Nordic Naturals announced that to date their fish oils have been used in 14 published studies by independent research institutions such as Stanford University, Cedars- Sinai medical Center and Massachusetts General Hospital. The 14 published studies show a full range of application including support for cardiovascular health, behavior/mood, blood sugar, brain health, circulation, bronchial health and the natural anti-inflammatory response. Thirty additional studies using Nordic Naturals oils are currently underway. JoarOpheim, CEO and founder of Nordic Naturals noted that “many of these studies are funded by the National Institute of Health (NIH), which requires rigorous documentation for purity, potency, and stability of the oils used….Our fish oils exceed both medical and pharmaceutical standards with unsurpassed quality and assurance.” Abstracts for these and additional studies can be viewed at www. omega-research.com.
UAS Labs Featured in Finance Monthly
UAS Laboratories was featured in the January 2011 issue of Finance Monthly. In its “Feature Deals of the Month” department, the magazine noted that UAS Labs recent developments include winning a 2010 Frost & Sullivan Award for Customer Value Enhancement of the Year in North
American probiotics market and the acquisition of APN Labs, a nutraceutical contract manufacturer offering condition- specific probiotic blends. The company’s president, Dr. S.K. Dash, was quoted: “Significantly, UAS Labs ensures a strong scientific validation for its products through nearly 200 studies as individual, collaborative, and sponsored efforts between the industry and academia.”
Seroyal Sponsors 2010-11 Graduate Assistance Program (GAP)
Seroyal has announced its sponsorship of the 2010-11 Ontario Association of Naturopathic Doctors (OAND) Graduate Assistance Program (GAP). Designed to help support graduates of naturopathic medicine, GAP provides numerous product, business, marketing and insurance resources to help with the transition of setting up a naturopathic practice. In addition to the sponsorship, Seroyal also offers OAND and GAP members an unequalled continuing education program. Graduates and practitioners alike are able to learn from internationally-recognized experts in natural medicine.
Try this, come back, and we’ll see
Try this, come back, and we’ll see
Creating great treatment plans
Anew patient – let’s call her Joanna – recently mentioned that she’d been to another practitioner, but had decided to try her luck elsewhere. Her reason?
“She told me to ‘Try this, come back, and we’ll see.’”
At fi rst glance, this might not seem like much of a reason to abandon a doctor. After all, much of what is done in practice is… well, practice. Being 100% confi dent is a luxury we rarely have. “Try this and we’ll see,” simply isn’t that uncommon.
But what Joanna was upset about was not the lack of surety, or the possibility that her care was a bit of an experiment. She simply felt that there was no plan.
And she was right. Try this and we’ll see isn’t a treatment plan any more than one line on a page is a novel. Yet it’s surprisingly common.
Much of the reason stems from putting too much emphasis on treatments. e tools of our trades are deceptive – it’s easy to believe that what you do as a practitioner is about the needles or the herbs or the supplements. But an acupuncture treatment isn’t a plan. Nor is a homeopathic remedy or a supplement. ey’re treatments, not plans. And how they’re used is a prescription, not a plan.
Joanna had a prescription and it wasn’t enough. So what was she missing?
Why Plans Are Important Joanna was craving a plan. A way to understand her treatment in a larger time frame, and how it fi ts in with her symptoms and overall health. And she’s not alone. Patients love plans. Here’s why:
Plans Create Context
Joanna’s not an outlier. She might have been better at articulating her desire for a treatment plan, but she’s not alone in her desire to have one. Patients crave context for their treatment. It’s how they understand that they’re on a path from A to B, and how they manage the anxiety around their health and care.
Even if they’re terrible at making a plan for themselves, patients love when you make one.
They Help Results
The simple act of mapping out treatment seems to have a placebo eff ect of its own. It gives patients a sense that they’re in capable hands, on a journey with a clear route and destination.
Plans Demonstrate Progress
Care takes time. Not everything resolves overnight, particularly chronic complaints that are built over years. A plan provides the context for seeing the slow, steady progress that can be invisible at times.
They Help Create Successful Outcomes
Plans make for commitment, commitment helps compliance, compliance creates success, and success makes more of itself. Want better results? Make a better plan.
What a Good Plan Looks Like
From the patient perspective, a good plan is a clear map from where the patient is to where she wants to be. It’s a health care road trip, with a clear destination, a route and waypoints along the way.
Here are the key high points your plan should hit:
Goals
What are you trying to accomplish? Are you sure you know what success looks like? Better still: does your patient know? Ask them what success looks like for them and make sure you’re on the same page.
Measurement
How will you know when you get to success? What can you measure that will let you know when you’ve arrived, and whether or not you’re headed in the right direction? Objective testing isn’t just a diagnostic tool for doctors – it’s a marker for progress that is hugely benefi cial for patients.
Visit Frequency
Return visits matter, and for more than just treatment – they’re how you track the progress of the plan. But visit structure and rationale are particularly important to clarify for consult-based care.
It’s easy for patients to know why they should come back for their next acupuncture treatment: to get the treatment. But the importance of a follow-up consult may need more clarity. Clearly state the visit structure, and the why behind it.
Tools and Techniques
What are you using to get from where you are now, to where you want to be? is is the area we tend to be best at, but have you explained to your patient why you’re using the modalities you are?
Timelines
What are the timelines? How long until your patient will see results? How long until she reaches success?
Alternatives
What are the options if the proposed plan doesn’t seem to be working? At what point do you consider them? Why are they alternatives, and not part of the active plan?
Even the Best-Laid Plans
Planning is surprisingly easy, but also surprisingly prone to three key pitfalls.
1. Believing ere’s not Enough Time to Plan
It’s easy to think that describing a treatment plan this way is too complicated to work through in an already-crowded visit.
But don’t be fooled. Joanna’s plan, properly laid out, takes just a few moments to communicate. Not a huge investment given the potential return.
The following example might take two, perhaps three, minutes to clearly explain to a patient:
I suspect the root of your problem is a food intolerance. It’s creating a chronic infl ammatory response in your body and your digestive track. at result is a magnesium defi ciency, and that’s what’s causing your migraines.
We’re going to remove your reactive foods from your diet, and see you once a we ek for the next 4-8 weeks for IV magnesium to treat the headaches. If the treatment works, we’ll decrease to once a month treatments after that for a year.
If we’re on the right track, you’ll notice a diff erence within the next 4 weeks, but it’s likely going to take 10-12 months for you to fully recover.
CRP, which is a measure of infl ammation, is elevated in your blood. We’ll measure this every 3 months to make sure it’s decreasing. We’ll follow up every 3 months to discuss the lab result, monitor your symptoms and tweak your treatment plan as needed.
2. Not Sharing the Plan e most common planning pitfall among doctors is that they already think they have a plan. And to be fair, you probably do.
You just might have neglected to tell your patient.
It’s easy to forget that your knowledge and experience make most aspects of the treatment plan seem obvious. So obvious that you don’t need to mention them. Remember, though, that your patient is a) not an expert and b) possibly overwhelmed with everything else you’ve told them during their visit. ey likely can’t create the plan themselves. at’s why they’ve come to you.
3. Too Much Information
Don’t confuse plans with information. A binder full of recipes, handouts and information sheets isn’t a plan – it’s just more information, and if anything it makes it harder for patients to understand and embrace the larger context of their care.
Plans are about broad strokes. ey’re about how to get from Toronto to Vancouver, not about tire pressure or the price of gas. Err on the side of over-simplifi cation. State the obvious, briefl y and clearly. en state it again if you need to.
Plans Help Everyone
A great plan is good for you and your patients. It leads to higher success rates, greater patient satisfaction, and as a result, more referrals.
Used properly, great treatment plans can help you fi nd that unique junction where benefi ts to patient, practitioner and practice overlap – the sweet spot of professional success.
Losing patients? Try better planning. Need more referrals? Try better planning. Want to feel more confi dent about your professional skills? You guessed it…better planning.
Cognitive Behavioural Therapy
Cognitive Behavioural Therapy
Application in clinical practice
Most health practitioners are aware of cognitivebehavioural therapy (CBT). CBT is one of the most studied and most relevant treatments in psychotherapy, with very positive research outcomes. It is short term, focused in the present, and evidence based. Cognitive-behavioural techniques can be quite specifi c but they are also highly adaptable, making them an important component of many diff erent treatment approaches and applicable in a variety of conditions. CBT is well established as a core treatment for many psychiatric disorders (Butler 2006); however, the usefulness of CBT is not limited to mental health concerns. CBT has been shown to reduce distress and improve outcomes related to a number of medical conditions, including chronic fatigue (Malouff 2007), type 2 diabetes (Gonzalez 2009), HIV (Safren 2009), and cancer (Osborn 2006). Cognitivebehavioural therapies can also be applied to help a healthy person optimize their wellbeing. Using CBT to identify and challenge the specifi c belief systems that perpetuate disturbance can help to both shift physiologic experience and promote self-empowerment. ese goals are inherently a part of our naturopathic philosophy and an expression of the mind-body connection.
The CBT Story:
A psychiatrist named Aaron Beck, MD first presented CBT as a specific treatment for depression in the early 1960s. Trained as a psychoanalyst, Dr. Beck began noticing how particular streams of negative thought became active during therapy with depressed patients. Recognizing that these thoughts were not consciously derived or acknowledged, Dr. Beck termed them “automatic negative thoughts.” Dr. Beck also noticed that automatic negative thoughts tended to relate to unconscious beliefs in one of three areas: negative beliefs about oneself, negative beliefs about the future, and negative beliefs about the world. Rather than working through these streams from a strictly psychoanalytic perspective, Dr. Beck began developing tools to help patients directly identify and evaluate the content of their thoughts. By bringing conscious awareness to these automatic and unconscious beliefs, Dr. Beck was able to help his patients begin to think more realistically. As patients began to consciously shift their thinking patterns, they began to feel better emotionally and were able to behave in more effective ways.
From these beginnings, an evidence base spanning 50 years of research has challenged CBT to grow as a treatment modality. CBT has been specifically studied as a treatment for depression and other mood disorders, anxiety disorders, eating disorders, addictions, recovery from trauma, and in the treatment of many medical conditions (for detailed list see Beck Institute 2011). Current research continues to examine the efficacy of CBT in a variety of mental health conditions (Hofmann 2008, McCarthy 2007, Roberts 2009) as well as its role in mindfulness-based therapy (Hofmann 2010) and healthy psychological adjustment in non-clinical populations. With such a rich research history behind it, contemporary data on CBT has become increasingly refined. For example, Cape and colleagues (2010) evaluate the use of a brief-form CBT in primary care as a treatment for anxiety, depression, and mixed mood disorders. Hart and Hart (2010) consider the values and limitations of CBT in a behavioural medicine context. Another interesting perspective (Spek 2007) explores the use of internetbased CBT and alternate delivery forms that make CBT tools much more widely accessible. Although the literature will continue to evaluate and critique the clinical parameters of CBT, its value as a psychotherapeutic tool is evident.
Next Steps: With an abundance of data to draw from and clear indications for the use of cognitive-behavioural therapies, the next question is how a naturopathic doctor can utilize the resources provided by a CBT model. Even without specific training in CBT, there are practical ways that an ND can apply cognitive-behavioural principles within a naturopathic context. While cognitivebehavioural methods may be of particular interest to those who treat patients with anxiety and depression, it is important to remember that these tools are much more broadly applicable. If we consider maladaptive belief systems as an obstruction to Vis, then we can begin to understand how beneficial cognitivebehavioural restructuring can be with regards to overall health. Recent advances in the science of epigenetics demonstrate how deeply organizing our perceptual systems are with regards to our physiology. In his work on the biology of belief (Lipton 2005), Dr. Bruce Lipton, PhD, explains how perception mediates cellular behaviour and biological expression. Our core beliefs and perceptual habits exert a significant influence on cellular behaviour and are strong epigenetic regulators. Informed by this perspective, we can begin to harness the restorative biological power that comes with increased freedom from cognitive distortions and limiting beliefs.
There is not just one way to use CBT in a naturopathic practice. For people who prefer a more structured and linear approach, there are some great resources on the market that will take you through the process step by step. For practitioners who would like to integrate a more general use of cognitive-behavioural therapy within a naturopathic framework, I will offer some suggestions as to what this might look like. The most important components of any cognitive-behavioural therapy include the capacity for self-reflective awareness, learning to identify specific thought patterns, and restructuring maladaptive belief systems. NDs can implement a cognitive-behavioural approach to health by helping patients to first become more aware of their thoughts and then learn to observe them without reacting to them. The goal here is to identify specific words, expectations, and assumptions that have a negative influence on a person’s wellbeing and to understand the context in which those thoughts become active.
