BC Children’s Hospital and University of British Columbia (UBC) researchers have found that two existing screening tests are accurate in diagnosing development delays in children and could be incorporated in a busy family practice setting with relative ease. Parents can complete the Ages and Stages Questionnaire (ASQ) or the Parents’ Evaluation of Developmental Status (PEDS) at home or in the family physician’s office, with the physician scoring the tests and providing results in a matter of minutes. “Only 30 per cent of children with developmental delays are identified prior to school age – whether that’s social, physical or learning – and most experts would agree that we should be identifying those delays earlier through regular screening,” says Dr. Marjolaine Limbos, principal investigator and a psychologist at BC Children’s Hospital. However, researchers believe the tests are not routinely administered by Canadian family physicians because they have not been validated in a primary care setting and because of physicians’ concerns about having enough time to administer such tests in a busy practice. The authors of this research study, however, concluded that the ASQ and, to a lesser extent, the PEDS are accurate and can be administered effectively and at low cost.” (J Dev Behav Pediatr. 2011 Jul 14. PMID: 21760526)
U.S. physicians spend nearly four times more than their Canadian counterparts on administrative costs
A new University of Toronto study published in the August issue of Health Affairs, reports that U.S. physician practices spend almost four times as much money and 10 times as many hours on paperwork than Canadian practices. “The Canadian system is by no means perfect and often gets a lot of criticism, but there are points in time where we have to sit back and say our system does allow access for people and does provide quality care and it does deliver it in an efficient fashion compared to the U.S.,” said Dr. Dante Morra, lead author of the study. American practices spend $83,000 per doctor every year dealing with health insurers and other payers compared to Canadian practices that spend only $22,000. In addition, nurses and medical assistants in the U.S. spend 20.6 hours per week on administrative duties associated with payers and health insurers, whereas in Ontario, that work amounts to just 2.5 hours. Clinicians, physicians, and nurses are best suited to provide patient care but in the U.S., a significant amount of time spent is spent just dealing with paperwork associated with multiple different insurance companies. Despite the U.S.’s hefty spending, Morra said that Canada boasts better patient outcomes.
The presence of aluminum has been detected in PPC brand Calcium Gluconate Injection 10%
A recent notice from Health Canada and Pharmaceutical Partners of Canada Inc. (PPC) stated that PPC brand Calcium Gluconate Injection 10% has been found to contain aluminum. The Warning section of the package insert states: “This product contains aluminum that might be toxic. Aluminum might reach toxic levels with prolonged administration if kidney function is impaired. Premature neonates are particularly at risk because their kidneys are immature, and because they require large amounts of calcium and phosphate solutions which contain aluminum. Research indicates that patients with impaired kidney function, including premature neonates, who receive parenteral levels of aluminum at greater than 4 to 5 μg/kg/day accumulate aluminum at levels associated with central nervous system and bone toxicity.” To address the issue of aluminum toxicity, PPC is investigating alternate processes and alternate container materials, such as plastic vials, to determine if they will reduce the amount of aluminum present in the product and if they are suitable for utilization. PPC will inform healthcare professionals of the date at which the newly manufactured vials are expected to be available. Any cases of serious or unexpected adverse reactions in patients receiving Calcium Gluconate Injection 10% should be reported to PPI or Health Canada.
ZRT Laboratory saliva testing included in at-home testing study from John Hopkins
A recent study conducted by the Department of Gynecology and Obstetrics at John Hopkins Medical Institutions in Maryland, includes ZRT Laboratory saliva testing and ZRT internal research data in their examination of remote testing options. In the study, published in the May 2011 issue of Fertility and Sterility, investigators review and describe various over-the-counter testing products available to infertility patients. Doctors Brezina, Wallach, and Habel conclude that at-home testing represents an opportunity for physicians to involve patients actively in their care. “When properly used, these tests also may result in cost savings. Many of the technologies used are innovative and, with proper evaluation and implementation,could serve as valuable adjuncts to medical practices,“ the study observes. The article examines ZRT Laboratory salivary test kits, which measure a wide variety of hormones including estradiol, progesterone, testosterone, DHEA, and cortisol. The authors mention accuracy and the ease of use as the advantage of ZRT saliva testing. ZRT Laboratory, based in Beaverton, Oregon, is an industry leader for offering simple, at-home hormone and wellness test kits.
