MA, & Krzysciak W. The dual effect of curcumin on etoposide action in leukemic and healthy bone marrow cells of rats with acute myeloid leukemia. Folia Med Cracov. 2014; 54(2); 71-9.
Pedroso L, Fávero G, de Camargo L, Mainardes R, & Khalil N. Effect of the o-methyl catechols apocynin, curcumin and vanillin on the cytotoxicity activity of tamoxifen. Journal of Enzyme Inhibition and Medicinal Chemistry. 2013;28(4):734-740.
Pesakhov S, Khanin M, Studzinski G, & Danilenko M. Distinct Combinatorial Effects of the Plant Polyphenols Curcumin, Carnosic Acid, and Silibinin on Proliferation and Apoptosis in Acute Myeloid Leukemia Cells. Nutrition and Cancer. 2010;62(6):811-824.
Purkayastha S, Berliner A, Fernando SS, Ranasinghe B, Ray I, Tariq H, & Banerjee P. Curcumin blocks brain tumor formation. Brain Research. 2009;1266:130–138. doi:10.1016/j.brainres.2009.01.066
Qian H, Yang Y, & Wang X. Curcumin enhanced adriamycin-induced human liver-derived Hepatoma G2 cell death through activation of mitochondria-mediated apoptosis and autophagy. European Journal of Pharmaceutical Sciences. 2011;43(3):125-131.
Rahman, S, Cao S, Steadman K. J, Wei M, & Parekh H. S. Native and β-cyclodextrin-enclosed curcumin: entrapment within liposomes and their in vitro cytotoxicity in lung and colon cancer. Drug Delivery.2012;19(7):346–353. doi:10.3109/10717544.2012.721143
Rana C, Piplani H, Vaish V, Nehru B, & Sanyal SN. Downregulation of PI3-K/Akt/PTEN pathway and activation of mitochondrial intrinsic apoptosis by Diclofenac and Curcumin in colon cancer. Mol Cell Biochem. 2015 Apr;402(1-2):225-41. doi: 10.1007/s11010-015-2330-5. Epub 2015 Feb 3.
Rozzo C, Fanciulli M, Fraumene C, Corrias A, Cubeddu T, Sassu I, Cossu S, Nieddu V, Galleri G, Azara E, Dettori MA, Fabbri D, Palmieri G, & Pisano M. Molecular changes induced by the curcumin analogue D6 in human melanoma cells. Molecular cancer. 2013;12(1): 37.
Roy M, Mukherjee S. Reversal of resistance towards cisplatin by curcumin in cervical cancer cells. Asian Pac J Cancer Prev. 2014;15(3):1403-10.
Sanchez Y, Simon G, Calvino E, de Blas E, & Aller P. Curcumin Stimulates Reactive Oxygen Species Production and Potentiates Apoptosis Induction by the Antitumor Drugs Arsenic Trioxide and Lonidamine in Human Myeloid Leukemia Cell Lines. Journal of Pharmacology and Experimental Therapeutics. 2010;335(1):114-123.
Shakibaei M, Buhrmann C, Kraehe P, Shayan P, Lueders C, & Goel A. Curcumin chemosensitizes 5-fluorouracil resistant MMR-deficient human colon cancer cells in high density cultures. PLoS One. 2014 Jan 3;9(1):e85397. doi: 10.1371/journal.pone.0085397. eCollection 2014.
Shao ZM, Shen ZZ, Liu CH, Sartippour MR, Go VL, Heber D, & Nguyen M. Curcumin exerts multiple suppressive effects on human breast carcinoma cells. International Journal of Cancer. 2002;98(2): 234-240.
Sharma RA, McLelland HR, Hill KA, Ireson CR, Euden SA, Manson MM, Pirmohamed M, Marnett LJ, Gescher AJ, & Steward WP. Pharmacodynamic and pharmacokinetic study of oral Curcuma extract in patients with colorectal cancer. Clin Cancer Res. 2001 Jul;7(7):1894-900.