Begin by noticing if there are certain words that a patient frequently uses to describe themselves, their life situation, or their relationships. What words have adverse energy for that person and in what context? Do the automatic negative thoughts tend to be active in all domains of life or are they more prevalent with regards to the way a person views a specific aspect of their life? (eg: self-image, home, work, relationships, family, health, finances, etc). What automatic assumptions are attached to those thoughts? How do those thoughts impact the person’s emotional state? Their body sensations? Their relationships? Their behaviours? By consciously identifying automatic negative thoughts and describing their impact on other aspects of experience, patients are able to begin separating themselves from their thinking and can better see the connections between thoughts, feelings, and behaviours.
As you teach patients to become more aware of their own thought patterns, you can actively challenge negative self-talk within an appointment by reflecting negative thought streams back to patients and asking them to consider the validity of that thought (“I hear you say quite often that you’ll never be healthy; do you believe that is true?” or “I’ve noticed that you tend to put yourself down when you are talking about your family; do you believe that you are less worthy of love than your siblings?”). Once you have identified a problematic thought, you can challenge the patient to discover whether or not that thought is true. Although it may feel 100% true, it often is not. Engage the patient by asking them to give evidence for the truth of a particular thought. Then ask them if there is any evidence that the thought is not completely true. You can expand on this by asking them to consider at least one alternate perspective (“How else might you explain this? What else could it mean?”). This kind of therapeutic conversation challenges patients to begin questioning and changing their cognitive habits.
Thought records are another way to help patients develop self-reflective awareness and learn to identify cognitive patterns. Patients are asked to track their thoughts, writing them down each time they become aware of them throughout the day. There are many resources available for learning to keep thought records, and essentially they all teach people to notice and evaluate their self-talk. Many thought records also include instructions on how to identify and label automatic negative thoughts (for example, is this thought a catastrophizing thought? A “should” thought? An all-or-nothing thought?), as well as how to create a habit of thinking more realistically (eg: changing “I’m never good at anything” to “even though I didn’t do as well as I would have liked, I still passed the exam”). Teaching people to become aware of and challenge their thinking can be an empowering step towards change.
Once a person has become more conscious of their self-talk, it is usually fairly simple to see that these automatic negative thoughts cluster around certain themes. As themes begin to appear within the automatic thought patterns, you can track them to underlying core beliefs. For example, automatic negative thoughts could cluster around themes of mistrust, autonomy, shame and guilt, competence, inferiority, perfection, intimacy, protection, or meaning. While there are limitless possibilities as to how these themes express themselves, they typically relate to the ways we view ourselves, the world around us, and our future potentials. Often they have to do with perceptions related to one’s worth as a person, the ability to give/receive love, the expression of power/control, and the desire for acceptance. As we explore the belief systems that motivate our feelings and behaviours, we start to identify perceptual distortions that limit our experience of the world. In homeopathic terms, these can be considered “delusions” and those with a homeopathic mindset will find that they can prescribe on the experience of these themes. The same idea applies to TCM: as you clarify the energy of the belief system you can correlate it with the five elements or mental-emotional patterns of specific organ imbalances and treat accordingly. From this perspective, cognitivebehavioural approaches are not only independent therapeutic tools but also provide a level of understanding that is helpful when applying other treatment modalities. Prescribing from this level allows for an expression of “treat root cause.” What we are trying to do is isolate belief systems that perpetuate disturbance in physiology, psychology, and behaviour. In other words, the belief system itself is pathogenic and once we understand its role, we can apply any of our naturopathic modalities to help correct it.
Belief systems create the perceptual configurations for the experience of life. As these belief systems become more visible, a range of options opens up to you and you can actively engage in a restructuring process. You can recognize the origins of some beliefs (“my father told me that winning is the only thing that matters”) and with compassion begin to let them go. You will find that some beliefs shift easily and others are much more entrenched. Sometimes it helps to directly challenge the truth of these belief systems and evaluate the data that supports/refutes them, and at other times it is better to just question whether or not the belief is helpful in the present. A good question to ask is, “how does this belief contribute to my health, happiness, and wellbeing?” If the answer is, “it doesn’t” then ask the patient what holds the belief in place, what sustains its presence in their psyche. Evaluate the justifications for keeping the belief and consider the possibilities for releasing it. “Who would I be without this belief” is another good question for discussion.
It is important to remember that cognitive-behavioural tools are most efficacious within a comprehensive and individualized approach to treatment. Some patients may just need direction to available resources and encouragement towards self-study; others may appreciate a workbook-style approach where you work as a team. Some patients may respond well when you engage in therapeutic conversation around a belief, allowing them to recognize it for themselves and encouraging them to examine the patterns in place around that belief in their lives. Some patients will enjoy a cognitively-oriented approach to working with belief systems but others may benefit from focusing more on the emotions, images, or sensations associated with the belief. Other people will do best when they are behaviourally-oriented. Understanding your patient as an individual keeps you true to the philosophy of naturopathic medicine and it also allows you to customize treatment so that it is as effective as possible. Whether you are drawn to a more cognitive approach to CBT or if you can better relate to cognitive work through another form like homeopathy, guided imagery, TCM, or body-based therapies, there are many ways that our profession can serve others through responsible use of functional psychotherapy.
References:
Beck Institute. www.beckinstitute.org Accessed January 2011.
Butler AC, Chapman JE, Forman EM, Beck AT. The empirical status of cognitive-behavioral therapy: a review of meta-analyses. Clin Psychol Rev. 2006 Jan;26(1):17-31.
Cape J, Whittington C, Buszewicz M, Wallace P, Underwood L. Brief psychological therapies for anxiety and depression in primary care: meta-analysis and meta-regression. BMC Med. 2010 Jun 25;8:38.
Gonzalez JS, McCarl LA, Wexler DJ, Cagliero E, Delahanty L, Soper TD, Goldman V, Knauz R, Safren SA. Cognitive behavioral therapy for adherence and depression (CBT-AD) in type 2 diabetes. Journal of Cognitive Psychotherapy. 2010;24(4):329-343.
Hart SL, Hart TA. The future of cognitive behavioral interventions within behavioral medicine. Journal of Cognitive Psychotherapy. 2010;24(4):344-353.
Hofmann SG, Sawyer AT, Witt AA, Oh D. The effect of mindfulness-based therapy on anxiety and depression: A meta-analytic review. J Consult Clin Psychol. 2010 Apr;78(2):169-83.
Hofmann SG, Smits JA. Cognitive-behavioral therapy for adult anxiety disorders: a meta-analysis of randomized placebo-controlled trials. J Clin Psychiatry. 2008 Apr;69(4):621-32.
Lipton, Bruce. The Biology of Belief. 2005. Malouff JM, Thorsteinsson EB, Rooke SE, Bhullar N, Schutte NS. Efficacy of cognitive behavioral therapy for chronic fatigue syndrome: a meta-analysis. Clin Psychol Rev. 2008 Jun;28(5):736-45.
McCarty CA, Weisz JR. Effects of psychotherapy for depression in children and adolescents: what we can (and can’t) learn from meta-analysis and component profiling. J Am Acad Child Adolesc Psychiatry. 2007 Jul;46(7):879-86.
Osborn RL, Demoncada AC, Feuerstein M. Psychosocial interventions for depression, anxiety, and quality of life in cancer survivors: meta-analyses. Int J Psychiatry Med. 2006;36(1):13-34.
Roberts NP, Kitchiner NJ, Kenardy J, Bisson JI. Systematic review and meta-analysis of multiplesession early interventions following traumatic events. Am J Psychiatry. 2009 Mar;166(3):293- 301.
Safren SA, O’Cleirigh C, Tan JY, Raminani SR, Reilly LC, Otto MW, Mayer KH. A randomized controlled trial of cognitive behavioral therapy for adherence and depression (CBT-AD) in HIV-infected individuals. Health Psychol. 2009 Jan;28(1):1-10.
Spek V, Cuijpers P, Nyklicek I, Riper H, Keyzer J, Pop V. Internet-based cognitive behaviour therapy for symptoms of depression and anxiety: a meta-analysis. Psychol Med. 2007 Mar;37(3):319-28.
James Excellence in Integrative Psychiatry
James Excellence in Integrative Psychiatry
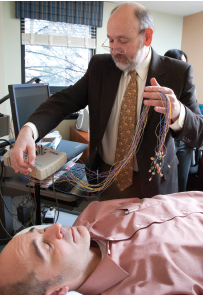
I arrived in medical school thinking brown rice and yoga was a cure for everything” states Dr James Greenblatt, a Psychiatrist practicing in the Waltham, Massachusetts (greater Boston) area. He received his license to practice adult psychiatry from George Washington University in 1990, and followed this with training in child psychiatry from John Hopkins. His interest in integrative medicine began early in his career, as he describes a seminar on “the evolution of the biomedical model” in 1988 as instrumental in the manner in which he approaches the practice of psychiatry today. Dr. Greenblatt’s commitment to integrative medicine for mental health is seen in his private outpatient practice (named Comprehensive Psychiatric Resources), and in his many lectures and seminars given throughout the US and Canada.
Dr Greenblatt exemplifies the true essence of an integrative practitioner. The formulary Dr Greenblatt calls upon is a welldefined selection of essential vitamins and minerals, functional food/ nutraceutical agents, and a selection of botanical medicines. He also relies upon a selection of integrative diagnostic tests to help individualize treatments for patients. Commonly employed integrative tests include the urinary organic acids profile, fasting plasma amino acid quantification, an opiate peptides test (looking for gliadorphin and/ or casomorphin), provocative heavy metal testing, food IgG sensitivity panels, and evaluation of essential fatty acid status.

 A new patient visit involves a one- hour interview with a trained social worker. A thorough history is gathered, including a genogram to track family history of illness. A thorough battery of routine lab analysis is ordered, which includes basic blood chemistry, methyl malonic acid, homocysteine and free B12 assessment as markers of B12 status, iron panel, plasma vitamin D, DHEA, testosterone, and assessment of thyroid function.
A new patient visit involves a one- hour interview with a trained social worker. A thorough history is gathered, including a genogram to track family history of illness. A thorough battery of routine lab analysis is ordered, which includes basic blood chemistry, methyl malonic acid, homocysteine and free B12 assessment as markers of B12 status, iron panel, plasma vitamin D, DHEA, testosterone, and assessment of thyroid function.
Dr Greenblatt has also championed the use of an assessment tool known as referenced EEG. The tool correlates comparative EEG data and provides a novel neuroinformatic view to medication sensitivity. Over 100 peer reviewed publications of the methodology exist, with a recent paper in the Journal of Psychiatric Research demonstrating that when the tool was used to select a prescription medication for a patient with depression, the response rate was dramatically improved. Given the generally poor response rates to prescription medications used for management of depression and other psychiatric illnesses, the tool provides a valuable strategy in improving patient compliance and ultimately patient outcomes. Patients should be medication free when undergoing the assessment, and thus for patients already medicated wishing to try the technology, supervised discontinuation of the medication is imposed. Dr Greenblatt and colleagues certainly use prescription medications with their patients when required, but the use of referenced EEG has made application of prescription medications infinitely more successful.
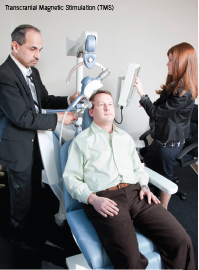
The Comprehensive Psychiatric Resources team has recently introduced TMS as a treatment option for patients. Transcranial Magnetic Stimulation (TMS) therapy uses a highly focused pulsed magnetic field to stimulate nerve cells in the area of the brain thought to control mood. Patients undergo five sessions per week for four to six weeks, with each session taking approximately 40 minutes. TMS has received FDA approval for patients suffering from depression who have not achieved satisfactory improvement from prior antidepressant medications.
Dr. Greenblatt is outraged and dismayed by the polypharmacy strategy that dominates mainstream psychiatric practice. Patients arrive at his treatment facility on as many as 12 or 13 psychoactive medications. “It’s an embarrassment to the fi eld of psychiatry”, states Greenblatt. e implementation of referenced EEG in-andof- itself has gone a long way to correcting the polypharmacy phenomena for the patients he manages.