Telehealth use in Canada grew by more than 35% annually over the past 5 years
Canada is a global leader in improving access to healthcare according to a new national study. “Across the country, use of telehealth is growing rapidly, bridging the distance between patients and their care providers,” said Dr. Jennifer Zelmer, Senior Vice-President, Clinical Adoption and Innovation, Canada Health Infoway. “Canadians do not have to travel as often to receive care, and the study reports improvements in access to care, quality, and productivity valued at millions of dollars last year.” Every province and territory in Canada is using Telehealth and almost 260,000 Telehealth sessions were held in 2010, supporting services such as remote care, education for health providers and administrative meetings. Nearly half of the clinical Telehealth sessions delivered care to patients from rural and remote communities, which are home to 21% of Canadians. The report estimates that Canadians who received care via telehealth rather than travelling to other communities for care saved about $70 million in personal travel costs in 2010. In addition, there were benefits to the health system valued at $55 million per year (e.g., because of avoided federal or provincial subsidized travel costs or reduced hospitalizations for patients with chronic diseases).
Possible link between Finasteride and male breast cancer
Health Canada reports that male breast cancer has been reported in a small number of patients worldwide taking the prescription drug finasteride for male pattern baldness and enlarged prostate. Finasteride is sold under two brand names in Canada, Propecia (1 mg) to treat male-pattern hair loss and Proscar (5 mg), which is used for benign prostatic hyperplasia. Generic versions of both are also sold. Male breast cancer has been reported with both the 1 mg and 5 mg formulations of finasteride but most of the reports have been in association with the 5mg formulation. Based on the currently available evidence, it is not known with certainty whether finasteride can cause breast cancer, nor can this possibility be ruled out at this point in time. The labeling for Propecia, Proscar and several of the generic finasteride products has already been updated to include information on the potential risk of male breast cancer. Updates to the remaining generic drugs will follow. Healthcare professionals should encourage their patients taking finasteride to report any changes in their breasts, such as breast enlargement, lumps, tenderness, pain or nipple discharge.
SickKids app helps patients manage IBD
The Hospital for Sick Children in Toronto has launched a free mobile app to empower people with Inflammatory Bowel Disease (IBD) to take control of the disease. myIBD app allows patients to track their food, stool, pain and frequency of washroom visits all on mobile devices. The app offers a visual tracking system so patients can monitor their disease activity and seek help when necessary in an effort to avoid flare-ups and hospital admissions. “We support our patients and their families as they adjust to the lifestyle changes required to effectively manage IBD, whether it be diet and lifestyle changes, medication, or simply monitoring symptoms. The app provides patients with immediate, round-the-clock access to current, reliable and educational IBD information to supplement the information they get in the clinic,” says Karen Frost, one of the Project Leaders and IBD Nurse Practitioner at SickKids. “With education comes a good foundation for long term remission of disease. We stick to our mantra that ‘remission is the mission’.” myIBD app is currently available for free in the App Store for iPhones, iPad, and the iPod touch. It will also be available to Android devices later this year.
Low magnesium levels can be associated with long- term use of Proton Pump Inhibitor drugs
The U.S. Food and Drug Administration (FDA) is informing the public that prescription proton pump inhibitor (PPI) drugs may cause low serum magnesium levels if taken for prolonged periods of time (i.e., longer than one year). In approximately one-quarter of the cases reviewed, magnesium supplementation alone did not improve low serum magnesium levels and the PPI had to be discontinued. In contrast to prescription PPIs, over-the- counter (OTC) PPIs are marketed at low doses and are only intended for a 14 day course of treatment up to three times per year. The FDA believes that there is very little risk of hypomagnesemia when OTC PPIs are used according to the directions on the OTC label. Low serum magnesium levels can result in serious adverse events including tetany, arrhythmias, and seizures. The FDA urges healthcare professionals to obtain serum magnesium levels prior to initiation of prescription PPI treatment in patients expected to be on these drugs for long periods of time, as well as patients who take PPIs with medications such as digoxin, diuretics, or other drugs that may cause hypomagnesemia.
Long-term cannabis use
Long-term cannabis use
increased risk of psychosis?
Cognitive impairments that ensue with long-term cannabis use are well documented (Solowij 1995a, Solowij 1991, Solowij 2011a, Solowij 1995b, Solowij 2002) and evidence is emerging that suggests an infl uence in the development of psychosis and schizophrenia (Di Forti 2009, Leweke 1999, omas 1996, Tien 1990, van Os 2002). ough the risks of acquiring mental illness from long term daily or near-daily cannabis use remains a debate between the proponents of its legislation and its partisans of continued prohibition (Hall 1994), there is an emerging body of evidence that researchers implore not be ignored. Studies have surfaced that clarify the understanding of the eff ects of cannabis use on the brain and that have quantifi ed the extent of the risks of long-term use (Leweke 2008, Murray 2007).
The Endocannabinoid System
Historically, explanation of reports of adverse eff ects associated with cannabis consumption, including psychotic episodes have been signifi cantly hindered by a lack of knowledge regarding their underlying neurobiological and pharmacological processes (Leweke 2004). However, the discovery of the endogenous cannabinoid system in the late 1980s has assisted in elucidating the molecular basis of cannabis, the action of cannabis and cannabinoids in the brain, and the relationship these cannabinoids have with psychotic symptoms and disorders (Leweke 2004).