Shishodia S. Curcumin: Getting Back to the Roots. Annals Of The New York Academy Of Sciences. 2005;1056(1):206-217. doi:10.1196/annals.1352.010
Sinha R, Anderson DE, McDonald SS, & Greenwald P. Cancer risk and diet in India. J Postgrad Med. 2003;49(3):222-228.
Sreekanth CN, Bava S V, Sreekumar E, & Anto RJ. Molecular evidences for the chemosensitizing efficacy of liposomal curcumin in paclitaxel chemotherapy in mouse models of cervical cancer. Oncogene. 2011;30(28): 3139–3152. doi:10.1038/onc.2011.23
Tharakan ST, Inamoto T, Sung B, Aggarwal BB, & Kamat AM. Curcumin potentiates the antitumor effects of gemcitabine in an orthotopic model of human bladder cancer through suppression of proliferative and angiogenic biomarkers. Biochemical pharmacology. 2010;79(2): 218-228. DOI: 10.1016/j.bcp.2009.08.007.
Tian B, Wang Z, Zhao Y, Wang D, Li Y, Ma L, Li X, Li J, Xiao N, Tian J, & Rodriguez R. Effects of curcumin on bladder cancer cells and development of urothelial tumors in a rat bladder carcinogenesis model. Cancer letters. 2008;264(2): 299-308.
Tian F, Fan T, Zhang Y, Jiang Y, & Zhang X. Curcumin potentiates the antitumor effects of 5-FU in treatment of esophageal squamous carcinoma cells through downregulating the activation of NF- B signaling pathway in vitro and in vivo. Acta Biochimica et Biophysica Sinica. 2012;44(10):847-855.
Toden S, Okugawa Y, Jascur T, Wodarz D, Komarova NL, Buhrmann C, Shakibaei M, Boland CR, & Goel A. Curcumin mediates chemosensitization to 5-fluorouracil through miRNA-induced suppression of epithelial-to-mesenchymal transition in chemoresistant colorectal cancer. Carcinogenesis. 2015 Mar;36(3):355-67. doi: 10.1093/carcin/bgv006. Epub 2015 Feb 4.
Vinod BS, Antony J, Nair HH, Puliyappadamba VT, Saikia M, Narayanan SS, Bevin A, & Anto RJ. Mechanistic evaluation of the signaling events regulating curcumin-mediated chemosensitization of breast cancer cells to 5-fluorouracil. Cell death & disease. 2013;4(2): e505.
Wang D, Veena M, Stevenson K, Tang C, Ho B, & Suh J. Liposome-Encapsulated Curcumin Suppresses Growth of Head and Neck Squamous Cell Carcinoma In vitro and in Xenografts through the Inhibition of Nuclear Factor B by an AKT-Independent Pathway. Clinical Cancer Research. 2008;14(19):6228-6236.
Wang H, Geng Q, Wang L, & Lu Y. Curcumin potentiates antitumor activity of l -asparaginase via inhibition of the AKT signaling pathway in acute lymphoblastic leukemia. Leukemia & Lymphoma. 2012;53(7):1376-1382.
Wang K, Fan H, Chen Q, Ma G, Zhu M, Zhang X, Zhang Y, & Yu J. Curcumin inhibits aerobic glycolysis and induces mitochondrial-mediated apoptosis through hexokinase II in human colorectal cancer cells in vitro. Anticancer Drugs. 2015 Jan;26(1):15-24. doi: 10.1097/CAD.0000000000000132.
Wang K, Wu X, Wang J, & Huang J. Cancer stem cell theory: therapeutic implications for nanomedicine. International journal of nanomedicine. 2013;8:899.
William B, Goodrich A, Peng C, & Li S. Curcumin inhibits proliferation and induces apoptosis of leukemic cells expressing wild-type or T315I-BCR-ABL and prolongs survival of mice with acute lymphoblastic leukemia. Hematology. 2008;13(6):333-343.
Yallapu MM, Ebeling MC, Khan S, Sundram V, Chauhan N, Gupta BK, Puumala SE, Jaggi M, & Chauhan SC. Novel curcumin-loaded magnetic nanoparticles for pancreatic cancer treatment. Mol Cancer Ther. 2013 Aug;12(8):1471-80. doi: 10.1158/1535-7163.MCT-12-1227. Epub 2013 May 23.