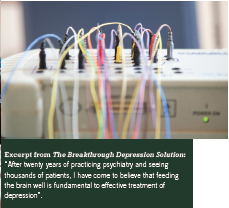
The philosophy that governs Dr Greenblatt’s approach to management of the psychiatric patient is one familiar to all practitioners of integrative medicine. Dr Greenblatt describes optimal nutritional support and correcting of nutrient defi ciencies as the fi rst steps to recovery. He has published two books addressing integrative psychiatry, one entitled “Answers to Anorexia” and the other entitled “ e Breakthrough Depression Solution”. He describes the concept of biochemical individuality, citing that the majority of known polymorphisms relate to some aspect of how an individual processes a particular nutrient. e book on anorexia makes mention of “feeding the starving brain”, a notion that has since become the title of a book for strategies in autism management (Children with Starving Brains by Jacquelyn McCandless, MD).
The Comprehensive Psychiatric Resources team is in the process of moving its head offi ces. “ e Watch Factory”, a historic facility built by the Waltham Watch Company in the 1860’s, will be the new home for the clinic beginning this May. Dr. Greenblatt’s desire to relocate to this historic location parallels a quote attributable to Royal Lee, founder of the supplement manufacturer Standard Process, “it takes over 250 parts to make a watch…which component is responsible for telling time?” Dr. Greenblatt likens this to the practice of integrative psychiatry and the importance of whole food supplementation. e concept of physician as detective, sifting through medical history and laboratory analysis to attempt to discover the selection of malfunctioning “gears” responsible for a particular patient’s presenting concerns.
Psychiatry is a discipline desperately requiring more widespread application of integrative techniques. Conventional therapeutics are outright failing. Several recent meta analytic reviews in JAMA and other leading medical journals are calling for reappraisals of current guidelines specifi cally in the area of depression and other aff ective disorders. Integrative strategies prove themselves highly effi cacious, even though remaining highly underutilized. As the community of integrative healthcare providers learns of the techniques of Dr Greenblatt and others, their pioneering eff orts will invariably lead to access to these safe and eff ective techniques across the continent and beyond.
Garrett Swetlikoff, ND
Garrett Swetlikoff, ND
Kelowna Naturopathic Clinic
Garrett Swetlikoff, ND, is a pioneer of the naturopathic profession in Canada. Among the first licensed ND’s in British Columbia, Garrett has become renown as an individual who likes to push the profession to its deserved heights, blazing a path that hundreds of others have had the good fortune to follow. This passion for progress is identified in many areas of Garrett’s life and practice. Politically, Garrett has worked tirelessly to help secure scope of practice recognition as well as the prescription rights ND’s in British Columbia are presently enjoying. He served as the President of the BCNA between 1999 and 2007, and also served on the Pharmacy Committee of British Columbia, the Scope of Practice Committee, and acted as a liaison with the British Columbia colleges of acupuncture and TCM. Clinically, Garrett pushes the envelop of therapeutics to deliver safe, effective, and often novel therapies to his patients.
 Garrett has coined a phrase that helps create an important differentiation in systems of naturopathic practice; “interventional natural medicine”. While an eclectic, whole- body approach, which he too employs, works for many presenting patient concerns, Garrett felt this traditional naturopathic model often failed to deliver prompt and effective treatment for acute, progressed pathologies. The more traditional naturopathic strategies may be effective for long- term management of these presentations, but a more robust, directed, and specific style of intervention is required for quick and clinically important symptom management of several common acute pathologies.
Garrett has coined a phrase that helps create an important differentiation in systems of naturopathic practice; “interventional natural medicine”. While an eclectic, whole- body approach, which he too employs, works for many presenting patient concerns, Garrett felt this traditional naturopathic model often failed to deliver prompt and effective treatment for acute, progressed pathologies. The more traditional naturopathic strategies may be effective for long- term management of these presentations, but a more robust, directed, and specific style of intervention is required for quick and clinically important symptom management of several common acute pathologies.
While the Kelowna Naturopathic Clinic has successfully utilized traditional eclectic strategies, it is the interventional techniques that have established a unique reputation in the community and surrounding areas. Garrett believes IV and injection therapeutics are the tools of choice for application of interventional medicine. His facility employs all of the following; prolotherapy, ozone/photo oxidation, hydrogen peroxide infusions, Myer’s cocktails, amino acid injections, etc… Garrett describes an 18-month internship with Dr Craig Wagstaff, where he learned early principles of neural therapy (injection of anesthetic and other agents into scars, tendons, ligaments, nerve ganglia in an effort to reset the autonomic nervous system) as a key moment in developing his interest in application of injection therapies. As such, the facility has built a reputation as a centre for such treatments, resulting in secondary referrals from other ND’s wishing to have the strategies employed on their patients.
The clinic remains true to naturopathic roots of eclectic assessment and treatment strategies. The goal of interventional strategies is often to transition patients into more long- term prevention models of care. The clinic has developed an impressive offering of integrative assessment tools. Most impressively, Garrett offers the Cardiovision assessment tool, which allows for assessment of flow mediated dilatation of the brachial artery. This strategy has been the focus of much recent research attention, and was applied by investigators during the construction of the Framingham algorithm. The clinic also offers assessment of heart rate variability, blood viscosity/ platelet aggregation, food IgG/ IgA sensitivity panels, electrodermal screening, complete stool analysis, breast and whole body thermography, redox, PH and resistivity of blood, urine, and saliva, and a wide array of standard blood tests and tumour markers.
The clinic has also employed a unique strategy in terms of dispensary management. Often, from a practice management perspective, clinicians are encouraged to limit the number of companies they order products from. Garrett has adopted quite an opposite approach, and the basis is obvious and simple; while each company may have several hundred product offerings, there are typically three to five products that distinguish the company from others. A handful of key products deliver phenomenal efficacy, and a massive product line ends up being built around this small selection of truly novel and effective offerings. Garrett chooses to identify each company’s unique and important offerings, and carries these. As such, the Kelowna Naturopathic Clinic dispensary carries offerings from over 80 different companies! The strategy may cause an increase in administrative duties, but the rewards grossly outweigh the inconvenience.
The naturopathic profession has been on a path of exponential growth for several years, possibly decades. Often we hear mentors of the profession questioning the strategies employed by the newer graduates. Some of the professions elders have expressed a concern over loss of traditional systems of practice. Garrett provided a fresh and reassuring view on the topic. Garrett feels that new graduates are better trained, better prepared, and simply more ready than graduates of the profession have ever been. He feels that as a result of this better training, the profession is reaching a critical mass, poised to position itself in a manner long overdue. The basis for such a view is becoming obvious. The thought of naturopathic doctors performing rotations in hospitals, partnering with medical doctors, brought on as part of the healthcare team of professional sport franchises, seemed like a far off dream in the not too distant past. Today, ND’s are filling such positions routinely.
What Garrett fails to highlight is his own contribution to this continually improving system of practice and education. Through the pioneering efforts of Garrett and others, safe and effective therapies have trickled down to the ranks of colleges. Students are leaving their ND training with knowledge that pioneers spent decades to compile. We at IHP agree that the profession has reached its critical mass, and the result is naturopathic appointment to overdue and deserved positions. Moving forward, the phenomena can only grow. The most valuable asset to solidifying the reputation of the profession is to blanket the country with practitioners who deliver safe and effective therapies. Garrett began this task over two decades ago, and set the bar very high.
A cetyl-L-Carnitine
A cetyl-L-Carnitine
Clinical application as a neuroprotective agent
Introduction
As an essential regulator of mitochondrial function and fatty acid metabolism, L-carnitine is relatively well known for its ability to improve muscle function in conditions such as cardiac ischemia, and is often used in the treatment of other muscular conditions such as fibromyalgia. The ability of carnitine to impact neurological function, however, is perhaps less well known. Surprisingly, there is a fairly well developed corpus around the use of acetyl-L-carnitine (ALC) in patients with neurological conditions ranging from age related dementia, substance withdrawal, depression, Down syndrome, attention deficient hyperactivity disorder, neuropathy, and encephalopathy secondary to liver failure (Arnold 2007, Janiri 2009, Malaguarnera 2008, Montgomery 2003, Pueschel 2006, Youle 2007, Zanardi 2006). This paper reviews the neuroprotective effects of ALC, with a narrowed focus on its effects in Alzheimer’s disease (AD), substance withdrawal, and toxic peripheral neuropathies.
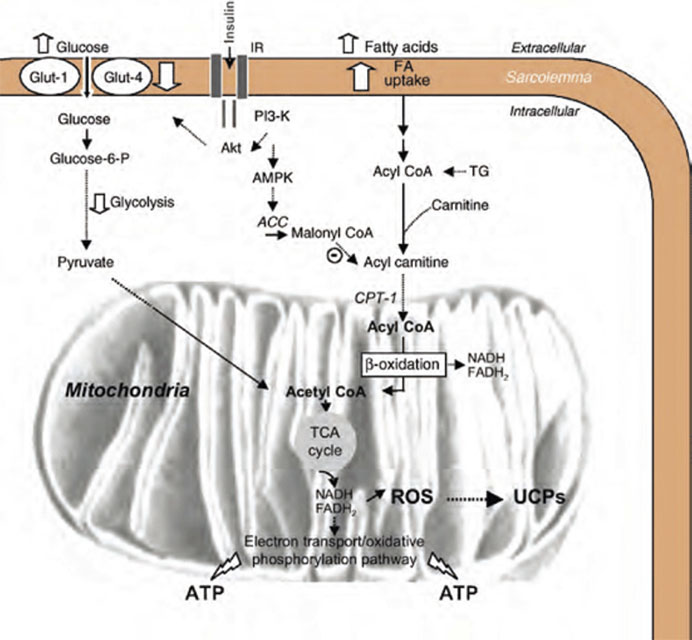
Physiology
As reviewed previously, carnitine is a key factor in the transport of fatty acids between the cytoplasm and the mitochondria, facilitating production of ATP, and appears to be in especially high demand under ischemic conditions. Acetyl-L-carnitine passes the blood-brain barrier through an active transport process and is highly concentrated in the brain, especially in the hypothalamus (Thal 2000, Virmani 2004), and is therefore the main focus of carnitine research in the area of neurological disease.
ALC exerts its neuroprotective effect through a variety of mechanisms. According to Quatraro, ALC has analgesic effects by acutely increasing blood levels of the endogenous opioid peptide beta-endorphin, neurotropic effects by upregulating nerve growth factor receptors on the brain and preventing accumulation of lipofuscin, and metabolic effects by increasing the oxidative metabolism of neurons (Calvani 1992, Quatraro 1995). Pettegrew used magnetic resonance spectroscopy to show that administration of ALC normalized brain levels of high energy phosphates in patients with AD compared to healthy controls (1995). ALC also has antioxidant effects, increasing glutathione and decreasing malondialdehyde (Thal 2000). Finally, acetyl-L-carnitine is thought to have cholinergic effects through facilitation of intracellular acetylcholine synthesis; carnitine transports acetyl groups out of the mitochondra into the cytoplasm where they can be used for ACh production (Thal 2000, Virmani 2004).
Clinical Evidence: Cognitive Impairment and/ or Alzheieimer’s DiDisease
ALC has been shown to improve cognitive function in elderly patients without dementia. Two randomized double blind placebo controlled trials showed ALC to improve mental fatigue in patients over 70 and over 100 years of age, respectively, along with concomitant improvements in physical function (Malaguarnera 2008, 2007). A third trial reported significant improvements in cognitive function, memory, emotional affect, and mood when evaluated in 481 elderly patients without AD (Salvioli 1994).
Two meta analyses have evaluated the effect of ALC for use in AD and/ or mild cognitive impairment (MCI) (Hudson 2003, Montgomery 2003). Montgomery included 21 randomized double blind placebo controlled trials assessing ALC for AD or MCI and concluded that ACL should be considered for the treatment of these conditions (2003). Doses ranged from 1.5 to 3.0 g/d for between 3 to 12 months. A combined effect size was calculated that incorporated both clinical and psychometric outcomes from the trials. There was significant benefit from treatment with ALC compared to placebo, effect size ES 0.201 (95% CI 0.107-0.295), and there was also a significant effect in the pooled Clinical Global Impression of Change, ES 0.32 (95% CI 0.18-0.47). ALC was well tolerated (Montgomery 2003).
