Antiviral Paxlovid reduces rate of hospitalization and death among high-risk COVID-19 patients
Health Canada has approved Pfizer’s COVID-19 therapeutic for use in adults 18 and older, paving the way for the distribution of a potentially lifesaving drug at a time when the country’s hospitals are overwhelmed.
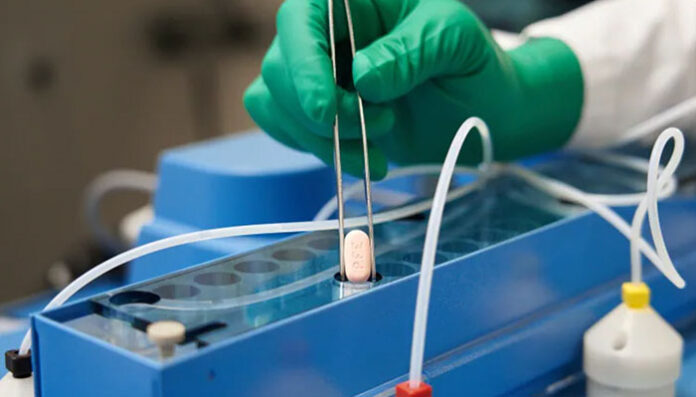
Pfizer’s Paxlovid, an oral antiviral prescribed by a doctor and administered in pill form, is designed to help the body fight off the SARS-CoV-2 virus, reduce symptoms from an infection and shorten the period of illness.
After months of clinical trials, Pfizer reported in November that Paxlovid reduced the risk of hospitalization or death by an impressive 89 per cent compared to a placebo in non-hospitalized high-risk adults with COVID-19.
- Have a coronavirus question or news tip for CBC News? Email: ask@cbc.ca or join us live in the comments now.
The drug company’s laboratory studies also indicate the drug is likely to work against the Omicron variant, now the dominant variant among new cases in Canada.
The product has been hailed as a pandemic “game changer” by some doctors because it could reduce hospitalizations and deaths among COVID-19 patients.
Experts say an effective pill that’s easy to self-administer at home could relieve some of the pressure on the health care system and change the trajectory of the pandemic. Existing therapeutics approved for use in Canada — products like monoclonal antibodies and remdesivir — must be administered intravenously in a hospital setting.
Speaking at a press conference with reporters Monday, Dr. Theresa Tam, Canada’s chief public health officer, said the regulator’s approval is “great news” because Paxlovid could drive down severe outcomes in the current wave and beyond.
“The regulator as well as the experts helping us with the guidance and the supply are all coming together at once and I think Canadians should be very happy today to hear that oral antivirals are beginning to become available in Canada,” she said.
Canada has placed an order for an initial quantity of one million treatment courses, with an option to buy up to 500,000 more. With global interest in antivirals running high as the Omicron variant wreaks havoc, Pfizer is promising to churn out 120 million courses of the treatment by year’s end.
Health Minister Jean-Yves Duclos said 30,000 treatment courses have arrived in Canada already and will be distributed to the provinces and territories on a per-capita basis.
Duclos said another 120,000 Paxlovid treatments will arrive between now and the end of March. The federal government is working with Pfizer to bring “additional treatment courses to Canada as quickly as possible,” he said.
Health Minister Jean-Yves Duclos says over 120,000 Paxlovid treatments are expected by March, in addition to the 30,000 already distributed. 1:02
While championing Paxlovid as a treatment that will “save lives, reduce illness and lighten the load on our health care system,” Duclos said this antiviral is not a replacement for vaccines, which remain the best way to keep people out of hospital.
“This is welcome news — we have one more tool in our toolbox. But no drug, including Paxlovid, can replace vaccination and public health measures,” he said. “You don’t want to have to use that pill if you can instead be vaccinated. Vaccination will be a lot better in protecting you.”
Tam said the Public Health Agency of Canada (PHAC) is working with its provincial and territorial counterparts to determine how best to distribute antivirals, which are expected to be in short supply for the foreseeable future.
“This treatment, the first treatment taken orally and at home, will be in high demand,” she said. “We anticipate supply at the beginning will not be great anywhere.”
The product, which doesn’t prevent infection, has been authorized by Health Canada for use in high-risk adults with mild or moderate COVID-19 symptoms.
A spokesperson for Ontario’s Health Minister Christine Elliott said the country’s largest province will earmark its share — about 10,000 courses of treatment to start — for adults with “the highest risk of severe outcomes, including immunocompromised patients.”
A person should start taking Paxlovid no more than five days after symptoms start, which could be “one of the key challenges of these antivirals,” Tam said.
‘An important tool’
“They have to be given really early. Not easy, but everybody needs to give it a good try because it could be an important tool going forward,” she said. “It could potentially blunt the severity of the virus, which is a key goal.”
Health Canada said Pfizer’s pills should only be used by patients who have tested positive on a SARS-CoV-2 viral test. Such tests are currently in short supply in some provinces and territories. If a PCR test is not available, Tam said a positive result on a rapid antigen test would also suffice.
Dr. Isaac Bogoch, an infectious disease specialist and researcher based at Toronto General Hospital, said Health Canada’s approval is “a very positive first step.”
“Everything we’ve heard about this pill is very promising but there are clearly logistical challenges ahead,” he said, adding that careful planning is required to make sure the pills get to those who need them most.
This drug regimen could be useful for people who have underlying conditions that increase the risk of hospitalization and death related to the coronavirus, such as heart disease or diabetes.
It could also be given to the unvaccinated, who are much more likely to experience severe outcomes. Tam pointed to PHAC data that suggest unvaccinated people are 19 times more likely to be hospitalized with COVID-19 than fully vaccinated people.
Health Canada has warned, however, that the product shouldn’t be used while a patient is on any of a long list of other drugs, including common medications used to treat erectile dysfunction, high cholesterol and seasonal allergies, among others.
“If you’re on certain medications, you have to be careful when using this drug,” Tam said, urging prescribers to review contraindications before writing a script for Paxlovid.
Pfizer’s treatment is meant to be taken as 30 pills over five days. Patients take three pills at a time: two of Pfizer’s pills and one of a low-dose HIV drug known as ritonavir, which helps Pfizer’s drug remain active in the body longer.
Sorce: www.cbc.ca