Yallapu MM, Othman SF, Curtis ET, Bauer NA, Chauhan N, Kumar D, Jaggi M, & Chauhan SC. Curcumin-loaded magnetic nanoparticles for breast cancer therapeutics and imaging applications. Int J Nanomedicine. 2012;7:1761-79. doi: 10.2147/IJN.S29290. Epub 2012 Apr 17.
Yan G, Graham K, & Lanza‐Jacoby S. Curcumin enhances the anticancer effects of trichostatin a in breast cancer cells. Molecular carcinogenesis. 2013;52(5):404-411.
Yen F, Wu T, Tzeng C, Lin L, & Lin C. Curcumin Nanoparticles Improve the Physicochemical Properties of Curcumin and Effectively Enhance Its Antioxidant and Antihepatoma Activities. Journal of Agricultural and Food Chemistry. 2010;58(12):7376-7382.
Yin H, Guo R, Xu Y, Zheng Y, Hou Z, Dai X, Zhang Z, Zheng D, Xu H. Synergistic antitumor efficiency of docetaxel and curcumin against lung cancer. Acta Biochimica et Biophysica Sinica. 2012;44(2):147–153. doi:10.1093/abbs/gmr106
Yin H, Zhang H, & Liu B. Superior anticancer efficacy of curcumin-loaded nanoparticles against lung cancer. Acta biochimica et biophysica Sinica. 2013;45(8): 634-640.
Yoysungnoen P. Anti-cancer and anti-angiogenic effects of curcumin and tetrahydrocurcumin on implanted hepatocellular carcinoma in nude mice. WJG. 2008;14(13):2003.
Yu J, Peng Y, Wu LC, Xie Z, Deng Y, Hughes T, He S, Mo X, Chiu M, Wang QE, He X, Liu S, Grever MR, Chan KK, & Liu Z. Curcumin down-regulates DNA methyltransferase 1 and plays an anti-leukemic role in acute myeloid leukemia. PloS one. 2013;8(2):e55934.
Yu LL, Wu JG, Dai N, Yu HG, & Si JM. Curcumin reverses chemoresistance of human gastric cancer cells by downregulating the NF-κB transcription factor. Oncology reports. 2011; 26(5):1197. DOI: 10.3892/or.2011.1410
Zhao X, Chen Q, Li Y, Tang H, Liu X et al. Codelivery of doxorubicin and curcumin with lipid nanoparticles results in improved efficacy of chemotherapy in liver cancer. IJN. 2014;10:257-70.
Zhou QM, Zhang H, Lu YY, Wang XF, & Su SB. Curcumin reduced the side effects of mitomycin C by inhibiting GRP58‐mediated DNA cross‐linking in MCF‐7 breast cancer xenografts. Cancer science. 2009;100(11):2040-2045.
Zhou QM, Wang XF, Liu XJ, Zhang H, Lu YY, Huang S, & Su SB. Curcumin improves MMC-based chemotherapy by simultaneously sensitising cancer cells to MMC and reducing MMC-associated side-effects. European Journal of Cancer. 2011;47(14): 2240-2247.
Zhu R, Wu X, Xiao Y, Gao B, Xie Q, Liu H, & Wang S. Synergetic Effect of SLN-Curcumin and LDH-5-Fu on SMMC-7721 Liver Cancer Cell Line. Cancer Biotherapy & Radiopharmaceuticals. 2013;28(8):579-587.
Zhu S, Moore T, Lin X, Morii N, Mancini A, & Howard R. Synthetic curcumin analog EF31 inhibits the growth of head and neck squamous cell carcinoma xenografts. Integrative Biology. 2012;4(6):633.
Zunino S. Oral or parenteral administration of curcumin does not prevent the growth of high-risk t(4;11) acute lymphoblastic leukemia cells engrafted into a NOD/SCID mouse model. International Journal of Oncology. 2013;42(2): 741-748.