The Cochrane review updated in 2008 included 16 randomized double blind placebo controlled trials (Hudson 2003). By contrast, this analysis found that while there was a statistically significant treatment effect in favour of ALC on the Clinical Global Impression at 12 and 24 wk (OR 1.90, 95% CI 1.31-2.76 and OR 2.33, 95% CI 1.31-4.14 respectively) this was not sustained at 52 weeks (OR 0.91, 95% CI 0.58-1.43). Similar effects were seen in pooled results for the Mini Mental State Exam (MMSE), leading authors to conclude that ALC may be of limited benefit.
This review excluded several studies that were included by Mongomery on the basis of poor reporting and/ or trial design. These trials were largely earlier trials out of Italy, so poor reporting standards may be to blame. In any case, the consequent discrepancy in included studies partially explains the divergent conclusions reached by the two studies. In addition, Mongomery utilized a unique composite effect measure that may not be directly comparable to those used by Hudson by pooling data across different assessment scales. However, it seems that the possibility of ALC benefiting at least a subset of patients with Alzheimer’s or MCI should not be ruled out. Hudson explains that the mechanisms by which ALC is absorbed and metabolized in the gut and liver “suggest that a large inter-individual variability should be expected in the general population. Studies have not taken this into account in selecting the dose of ALC, and this may be a source of error” (2003). Select trials of ALC for cognitive impairment are summarized in Table 1.

One uncontrolled trial found that the addition of ALC to the treatment of acetylcholinesterase inhibitor resistant patients with AD increased the response rate from 38% to 50% (p value not given) (Bianchetti 2003). ALC may therefore be useful for augmenting the effect of other therapies, even when it is not sufficient as a stand alone intervention.
Substance Withdrawal
A handful of trials have examined the potential effectiveness of ALC in ameliorating the symptoms of withdrawal from substances including methadone, cocaine, and alcohol. Janiri 2009 found that oral ALC 2g/d significantly decreased symptoms of methadone (opiate) withdrawal compared to placebo in 30 patients undergoing a three week detoxification program. Total symptom scores during the first five days were significantly lower in the ALC group (p<0.05), and in particular the following symptoms were improved: muscle tension and muscle spasm (p<0.05), insomnia (p<0.005), and feelings of coldness (p<0.05). Pain as assessed by the Huskisson analog scale was also “considerably lower” in the ALC group from after week one until the end of the study.
One study found no impact on symptoms of cocaine withdrawal or drug cravings (Reid 2005), while two RCTs reported significant improvements in cognitive impairment, mood, and anhedonia associated with chronic alcohol abuse in abstinent patients (Martinotti 2011, Tempesta 1990).
Peripheral Neuropathy
ALC appears to protect nerves from the effects of toxic drugs such as antiretroviral therapy and chemotherapy, and from the damaging effects of diabetes. One RCT and three open trials have reported improvements in pain and/ or symptom scores from administration of ALC in patients with antiretroviral toxic neuropathy (Hart 2004, Osio 2006, Scarpini 1997, Youle 2007). Two open trials have found reductions in neuropathy grade and/ or symptomatic improvements in patients with neuropathy of chemotherapy (Bianchi 2005, Maestri 2005). Four RCTs and one open trial reported improvements in pain and/ or neuropathy grade when ALC was given to patients with diabetes (De Grandis 2002, Quatraro 1995, Sima 2005). These studies are summarized in Table 2.
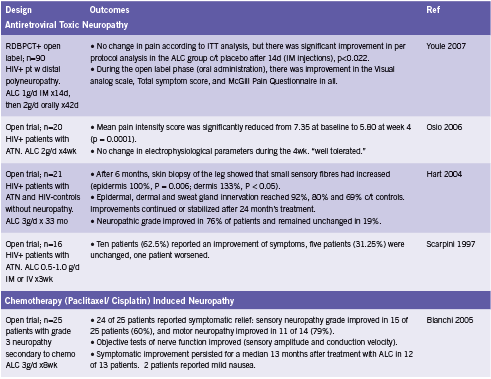
ALC has been shown to aid in the regeneration of damaged nerve tissue. Hart found that on biopsy, the skin of patients taking ALC for six months showed significant increases in the number of small sensory fibers, a 133% increase in the dermis, and 100% in the epidermis (2004). Furthermore, epidermal innervation reached up to 92% of that compared to healthy HIV- controls without neuropathy (Hart 2004). Sima demonstrated similar results in diabetic patients (2005). ALC has also been shown to improve of nerve conduction parameters, velocity and amplitude (Bianchi 2005, De Grandis 2002). Sima reported no effect on this outcome (2005), while Uzun found improvement in those patients with stage 1a diabetic neuropathy, but not those with stage 1b disease (2005).
Cerebrovascular Insufficiency
Finally, two small controlled trials have examined ALC in patients with cerebrovascular insufficiency (Arrigo 1990, Postiglione 1991). Intravenous administration of a single dose of ALC 1.5g increased cerebral blood flow compared to placebo (p<0.05) to the ischemic area but not the area corresponding to the stroke among 20 patients with a history of ischemic stroke when assessed by single proton emission computed tomography (SPECT) imaging (Postiglione 1991, Abstr). A cross over study in 12 patients undergoing rehab for cerebrovascular insufficiency reported that “significant differences between the drug [ALC] and placebo were found in memory, number and word tests and in the responses to simple stimuli and the performance of the maze test” (Arrigo 1990, Abstr).
Conclusion
Acetyl-L-carnitine possesses analgesic, neuroprotective, metabolic, and cholinergic activities. Human studies have demonstrated clinical improvements associated with ALC use in a range of neurological conditions including Alzheimer’s disease and age related cognitive impairment, psychological and cognitive effects of chronic alcohol abuse, methodone withdrawal, toxic neuropathy of various etiologies, and chronic cerebrovascular insufficiency. Although there is some question as to the consistency of its effects between individual patients especially in AD, the majority of the evidence suggests that acetyl-L-carnitine would be a valuable intervention for use in the treatment of toxic or degenerative neurological conditions where permitted by jurisdiction of practice.
Acknowledgements
The neuroprotective properties of ALC were first brought to this author’s attention by Paul Saunders ND, PhD.
References
Arnold LE, Amato A, Bozzolo H, Hollway J, Cook A, Ramadan Y, Crowl L, Zhang D, Thompson S, Testa G, Kliewer V, Wigal T, McBurnett K, Manos M. Acetyl-L-carnitine (ALC) in attention-deficit/hyperactivity disorder: a multi-site, placebo-controlled pilot trial. J Child Adolesc Psychopharmacol. 2007 Dec;17(6):791-802.
Arrigo A, Casale R, Buonocore M, Ciano C. Effects of acetyl-L-carnitine on reaction times in patients with cerebrovascular insufficiency. Int J Clin Pharmacol Res. 1990;10(1-2):133-7.
Bianchetti A, Rozzini R, Trabucchi M. Effects of acetyl-L-carnitine in Alzheimer’s disease patients unresponsive to acetylcholinesterase inhibitors. Curr Med Res Opin. 2003;19(4):350-3.
Bianchi G, Vitali G, Caraceni A, Ravaglia S, Capri G, Cundari S, Zanna C, Gianni L. Symptomatic and neurophysiological responses of paclitaxel- or cisplatin-induced neuropathy to oral acetyl-L-carnitine. Eur J Cancer. 2005 Aug;41(12):1746-50.
Bonavita E. Study of the efficacy and tolerability of L-acetylcarnitine therapy in the senile brain. Int J Clin Pharmacol Ther Toxicol. 1986 Sep;24(9):511-6.
Brooks JO 3rd, Yesavage JA, Carta A, Bravi D. Acetyl L-carnitine slows decline in younger patients with Alzheimer’s disease: a reanalysis of a double-blind, placebo-controlled study using the trilinear approach. Int Psychogeriatr. 1998 Jun;10(2):193-203.
Calvani M, Carta A, Caruso G, Benedetti N, Iannuccelli M. Action of acetyl-L-carnitine in neurodegeneration and Alzheimer’s disease. Ann N Y Acad Sci. 1992 Nov 21;663:483-6.
Cipolli C, Chiari G. [Effects of L-acetylcarnitine on mental deterioration in the aged: initial results]. Clin Ter. 1990 Mar 31;132(6 Suppl):479-510. [Abstr]
De Grandis D, Minardi C. Acetyl-L-carnitine (levacecarnine) in the treatment of diabetic neuropathy. A long-term, randomised, double-blind, placebo-controlled study. Drugs R D. 2002;3(4):223-31.
Hart AM, Wilson AD, Montovani C, Smith C, Johnson M, Terenghi G, Youle M. Acetyl-l-carnitine: a pathogenesis based treatment for HIV-associated antiretroviral toxic neuropathy. AIDS. 2004 Jul 23;18(11):1549-60.
Hudson S, Tabet N. Acetyl-L-carnitine for dementia. Cochrane Database Syst Rev. 2003;(2):CD003158. Review. PubMed PMID: 12804452.
Janiri L, Martinotti G, Tonioni F, Ghelardini C, Nicolai R, Galeotti N, Mosconi L, Calvani M, Bartolini A, Iannoni E. Acetyl-L-carnitine in the management of pain during methadone withdrawal syndrome. Clin Neuropharmacol. 2009 Jan-Feb;32(1):35-40.
Maestri A, De Pasquale Ceratti A, Cundari S, Zanna C, Cortesi E, Crinò L. A pilot study on the effect of acetyl-L-carnitine in paclitaxel- and cisplatin-induced peripheral neuropathy. Tumori. 2005 Mar-Apr;91(2):135-8.
Malaguarnera M, Cammalleri L, Gargante MP, Vacante M, Colonna V, Motta M. L-Carnitine treatment reduces severity of physical and mental fatigue and increases cognitive functions in centenarians: a randomized and controlled clinical trial. Am J Clin Nutr. 2007 Dec;86(6):1738-44.
Malaguarnera M, Gargante MP, Cristaldi E, Vacante M, Risino C, Cammalleri L, Pennisi G, Rampello L. Acetyl-L-carnitine treatment in minimal hepatic encephalopathy. Dig Dis Sci. 2008 Nov;53(11):3018-25.
Malaguarnera M, Gargante MP, Cristaldi E, Colonna V, Messano M, Koverech A, Neri S, Vacante M, Cammalleri L, Motta M. Acetyl L-carnitine (ALC) treatment in elderly patients with fatigue. Arch Gerontol Geriatr. 2008 Mar-Apr;46(2):181-90.
Martinotti G, Andreoli S, Reina D, Di Nicola M, Ortolani I, Tedeschi D, Fanella F, Pozzi G, Iannoni E, D’Iddio S, Prof LJ. Acetyl-l-Carnitine in the treatment of anhedonia, melancholic and negative symptoms in alcohol dependent subjects. Prog Neuropsychopharmacol Biol Psychiatry. 2011 Jan 20. [Epub ahead of print]
Montgomery SA, Thal LJ, Amrein R. Meta-analysis of double blind randomized controlled clinical trials of acetyl-L-carnitine versus placebo in the treatment of mild cognitive impairment and mild Alzheimer’s disease. Int Clin Psychopharmacol. 2003 Mar;18(2):61-71.
Neri M, Cortelloni C, De Vreese L. [Methodology of a clinical controlled study of L-acetylcarnitine]. Clin Ter. 1990 Mar 31;132(6 Suppl):457-68. Italian.
Osio M, Muscia F, Zampini L, Nascimbene C, Mailland E, Cargnel A, Mariani C. Acetyl-l-carnitine in the treatment of painful antiretroviral toxic neuropathy in human immunodeficiency virus patients: an open label study. J Peripher Nerv Syst. 2006 Mar;11(1):72-6.
Passeri M, Cucinotta D, Bonati PA, Iannuccelli M, Parnetti L, Senin U. Acetyl-L-carnitine in the treatment of mildly demented elderly patients. Int J Clin Pharmacol Res. 1990;10(1-2):75-9.
Passeri M, Iannuccelli M, Ciotti G, Bonati PA, Nolfe G, Cucinotta D. Mental impairment in aging: selection of patients, methods of evaluation and therapeutic possibilities of acetyl-L-carnitine. Int J Clin Pharmacol Res. 1988;8(5):367-76.
Pettegrew JW, Klunk WE, Panchalingam K, Kanfer JN, McClure RJ. Clinical and neurochemical effects of acetyl-L-carnitine in Alzheimer’s disease. Neurobiol Aging. 1995 Jan-Feb;16(1):1-4.
Postiglione A, Soricelli A, Cicerano U, Mansi L, De Chiara S, Gallotta G, Schettini G, Salvatore M. Effect of acute administration of L-acetyl carnitine on cerebral blood flow in patients with chronic cerebral infarct. Pharmacol Res. 1991 Apr;23(3):241-6.
Pueschel SM. The effect of acetyl-L-carnitine administration on persons with Down syndrome. Res Dev Disabil. 2006 Nov-Dec;27(6):599-604.
Quatraro A, Roca P, Donzella C, Acampora R, Marfella R, Giugliano D. Acetyl-L-carnitine for symptomatic diabetic neuropathy. Diabetologia. 1995 Jan;38(1):123.
Rai G, Wright G, Scott L, Beston B, Rest J, Exton-Smith AN. Double-blind, placebo controlled study of acetyl-l-carnitine in patients with Alzheimer’s dementia. Curr Med Res Opin. 1990;11(10):638-47.
Reid MS, Casadonte P, Baker S, Sanfilipo M, Braunstein D, Hitzemann R, Montgomery A, Majewska D, Robinson J, Rotrosen J. A placebo-controlled screening trial of olanzapine, valproate, and coenzyme Q10/L-carnitine for the treatment of cocaine dependence. Addiction. 2005 Mar;100 Suppl 1:43-57.
Salvioli G, Neri M. L-acetylcarnitine treatment of mental decline in the elderly. Drugs Exp Clin Res. 1994;20(4):169-76.
Sano M, Bell K, Cote L, Dooneief G, Lawton A, Legler L, Marder K, Naini A, Stern Y, Mayeux R. Double-blind parallel design pilot study of acetyl levocarnitine in patients with Alzheimer’s disease. Arch Neurol. 1992 Nov;49(11):1137-41.
Scarpini E, Sacilotto G, Baron P, Cusini M, Scarlato G. Effect of acetyl-L-carnitine in the treatment of painful peripheral neuropathies in HIV+ patients. J Peripher Nerv Syst. 1997;2(3):250-2.
Sima AA, Calvani M, Mehra M, Amato A; Acetyl-L-Carnitine Study Group. Acetyl-L-carnitine improves pain, nerve regeneration, and vibratory perception in patients with chronic diabetic neuropathy: an analysis of two randomized placebo-controlled trials. Diabetes Care. 2005 Jan;28(1):89-94.
Spagnoli A, Lucca U, Menasce G, Bandera L, Cizza G, Forloni G, Tettamanti M, Frattura L, Tiraboschi P, Comelli M, et al. Long-term acetyl-L-carnitine treatment in Alzheimer’s disease. Neurology. 1991 Nov;41(11):1726-32.
Tempesta E, Troncon R, Janiri L, Colusso L, Riscica P, Saraceni G, Gesmundo E, Calvani M, Benedetti N, Pola P. Role of acetyl-L-carnitine in the treatment of cognitive deficit in chronic alcoholism. Int J Clin Pharmacol Res. 1990;10(1-2):101-7.
Thal LJ, Calvani M, Amato A, Carta A. A 1-year controlled trial of acetyl-l-carnitine in early-onset AD. Neurology. 2000 Sep 26;55(6):805-10.
Thal LJ, Carta A, Clarke WR, Ferris SH, Friedland RP, Petersen RC, Pettegrew JW, Pfeiffer E, Raskind MA, Sano M, Tuszynski MH, Woolson RF. A 1-year multicenter placebo-controlled study of acetyl-L-carnitine in patients with Alzheimer’s disease. Neurology. 1996 Sep;47(3):705-11.
Uzun N, Sarikaya S, Uluduz D, Aydin A. Peripheric and automatic neuropathy in children with type 1 diabetes mellitus: the effect of L-carnitine treatment on the peripheral and autonomic nervous system. Electromyogr Clin Neurophysiol. 2005 Sep-Oct;45(6):343-51.
Virmani A, Binienda Z. Role of carnitine esters in brain neuropathology. Mol Aspects Med. 2004 Oct-Dec;25(5-6):533-49.
Youle M, Osio M; ALCAR Study Group. A double-blind, parallel-group, placebo-controlled, multicentre study of acetyl L-carnitine in the symptomatic treatment of antiretroviral toxic neuropathy in patients with HIV-1 infection. HIV Med. 2007 May;8(4):241-50.
Zanardi R, Smeraldi E. A double-blind, randomised, controlled clinical trial of acetyl-L-carnitine vs. amisulpride in the treatment of dysthymia. Eur Neuropsychopharmacol. 2006 May;16(4):281-7.
CoEnzyme Q10
CoEnzyme Q10
Cardiovascular applications
Introduction
In 2007, heart disease was one of the two leading causes of death for Canadians and was responsible for 22% of the 235,217 deaths (Statistics Canada 2010). Heart disease includes heart failure, angina, cardiomyopathy, coronary artery disease, valve disorders and other conditions related to the heart. ese staggering numbers call for the best possible treatments for heart disease whereupon Coenzyme Q10 (CoQ10) has appeared as a promising therapy.
Heart Facts e adult human heart weighs from 200 to 425g and is able to convert chemical energy to mechanical energy extremely effi ciently. To put it in numbers, it is capable of pumping out fi ve liters of blood every minute, 7200 liters per day and over 2.6 million liters every year. In order to execute this, the heart hydrolyzes an estimated six kilograms of adenosine-5’-triphosphate (ATP) per day (Soukoulis 2009).
High blood pressure (BP) is one of the most important cardiovascular risk factors worldwide. Approximately two-thirds of patients do not achieve optimal BP control using drug therapy (Yusuf 2004). Other interventions are extremely important, as a reduction of 5mmHg in systolic BP has been associated with a 7% reduction in all-cause mortality (Whelton 2002).
The consequences of heart failure are concerning and despite sophisticated diagnostic techniques and treatments, the risk of death within fi ve years of diagnosis is greater than 50% (Dunn 2009). CoQ10 levels in the blood have been found to be an independent predictor of mortality in congestive heart failure (Molyneux 2008).
About CoQ10
First isolated in 1957 from beef mitochondria, CoQ10, or ubiquinone is highly concentrated in heart-muscle cells due to their increased energy requirements (Crane 1989). In addition to its role in the formation of ATP, CoQ10 serves to delay or prevent lipid peroxidation and enhances cell membrane stabilization (Sarter 2002, Shah 2007). Due to the fact that mitochondria are particularly vulnerable to oxidative damage, mitochondria targeted antioxidants like CoQ10 can be an effective therapeutic strategy in preventing or reducing the progression of cardiovascular and other disorders (Graham 2009).
CoQ10 is mostly found in active organs like the heart where a substantial decline can be observed with increasing age (Pravst 2010). An adult human body has approximately 2 grams of CoQ10 and 0.5 grams must be replaced daily making the average turnover rate in the body around 4 days (Pravst 2010). Human intervention trials have found supplemental CoQ10 to be effective in cardiovascular disorders like cardiomyopathy, hypertension, angina pectoris, atherosclerosis, ischemic heart disease, cardiovascular surgery, hypertension, valvular heart diseases and myocardial infarctions (Hadj 2007, Kumar 2009).
Sources
Common sources for CoQ10 are beef, poultry, broccoli, soya oil, fish oils, peanuts, sardines, and mackerel. However, an average dietary amount of CoQ10 is only 2-6mg/day, which is inadequate to provide levels in the body required to be beneficial in pathological states (Kumar 2009, Pravst 2010).
Metabolism and Excretion
CoQ10 is absorbed from the small intestine and transferred into the blood circulation. It is then carried to the liver where it undergoes biotransformation and is primarily excreted through the bile duct. In this process, only a fraction of the ingested amount is carried to other organs like the heart, adrenal glands, kidneys and lungs (Ozaki 2010, Wyman 2010). Approximately 60% of an oral dose is eliminated in the feces during chronic oral administration and the elimination half-life is about 34 hours (Greenberg 1990). Due to its excretion pathway, patients with biliary obstruction or hepatic impairment may accumulate CoQ10 in their bodies (Ozaki 2010, Wyman 2010).
Absorbability and Dosage
Due to its lipophilic nature, CoQ10 is best absorbed with meals or in emulsified form. Divided dosing also maximizes absorbability while minimizing potential side effects (Ozaki 2010).
What form of CoQ10 is best? According to Hosoe (2007), ubiquinol, the reduced form of CoQ10, is two times more absorbable than ubiquinone. In support of ubiquinol, Langsjoen (2008) found that ubiquinol dramatically improved absorption and was correlated to improvements in CoQ10 levels and positive clinical outcomes that were not possible with the use of ubiquinone at even up to 900mg/ day. However, it has also been reported that the form of CoQ10 ingested may not be so important. According to Pravst (2010), ubiquinone appears to be reduced during or following absorption in the intestine, and as a result more than 95% of CoQ10 in circulation exists in the more active ubiquinol form after its ingestion. Thus, the answer to this question is still in contention.
The recommended daily dose of CoQ10 ranges from 30 to 100mg per day for healthy people (Pravst 2010) or 60 to 200mg per day in treating different conditions (Kumar, 2009). A maximum dosage of 1200mg has recently been suggested for adult intake as it has been found to be well tolerated and safe (Hathcock 2006).
Normal blood levels range from 0.7 to 1.0 ug/ml; doses of 30-60mg can be used to prevent CoQ10 deficiency and to maintain normal serum concentrations. However, clinically useful levels necessitate above normal CoQ10 blood levels that may be two to four times higher. In fact, 450mg of CoQ10 a day was found to achieve a plasma level of 4ug/ml and was much more successful in reversing the course of severe heart failure (Kumar 2009).
These increased levels may take days or months to achieve. Oral administration of 100mg/day of CoQ10 for two to eight months resulted in an increase of 20-85% in myocardial CoQ10 levels in patients with cardiomyopathy (Folkers 1985, Sarter 2002). Due to this slow plasma increase of CoQ10, clinical improvement is normally seen one to four weeks after initiation of treatment and it may take months to reach maximal clinical benefit (Kumar 2009).
Mechanism of action
CoQ10 appears to work in several ways including targeting expression of multiple genes, such as those involved in intermediary metabolism and cell signaling. This gene regulation and control of metabolism may explain many of the cardiovascular and other actions of CoQ10 (Rosenfeldt 2007). The beneficial effect of CoQ10 in hypertensive cases is a result of decreased peripheral resistance that is due to its direct action on the vascular wall. Free radicals inactivate nitric oxide (NO), thus preventing NOmediated relaxation of the smooth muscle layer of the vascular wall. By acting as a free radical scavenger, CoQ10 is able to prevent vasoconstriction and the resulting increase in blood pressure (Ankola 2007). With regards to coronary heart failure and myocardial infarction, CoQ10 is hypothesized to benefit these states through its direct impact on energy production by mitochondria, improving ATP availability for the failing heart (Kumar 2009, Crane 2001).
Side Effects
A long-term noncomparative study was conducted in 173 Italian centers to specifically look at safety and efficacy of CoQ10 in 2664 patients with congestive heart failure (Baggio 1994). In this study, preexisting adverse reactions were taken at baseline and then recorded again after three months of treatment with only 36 patients (1.3%) complaining of side effects mainly to do with mild gastrointestinal upset (Baggio 1994). The maximum dose in this study was 150mg per day. However, CoQ10 supplementation has been found to be free of side effects at dosages up to 600mg/day and well tolerated and safe up to 1200mg/day (Hathcock 2006). Aside from possible abdominal discomfort, nausea, vomiting, diarrhea and anorexia, allergic rash and headache have also been reported in rare cases. It is reported that CoQ10’s antiplatelet effect may increase the risk of bleeding, especially in those on antiplatelet medication (Wyman 2010). However, due to its vitamin K like action it may also act to oppose the anticoagulant effects of warfarin (Jelin 2009).
Clinical evidence
Research for CoQ10 has resulted in many positive conclusions that include Langsjoen’s study published in 1994 illustrating the usefulness of CoQ10 in clinical cardiology. It tested the use of CoQ10 in 424 patients with different types of myocardial diseases. Patients were treated with an average of 240mg of CoQ10 per day and followed an average of approximately a year and a half. Significant improvement in New York Heart Association functional classification (Table 1) was seen. 58% of patients improved by one NYHA class, 28% improved by two classes, 1.2% by three NYHA classes. Within a month, myocardial function became measurably improved. By six months, maximal improvement was usually obtained and this improvement was sustained in the majority of patients. 43% stopped between one and three drugs and only 6% required the addition of one drug. The withdrawal of CoQ10 supplementation resulted in a measurable decline in myocardial function within 1 month and a regression to pretreatment measurements within three to six months (Langsjoen 1994a, Langsjoen 1994b).

New York Heart Association (NYHA)
Classification of Functional Capacity
(Chavey 2001)
In 1994, Baggio published the largest open trial in heart failure, which included 2664 patients treated with up to 150 mg of CoQ10 per day. At the end of three-months, improvements were seen in vertigo (73.1%), subjective arrhythmia (63.4%), insomnia (62.8%), cyanosis (78.1%), edema (78.6%), pulmonary rales (77.8%), hepatomegaly (49.3%), jugular reflux (71. 8%), dyspnea (52. 7%), palpitations (75.4%), sweating (79.8%), and nocturia (53.6%). Over half (54%) of patients had improvement of at least three symptoms. Furthermore, 89.7% entered as NYHA class II moved up to a NYHA class I classification and 28.8% of patients entered as NYHA class III classification moved up to a class II classification (Baggio 1994).
The elderly population is of particular interest as using antihypertensive agents can be problematic because of the increased incidence of postural hypotension with resulting risk of falls and associated morbidity. Two large clinical trials in 1991 and 1997 respectively confirmed that in adults over the age of 60 with isolated systolic hypertension (systolic blood pressure >140, diastolic blood pressure <90), reducing systolic blood pressure by 20 mm Hg reduces the incidence of stroke, heart failure and mortality (SHEP Cooperative Research Group 1991, Rosenfeld 1997). Clinical trials of CoQ10 in hypertension, as well as trials of CoQ10 in cardiovascular pathologies, are summarized in Table 2.
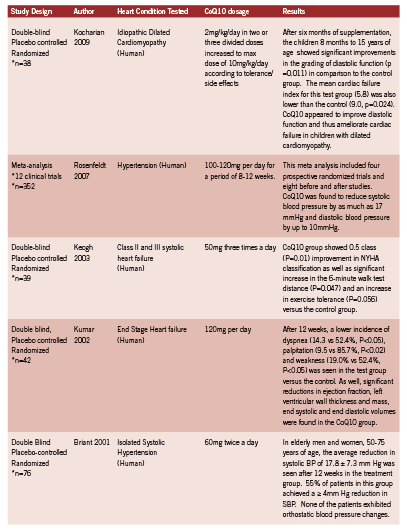
Clinical implications The use of CoQ10 in patients with heart disease may not only be useful but imperative in achieving clinically relevant improvements in heart disease patients. While more research in this area is needed with larger “n” numbers in order to statistically better quantify its merits, much research thus far points to positive outcomes in its utilization. However, its low side effect profile and non-toxic nature makes CoQ10 a viable treatment for all age groups and across a broad range of cardiovascular pathologies.
Following positive results outlined here and elsewhere, countries like Japan, Hungary, Italy, Norway and Denmark now grant licensed prescription of CoQ10 for heart failure and ischemic heart disease (Pepe 2007), a move that shows its valued acceptance and one that encourages a more prominent and wide-spread use of CoQ10.
References
Ankola DD, Viswanad B, Bhardwaj V, Ramarao P, Kumar MN. Development of potent oral nanoparticulate formulation of coenzyme Q10 for treatment of hypertension: can the simple nutritional supplements be used as first line therapeutic agents for prophylaxis/therapy? Eur J Pharm Biopharm. 2007;67(2):361-9. Epub 2007.
Baggio E, Gandini R, Plancher AC, Passeri M, Carmosino G. Italian multicenter study on the safety and efficacy of coenzyme Q10 as adjunctive therapy in heart failure. CoQ10 Drug Surveillance Investigators. Mol Aspects Med 1994;15(Suppl):S287-94.
Burke BE, Neuenschwander R, Olson RD. Randomized, double-blind, placebo-controlled trial of coenzyme Q10 in isolated systolic hypertension. South Med J. 2001;94(11):1112-7.
Chavey WE 2nd, Blaum CS, Bleske BE, Harrison RV, Kesterson S, Nicklas JM. Guideline for the management of heart failure caused by systolic dysfunction: Part I. Guideline development, etiology and diagnosis. Am Fam Physician. 2001;64(5):769-74.
Crane, FL, Hatefi Y, Lester RI, Widmer C. Isolation of a quinone from beef heart mitochondria. 1957. Biochim Biophys Acta. 1989;1000:362-3.
Crane FL. Biochemical functions of coenzyme Q10. J Am Coll Nutr. 2001;20(6):591-8.
Dunn SP, Bleske B, Dorsch M, Macaulay T, Van Tassell B, Vardeny O. Nutrition and heart failure: impact of drug therapies and management strategies. Nutr Clin Pract. 2009;24(1):60- 75.
Folkers K, Vadhanavikit S, Mortensen SA. Biochemical rationale and myocardial tissue data on the effective therapy of cardiomyopathy with coenzyme Q10. Proc Natl Acad Sci 1985;82:901–904.
Graham D, Huynh NN, Hamilton CA, Beattie E, Smith RA, Cochemé HM, Murphy MP, Dominiczak AF. Mitochondria-targeted antioxidant MitoQ10 improves endothelial function and attenuates cardiac hypertrophy. Hypertension. 2009;54(2):322-8. Epub 2009.
Greenberg S, Frishman WH. Co-enzyme Q10: a new drug for cardiovascular disease. J Clin Pharmacol 1990;30:596–608.
Hadj A, Pepe S, Rosenfeldt F. The clinical application of metabolic therapy for cardiovascular disease. Heart Lung Circ. 2007;16 Suppl 3:S56-64. Epub 2007.
Hathcock, J. N., & Shao, A. (2006). Risk assessment for coenzyme Q10 (ubiquinone). Regul Toxicol Pharmacol 45, 282–288.
Hosoe K, Kitano M, Kishida H, Kubo H, Fujii K, Kitahara M. Study on safety and bioavailability of ubiquinol after single and 4-week multiple oral administration to healthy volunteers. Regul Toxicol Pharmacol 2007;7(1):19–28.
Jelin JM, Gregory PJ, et al. Natural medicines comprehensive database/compiled by the editors of Pharmacist’s Letter, Prescriber’s Letter. 11th ed. Stockton, CA: Therapeutic Research Faculty; 2009:452–457.
Keogh A, Fenton S, Leslie C, Aboyoun C, Macdonald P, Zhao YC, Bailey M, Rosenfeldt F. Randomised double-blind, placebo-controlled trial of coenzyme Q , therapy in class II and III systolic heart failure. Heart Lung Circ. 2003;12(3):135-41.
Kocharian A, Shabanian R, Rafiei-Khorgami M, Kiani A, Heidari-Bateni G. Coenzyme Q10 improves diastolic function in children with idiopathic dilated cardiomyopathy. Cardiol Young. 2009;19(5):501-6. Epub 2009.
Kumar A, Kartikey NS, Niaz MA, Singh RB. Effect of Q gel (coenzyme Q10) in patients with end stage heart failure targetted for heart transplantation. 2002. Third Conference of the International Coenzyme Q10 Association, London, UK.
Kumar A, Kaur H, Devi P, Mohan V. Role of coenzyme Q10 (CoQ10) in cardiac disease, hypertension and Meniere-like syndrome. Pharmacol Ther. 2009;124(3):259-68. Epub 2009.
Langsjoen H, Langsjoen P, Langsjoen P, Willis R, Folkers K. Usefulness of coenzyme Q10 in clinical cardiology: a long-term study. Mol Aspects Med 1994a;15 Suppl:s165–s175.
Langsjoen P, Willisb R, Folkers K. Treatment of essential hypertension with coenzyme Q10. Mol Aspects Med 1994b;15:262–272.
Langsjoen PH, Langsjoen AM. Biofactors. Supplemental ubiquinol in patients with advanced congestive heart failure. 2008;32(1-4):119-28.
Molyneux SL, Young JM, Florkowski CM, Lever M, George PM. Coenzyme Q10: is there a clinical role and a case for measurement? Clin Biochem Rev 2008;29:71–82.
Ozaki A, Muromachi A, Sumi M, Sakai Y, Morishita K, Okamoto T. Emulsification of coenzyme Q10 using gum arabic increases bioavailability in rats and human and improves foodprocessing suitability. J Nutr Sci Vitaminol (Tokyo). 2010;56(1):41-7.
Pepe S, Marasco SF, Haas SJ, Sheeran FL, Krum H, Rosenfeldt FL. Coenzyme Q10 in cardiovascular disease. Mitochondrion. 2007;7 Suppl:S154-67. Epub 2007.
Pravst I, Zmitek K, Zmitek J. Coenzyme Q10 contents in foods and fortification strategies. Crit Rev Food Sci Nutr. 2010;50(4):269-80.
Rosenfeld J, Rodicio JL, Tuomilehto J, Zanchetti A. Randomized double-blind comparison of placebo and active treatment for older patients with isolated systolic hypertension. The Systolic Hypertension in Europe (Syst-Eur) Trial Investigators. Lancet 1997;350:757-764.
Rosenfeldt FL, Haas SJ, Krum H, Hadj A, Ng K, Leong JY, Watts GF. Coenzyme Q10 in the treatment of hypertension: a meta-analysis of the clinical trials. J Hum Hypertens. 2007;21(4):297-306. Epub 2007.
Sarter B. Coenzyme Q10 and cardiovascular disease: a review. J Cardiovasc Nurs. 2002;16(4):9-20.
Shah SA, Sander S, Cios D, Lipeika J, Kluger J, White CM. Electrocardiographic and hemodynamic effects of coenzyme Q10 in healthy individuals: a double-blind, randomized controlled trial. Ann Pharmacother. 2007;41(3):420-5. Epub 2007.
SHEP Cooperative Research Group. Prevention of stroke by antihypertensive drug treatment in older persons with isolated systolic hypertention. Final results of the Systolic Hypertension in the Elderly Program (SHEP). JAMA 1991;265:3255-3264.
Staessen JA, Fagard R, Thijs L, Celis H, Arabidze GG, Birkenhäger WH, Bulpitt CJ, de Leeuw PW, Dollery CT, Fletcher AE, Forette F, Leonetti G, Nachev C, O’Brien ET,
Soukoulis V, Dihu JB, Sole M, Anker SD, Cleland J, Fonarow GC, Metra M, Pasini E, Strzelczyk T, Taegtmeyer H, Gheorghiade M. Micronutrient deficiencies an unmet need in heart failure. J Am Coll Cardiol. 2009;54(18):1660-73.
Statistics Canada. Leading causes of death. http://www.statcan.gc.ca/daily-quotidien/101130/ dq101130b-eng.htm. Accessed December 2010.
Whelton PK, He J, Appel LJ, Cutler JA, Havas S, Kotchen TA, et al. Primary prevention of hypertension: clinical and public health advisory from the National High Blood Pressure Education Program. JAMA 2002;288(15):1882-8.
Wyman M, Leonard M, Morledge T. Coenzyme Q10: a therapy for hypertension and statininduced myalgia? Cleve Clin J Med. 2010;77(7):435-42.
Yusuf S, Hawken S, Ounpuu S, Dans T, Avezum A, Lanas F. Effect of potentially modifiable risk factors associated with myocardial infarction in 52 countries (the INTERHEART study): case-control study. Lancet 2004;364(9438):937-52.
L- Carnitine
L- Carnitine
Heidi Fritz MA, ND
Introduction
L-carnitine is a trimethylated amino acid- derivative whose most well described biological function is as a cofactor of fatty acid metabolism (Alt Med Rev 2005). L-carnitine is a key component of the intracellular shuttle system that transports free fatty acids from the cytosol and across the mitochondrial membrane where they undergo beta oxidation and transformation to ATP (Karlic 2004). Long chain fatty acids represent up to 80% of the body’s energy sources during most physiological states with the exception of high intensity exercise (Karlic 2004) and are the preferred substrate in cardiac muscle (Carvajal 2003). L-carnitine also impacts other cellular energy- producing pathways (Mate 2010). Optimization of mitochondrial metabolism through supplementation with L-carnitine, which is known to concentrate in cardiac and skeletal muscle (Flanagan 2010), could be an effective intervention for a variety of conditions characterized by fatigue, pain, and impaired muscle function. This paper focuses on the evidence base for L-carnitine in cardiovascular medicine.
Physiology
L-carnitine is considered to be conditionally essential, meaning that although there are rare genetic conditions causing primary deficiency, and secondary deficiency resulting commonly from hemodialysis in patients with kidney disease, most healthy individuals can synthesize L-carnitine from dietary lysine and methionine and cofactors vitamin C, iron, pyroxidine, and niacin (Flanagan 2010, Mate 2010). Despite this, alterations of L-carnitine metabolism are common in cardiovascular disease, most likely due to a combination of increased utilization and the inability of myocardiocytes to synthesize carnitine endogenously.
Cardiac ischemia is well recognized in the literature to be accompanied by a rapid depletion in myocardial carnitine content and a concurrent rise in intracellular long chain free fatty acids (Gurlek 2000, Regitz 1990A, Regitz 1990B, Tarantini 2006). In patients with heart failure, serum carnitine has been correlated with impaired left ventricular systolic function as measured by echocardiography (El-Aroussy 2000). During ischemia L-carnitine is thought to offset rising concentrations of free fatty acids by facilitating mitochondrial uptake and utilization, thereby preventing the damaging effects of elevated free fatty acids: membrane damage with consequent cell swelling and microvascular compression, arrhythmias, and metabolic inefficiency with consequent deterioration of myocardial function (Tarantini 2006).
In addition to its facilitating fatty acid transport into the mitochondria, L-carnitine performs a second key metabolic function: the removal from the mitochondria of short and medium chain fatty acids (acetyl groups) formed as products of beta oxidation and bound to CoA as acetyl-CoA (Mate 2010). Mitochondrial accumulation of this byproduct is toxic, inhibiting pyruvate dehydrogenase activity and glucose oxidation. On the other hand, the removal of acetyl groups and consequent increase in free CoA stimulates energy production through the Kreb’s cycle (Mate 2010). In this way, L-carnitine can regulate both fatty acid and glucose metabolism in ischemic cardiac tissue. See Figure 1. Importantly, since myocardium is unable to synthesize carnitine itself, exogenous administration may be necessary to restore optimal levels and metabolic function.
Human Evidence
Oral and intravenous L-carnitine has been shown to be benefi cial for a number of cardiovascular conditions including angina (Bartels 1996, Cacciatore 1991), acute myocardial infarction (Gurlek 2000), chronic heart failure (Rizos 2000), and peripheral vascular disease (Brevetti 1999); because it is more accessible, this summary focuses on oral L-carnitine in particular. e typical dosage is 2g per day in divided doses (Gurlek 2000, Singh 1996); some studies have used up to 6g per day (Iliceto 1995). No adverse events beyond mild gastrointestinal upset, which was rare, were reported (Kumar 2007). It should be noted that most of the studies cited here included patients also receiving standard medications for their conditions, and that L-carnitine was studied as an “add on” therapy; space constraints limit the detail in which this can be described for each study.
Angina ere are at least eight controlled human trials of oral L-carnitine for the treatment of angina. Of these, all found benefi t (Bartels 1995, Bartels 1996, Cacciatore 1991, Cherchi 1985, Cherchi 1990, Iyer 2000, Kamikawa 1984, Lagioia 1992, McMackin 2007). L carnitine has been shown to:
• Improve cardiac function (eg. cardiac output, maximal heart rate) during exercise and improve overall exercise performance (eg. exercise time, exercise workload) despite having no eff ect on myocardial oxygen requirements (Bartels 1996, Bartels 1995, Cacciatore 1991, Cherchi 1985, Iyer 2000, Kamikawa 1984, Lagioia 1992);
• Reduce myocardial ischemia upon exercise testing, measured as the time to ST-segment depression or the degree of ST-depression on ECG (Bartels 1996, Bartels 1995, Cacciatore 1991, Cherchi 1985, Cherchi 1990, Kamikawa 1984, Lagioia 1992);
• Reverse angina, with a number of patients becoming angina- free during treatment (up to 22.7% of patients compared to 9.1% in the placebo group according to Cherchi 1985, 2 out of 12 patients according to Kamikawa 1984); • Decrease frequency of angina attacks by up to 70% and nitroglycerin consumption by 57% according to Bartels 1996, comparable to treatment with diltiazem;
• One additional study in patients without angina but with established coronary artery disease showed L-carnitine to increase brachial artery diameter by 2.3%, consistent with reduced arterial tone (McMackin 2007).
L-carnitine exerts these eff ects without reducing myocardial oxygen demand, as do select cardiac medications such as beta blockers. Bartels showed that: “[proprionyl-L-carnitine] prevents ischemia-induced ventricular dysfunction, not by aff ecting the myocardial oxygen supply-demand ratio but as a result of its intrinsic metabolic actions, increasing pyruvate dehydrogenase activity and fl ux through the citric acid cycle” (1994).
Acute Myocardial Infarction
Eight controlled trials have examined the eff ect of L-carnitine in the treatment of acute myocardial infarction (AMI), often with intravenous administration for an initial period, followed by long term oral administration. ese are summarized in Table 1. When added to standard therapies, L-carnitine has been found to:
• Improve myocardial pumping ability: increasing left ventricular ejection fraction (LVEF) (Gurlek 2000), and end- diastolic and end- systolic volumes (Iliceto 1995)
• Reduce myocardial damage: reduced infarct size when administered acutely (Singh 1996), and reduced deleterious left ventricular remodeling and dilation when administered acutely and long term (Iliceto 1995)
• Decrease angina following MI by up to half that experienced by patients receiving placebo (Davini 1992, Singh 1996)
• Decrease incidence of arrhythmias following MI (Mondillo 1995, Singh 1996)
• Reduce number of second cardiac events to 15.6%, compared to 26% in placebo (Singh 1996)
• Reduce overall mortality, with 1.2% rate reported in LC group compared to 12.5% in the placebo group according to Davini 1992. Reduced mortality also demonstrated by De Pasquale (1995).
Two large randomized controlled trials have been conducted by Italian researchers. The CEDIM1 (Carnitine Ecocardiografia Digitalizzata Infarto Miocardico) and CEDIM2 trials investigated the use of intravenous L-carnitine 9g/d for the first five days after AMI, followed by 4 to 6 g/d orally for 6 to 12 months in 472 and 2230 patients respectively (Iliceto 1995, Tarantini 2006). Although the studies differed in their long term (6-12 months) findings, both demonstrate that L-carnitine can reduce early mortality post AMI. CEDIM2 found significantly reduced early mortality, risk of death at 5 days HR 0.61 (95% CI 0.37-0.98), and upon reanalysis, it was found that much of the apparent benefit seen in CEDIM1 was also due to lower early mortality (Tarantini 2006).
Of the eight studies, seven found significant positive effects (Davini 1992, De Pasquale 1995, Gurlek 2000, Iliceto 1995, Mondillo 1995, Singh 1996, Tarantini 2006). Iyer found no effect on cardiac function in a smaller trial of 60 patients using L-carnitine 6g/d intravenously for the first 7 days, then 3g/d orally for 3 months, and it has been suggested that this may be due to the smaller trial size compared to CEDIM 1 and 2, or the lower dosage used (1999).
Heart Failure
Nine controlled trials have investigated L-carnitine for patients with chronic/ congestive heart failure (CHF) (Anand 1998, Caponetto 1994, Ghidini 1988, Kumar 2007, Loster 1999, Mancini 1992, No authors 1999, Pucciarelli 1992, Rizos 2000). L-carnitine has been shown to:
• Improve exercise performance (maximum exercise time) (Caponetto 1994, Kumar 2007, Loster 1999, Mancini 1992, No authors 1999, Pucciarelli 1992, Rizos 2000)
• Improve cardiac function during exercise (eg. peak exercise heart rate, ejection fraction) (Anand 1998, Mancini 1992, No authors 1999)
• Increase cardiac pumping ability (eg. cardiac output, stroke volume index, left ventricular ejection fraction, pulmonary blood pressure) (Caponetto 1994, Pucciarelli 1992, Rizos 2000)
• Decrease left ventricular size (Anand 1998)
• Decrease signs and symptoms associated with CHF, including dyspnea, palpitations, fatigue, edema, and improved diuresis (Ghidini 1988, Kumar 2007)
• Reduce long term mortality: in one study 3-year mortality was 3% for patients receiving L-carnitine, compared to 18% for those on placebo (p<0.04) (Rizos 2000).
Interestingly, Loster found that the beneficial effects of L-carnitine continued to persist for up to 60 days after cessation of treatment (1999).
Peripheral Vascular Disease Nine controlled trials have investigated L-carnitine, mostly as proprionyl-L-carnitine (PLC), for the treatment of peripheral vascular diseases secondary to smoking or diabetes (Barker 2001, Brevetti 1999, Brevetti 1997, Brevetti 1995, Brevetti 1988, Dal Lago 1999, Greco 1992, Hiatt 2001, Santo 2006). PLC appears to benefit predominantly those patients with moderate to severe disease, with limited effect in mild disease (Brevetti 1999, 1997, Silvestro 2006). PLC has been shown to:
• Increase walking time and/ or distance, by up to 98% in those with moderate to severe disease (Brevetti 1999, Brevetti 1995, Brevetti 1988, Dal Lago 1999, Greco 1992, Hiatt 2001)
• Increase intermittent claudication distance (the distance walked until onset of claudication) (Brevetti 1999, Santo 2006)
• Improve ankle-brachial index (Greco 1992, Santo 2006)
• Improve quality of life (Brevetti 1997)
Intravenous PLC infused 3 times weekly has also been shown to augment the effects of a physical training program in patients with moderate to severe intermittent claudication (Andreozzi 2008). Biopsy of ischemic muscle has shown that supplementation with PLC increases muscle total carnitine content after 15 days (Brevetti 1988); and uncontrolled trials have shown an ability of PLC to attenuate cold induced decreases in blood flow in patients with vasospastic disease such as Raynaud’s (Gasser 1997) and reduce exercise induced increases in serum adhesion molecules, suggesting vasoprotective effects (Signorelli 2001).
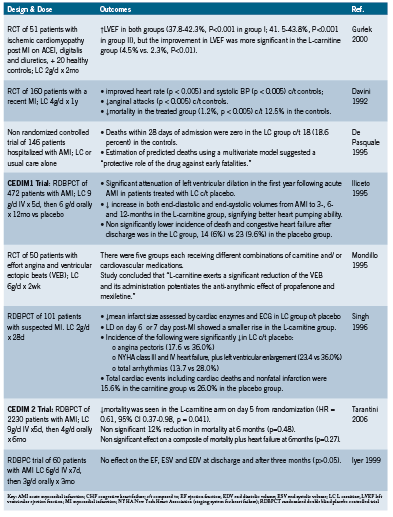
Conclusion
L-carnitine is a necessary cofactor for mitochondrial function, stimulating oxidative metabolism of glucose and fatty acids particularly under conditions of ischemia. L-carnitine protects against ischemiainduced myocardial dysfunction and has been demonstrated to improve cardiac function and exercise performance in patients with angina, myocardial infarction, and heart failure. L-carnitine can decrease frequency of angina attacks; reduce deleterious cardiac remodeling and arrhythmias, and improve survival after MI; and decrease symptoms of CHF while increasing long term survival. L-carnitine also benefits peripheral vascular disease. L-carnitine has been administered alongside standard cardioactive medications in many of the trials described above without report of any serious adverse events, and can be safely considered for comanagement of the cardiac patient.
References
Acetyl-L-carnitine. Monograph. Altern Med Rev. 2010 Apr;15(1):76-83.
Anand I, Chandrashekhan Y, De Giuli F, Pasini E, Mazzoletti A, Confortini R, Ferrari R. Acute and chronic eff ects of propionyl-L-carnitine on the hemodynamics, exercise capacity, and hormones in patients with congestive heart failure. Cardiovasc Drugs er. 1998 Jul;12(3):291-9.
Andreozzi GM, Leone A, Laudani R, Martin R, Deinit G, Cataldi V. Levo-propionylcarnitine improves the eff ectiveness of supervised physical training on the absolute claudication distance in patients with intermittent claudication. Angiology. 2008 Feb-Mar;59(1):84-9.
Barker GA, Green S, Askew CD, Green AA, Walker PJ. Eff ect of propionyl-L-carnitine on exercise performance in peripheral arterial disease. Med Sci Sports Exerc. 2001 Sep;33(9):1415-22.
Bartels GL, Remme WJ, Holwerda KJ, Kruijssen DA. Anti-ischaemic effi cacy of Lpropionylcarnitine– a promising novel metabolic approach to ischaemia? Eur Heart J. 1996 Mar;17(3):414-20.
Bartels GL, Remme WJ, den Hartog FR, Wielenga RP, Kruijssen DA. Additional antiischemic eff ects of long-term L-propionylcarnitine in anginal patients treated with conventional antianginal therapy. Cardiovasc Drugs er. 1995 Dec;9(6):749-53.
Bartels GL, Remme WJ, Pillay M, Schönfeld DH, Kruijssen DA. Eff ects of L-propionylcarnitine on ischemia-induced myocardial dysfunction in men with angina pectoris. Am J Cardiol. 1994 Jul 15;74(2):125-30.
Brevetti G, Attisano T, Perna S, Rossini A, Policicchio A, Corsi M. Eff ect of L-carnitine on the reactive hyperemia in patients aff ected by peripheral vascular disease: a double-blind, crossover study. Angiology. 1989 Oct;40(10):857-62.
Brevetti G, Chiariello M, Ferulano G, Policicchio A, Nevola E, Rossini A, Attisano T, Ambrosio G, Siliprandi N, Angelini C. Increases in walking distance in patients with peripheral vascular disease treated with L-carnitine: a double-blind, cross-over study. Circulation. 1988 Apr;77(4):767-73.
Brevetti G, Diehm C, Lambert D. European multicenter study on propionyl-L-carnitine in intermittent claudication. J Am Coll Cardiol. 1999 Nov 1;34(5):1618-24.
Brevetti G, Perna S, Sabba C, Martone VD, Di Iorio A, Barletta G. Eff ect of propionyl-L-carnitine on quality of life in intermittent claudication. Am J Cardiol. 1997 Mar 15;79(6):777-80.
Brevetti G, Perna S, Sabba C, Martone VD, Condorelli M. Propionyl-L-carnitine in intermittent claudication: double-blind, placebo-controlled, dose titration, multicenter study. J Am Coll Cardiol. 1995 Nov 15;26(6):1411-6.
Cacciatore L, Cerio R, Ciarimboli M, Cocozza M, Coto V, D’Alessandro A, D’Alessandro L, Grattarola G, Imparato L, Lingetti M, et al. e therapeutic eff ect of L-carnitine in patients with exercise-induced stable angina: a controlled study. Drugs Exp Clin Res. 1991;17(4):225-35. [Abstr]
Caponnetto S, Canale C, Masperone MA, Terracchini V, Valentini G, Brunelli C. Effi cacy of L-propionylcarnitine treatment in patients with left ventricular dysfunction. Eur Heart J. 1994 Sep;15(9):1267-73.
Carvajal K, Moreno-Sánchez R. Heart metabolic disturbances in cardiovascular diseases. Arch Med Res. 2003 Mar-Apr;34(2):89-99.
Cherchi A, Lai C, Angelino F, Trucco G, Caponnetto S, Mereto PE, Rosolen G, Manzoli U, Schiavoni G, Reale A, et al. Eff ects of L-carnitine on exercise tolerance in chronic stable angina: a multicenter, double-blind, randomized, placebo controlled crossover study. Int J Clin Pharmacol er Toxicol. 1985 Oct;23(10):569-72. [Abstr]
Cherchi A, Lai C, Onnis E, Orani E, Pirisi R, Pisano MR, Soro A, Corsi M. Propionyl carnitine in stable eff ort angina. Cardiovasc Drugs er. 1990 Apr;4(2):481-6.
Dal Lago A, De Martini D, Flore R, Gaetani E, Gasbarrini A, Gerardino L, Pola R, Santoliquido A, Serricchio M, Tondi P, Nolfe G. Eff ects of propionyl-L-carnitine on peripheral arterial obliterative disease of the lower limbs: a double-blind clinical trial. Drugs Exp Clin Res. 1999;25(1):29-36.
Davini P, Bigalli A, Lamanna F, Boem A. Controlled study on L-carnitine therapeutic effi cacy in post-infarction. Drugs Exp Clin Res. 1992;18(8):355-65.
De Pasquale B, Righetti G, Menotti A. [L-carnitine for the treatment of acute myocardial infarct]. Cardiologia. 1990 Jul;35(7):591-6.
El-Aroussy W, Rizk A, Mayhoub G, Aleem SA, El-Tobgy S, Mokhtar MS. Plasma carnitine levels as a marker of impaired left ventricular functions. Mol Cell Biochem. 2000 Oct;213(1-2):37-41.
Flanagan JL, Simmons PA, Vehige J, Willcox MD, Garrett Q. Role of carnitine in disease. Nutr Metab (Lond). 2010 Apr 16;7:30.
Gasser P, Martina B, Dubler B. Reaction of capillary blood cell velocity in nailfold capillaries to L-carnitine in patients with vasospastic disease. Drugs Exp Clin Res. 1997;23(1):39-43.
Ghidini O, Azzurro M, Vita G, Sartori G. Evaluation of the therapeutic effi cacy of L-carnitine in congestive heart failure. Int J Clin Pharmacol er Toxicol. 1988 Apr;26(4):217-20. Erratum in: Int J Clin Pharmacol er Toxicol 1989 Aug;27(8):418.
Greco AV, Mingrone G, Bianchi M, Ghirlanda G. Eff ect of propionyl-L-carnitine in the treatment of diabetic angiopathy: controlled double blind trial versus placebo. Drugs Exp Clin Res. 1992;18(2):69-80.
Gürlek A, Tutar E, Akçil E, Dinçer I, Erol C, Kocatürk PA, Oral D. e eff ects of L-carnitine treatment on left ventricular function and erythrocyte superoxide dismutase activity in patients with ischemic cardiomyopathy. Eur J Heart Fail. 2000 Jun;2(2):189-93.
Hiatt WR, Regensteiner JG, Creager MA, Hirsch AT, Cooke JP, Olin JW, Gorbunov GN, Isner J, Lukjanov YV, Tsitsiashvili MS, Zabelskaya TF, Amato A. Propionyl-L-carnitine improves exercise performance and functional status in patients with claudication. Am J Med. 2001 Jun 1;110(8):616-22.
Iliceto S, Scrutinio D, Bruzzi P, D’Ambrosio G, Boni L, Di Biase M, Biasco G, Hugenholtz PG, Rizzon P. Eff ects of L-carnitine administration on left ventricular remodeling after acute anterior myocardial infarction: the L-Carnitine Ecocardiografi a Digitalizzata Infarto Miocardico (CEDIM) Trial. J Am Coll Cardiol. 1995 Aug;26(2):380-7.
Iyer RN, Khan AA, Gupta A, Vajifdar BU, Lokhandwala YY. L-carnitine moderately improves the exercise tolerance in chronic stable angina. J Assoc Physicians India. 2000 Nov;48(11):1050-2.
Iyer R, Gupta A, Khan A, Hiremath S, Lokhandwala Y. Does left ventricular function improve with L-carnitine after acute myocardial infarction? J Postgrad Med. 1999 Apr-Jun;45(2):38-41.
Karlic H, Lohninger A. Supplementation of L-carnitine in athletes: does it make sense? Nutrition. 2004 Jul-Aug;20(7-8):709-15.
Kamikawa T, Suzuki Y, Kobayashi A, Hayashi H, Masumura Y, Nishihara K, Abe M, Yamazaki N. Eff ects of L-carnitine on exercise tolerance in patients with stable angina pectoris. Jpn Heart J. 1984 Jul;25(4):587-97.
Kumar A, Singh RB, Saxena M, Niaz MA, Josh SR, Chattopadhyay P, Mechirova V, Pella D, Fedacko J. Eff ect of carni Q-gel (ubiquinol and carnitine) on cytokines in patients with heart failure in the Tishcon study. Acta Cardiol. 2007 Aug;62(4):349-54.
Lagioia R, Scrutinio D, Mangini SG, Ricci A, Mastropasqua F, Valentini G, Ramunni G, Totaro Fila G, Rizzon P. Propionyl-L-carnitine: a new compound in the metabolic approach to the treatment of eff ort angina. Int J Cardiol. 1992 Feb;34(2):167-72. [Abstr]
Löster H, Miehe K, Punzel M, Stiller O, Pankau H, Schauer J. Prolonged oral L-carnitine substitution increases bicycle ergometer performance in patients with severe, ischemically induced cardiac insuffi ciency. Cardiovasc Drugs er. 1999 Nov;13(6):537-46.
Mancini M, Rengo F, Lingetti M, Sorrentino GP, Nolfe G. Controlled study on the therapeutic effi cacy of propionyl-L-carnitine in patients with congestive heart failure. Arzneimittelforschung. 1992 Sep;42(9):1101-4.
Mate A, Miguel-Carrasco JL, Vázquez CM. e therapeutic prospects of using L-carnitine to manage hypertension-related organ damage. Drug Discov Today. 2010 Jun;15(11-12):484-92.
McMackin CJ, Widlansky ME, Hamburg NM, Huang AL, Weller S, Holbrook M, Gokce N, Hagen TM, Keaney JF Jr, Vita JA. Eff ect of combined treatment with alpha-Lipoic acid and acetyl-L-carnitine on vascular function and blood pressure in patients with coronary artery disease. J Clin Hypertens (Greenwich). 2007 Apr;9(4):249-55.
Mondillo S, Faglia S, D’Aprile N, Mangiacotti L, Campolo MA, Agricola E, Palazzuoli V. [ erapy of arrhythmia induced by myocardial ischemia. Association of L-carnitine, propafenone and mexiletine]. Clin Ter. 1995 Dec;146(12):769-74. [Abstr]
No authors. Study on propionyl-L-carnitine in chronic heart failure. Eur Heart J. 1999 Jan;20(1):70-6.
Pucciarelli G, Mastursi M, Latte S, Sacra C, Setaro A, Lizzadro A, Nolfe G. [ e clinical and hemodynamic eff ects of propionyl-L-carnitine in the treatment of congestive heart failure]. Clin Ter. 1992 Nov;141(11):379-84. [Abstr]
Regitz V, Bossaller C, Strasser R, Müller M, Shug AL, Fleck E. Metabolic alterations in endstage and less severe heart failure–myocardial carnitine decrease. J Clin Chem Clin Biochem. 1990 Sep;28(9):611-7. A
Regitz V, Shug AL, Fleck E. Defective myocardial carnitine metabolism in congestive heart failure secondary to dilated cardiomyopathy and to coronary, hypertensive and valvular heart diseases. Am J Cardiol. 1990 Mar 15;65(11):755-60. B
Rizos I. ree-year survival of patients with heart failure caused by dilated cardiomyopathy and L-carnitine administration. Am Heart J. 2000 Feb;139(2 Pt 3):S120-3.
Santo SS, Sergio N, Luigi DP, Giuseppe M, Margherita F, Gea OC, Roberto F, Gabriella C, Giuseppe P, Massimiliano A. Eff ect of PLC on functional parameters and oxidative profi le in type 2 diabetes-associated PAD. Diabetes Res Clin Pract. 2006 Jun;72(3):231-7.
Signorelli SS, Malaponte G, Di Pino L, Digrandi D, Pennisi G, Mazzarino MC. Eff ects of ischaemic stress on leukocyte activation processes in patients with chronic peripheral occlusive arterial disease: role of L-propionyl carnitine administration. Pharmacol Res. 2001 Oct;44(4):305-9.
Singh RB, Niaz MA, Agarwal P, Beegum R, Rastogi SS, Sachan DS. A randomised, doubleblind, placebo-controlled trial of L-carnitine in suspected acute myocardial infarction. Postgrad Med J. 1996 Jan;72(843):45-50.
Tarantini G, Scrutinio D, Bruzzi P, Boni L, Rizzon P, Iliceto S. Metabolic treatment with L-carnitine in acute anterior ST segment elevation myocardial infarction. A randomized controlled trial. Cardiology. 2006;106(4):215-23.