The cannabinoid-1 (CB1) receptor has been discovered as the target for delta-9-tetrahydrocannabinol (Delta-9-THC) in the rat brain (Devane 1988). Delta-9-THC was the fi rst exogenous cannabinoid CB1 activator that was isolated from preparations of the cannabis sativa plant. Subsequently, two endogenous ligands for this receptor were identifi ed: anandamide and 2-arachidonylglycerol (2-AG). In recent years, many studies have investigated the CB1 receptor and the endogenous cannabinoid system (Elphick 2001, Fernandez 1997, Giuff rida 2000, Howlett 1995, Manzanares 1999, Schlicker 2001, Wilson 2002). Studies on various mammals have found that the anatomical distribution of the CB1 receptor is highly consistent among diff erent species (Herkenham 1990, Herkenham 1991, Matsuda 1993), concentrating in parts of the limbic system (particularly the hippocampus and the amygdala) and in the cerebellum (Solowij 2011b).
On a functional level, CB1 receptors are activated by endogenous cannabinoids such as anandamide and 2-AG at the presynaptic nerve terminal. Binding of these cannabinoids leads to a decrease in membrane permeability to calcium and potassium ions, as well as to a decrease in the activity of adenylate cyclase, thereby inhibiting the release of glutamate, dopamine, acetylcholine, and noradrenaline (norepinephrine) (Leweke 2004). GABA reuptake is also inhibited (Leweke 2004).
A role for the Endocannabinoid System in Psychosis and Schizophrenia
Since the discovery of the endocannabinoid system, a growing body of psychiatric research has focused on the role of this system in major psychiatric disorders (Leweke 2004). Evidence suggests that cannabinoid receptors, the pharmacological target of cannabis-derived drugs, and their accompanying system of endogenous activators may be dysfunctional in schizophrenia (Leweke 1999). Leweke et al purifi ed and quantifi ed endogenous cannabinoids from cerebrospinal fl uid (CSF) of 10 patients with schizophrenia and 11 non-schizophrenic controls. It was found that CSF concentrations of two endogenous cannabinoids (anandamide and palmitylethanolamide) were signifi cantly higher in schizophrenic patients than in non-schizophrenic controls (Leweke 1999). ese elevated endogenous cannabinoids in schizophrenic patients are thought to refl ect an imbalance in the endogenous cannabinoid signalling, which may contribute to the pathogenesis of schizophrenia (Leweke 1999).
Koethe (2009) explored the proposed homeostatic role of anandamide in schizophrenia. eir study included subjects not yet diagnosed with schizophrenia, but exhibiting signs of psychosis. CSF anandamide levels were assessed in subjects in this prodromal state of psychosis and in healthy volunteers. Results revealed that anandamide levels in subjects in the prodromal state were signifi cantly elevated and that those with lower levels of anandamide actually showed a higher risk for transiting to psychosis earlier. is anandamidergic up-regulation in the initial prodromal course may suggest a protective role of the endocannabinoid system in early schizophrenia (Koethe 2009), refl ecting a compensatory adaptation to the disease state (Giuff rida 2004). at CSF anandamide levels correlate inversely with psychotic symptoms suggests that anandamide release into the central nervous system may serve as an adaptive mechanism countering neurotransmitter abnormalities in acute psychosis (Leweke 2007).
Guiff rida and colleagues researched the infl uence that pharmaceutical treatment had on anandamide levels in patients with schizophrenia compared to anti-psychotic naïve individuals suff ering a fi rst-episode psychosis to further understand the relationship between the endocannabinoid system, neurotransmission and psychosis. ey found that individuals with schizophrenia treated with ‘typical antipsychotics’ (antagonists of the dopamine D2-like receptor) did not have the same elevations in anandamide levels, though individuals with schizophrenia treated with ‘atypical’ antipsychotics (antagonists of 5HT(2A) receptors) did (Giuff rida 2004). ese fi ndings are congruent with the hypotheses concerning the interactions between cannabis, endocannabinoids, and dopamine whereby cannabisinduced dopamine dysregulation may give rise to delusions and hallucinations (Kuepper 2010). Kuepper (2010) provide a review of possible dopamine pathways in psychosis based on animal research that suggests that delta-9-THC increases dopamine levels in several regions of the brain; however, such discussion, in its complexity, goes beyond the scope of this article.
The Relationship Between Cannabis Use, Psychosis and Schizophrenia
The consensus in the literature regarding the association between psychosis, schizophrenia, and cannabis use is one that is congruent with the vulnerability/stress model of schizophrenia developed by Nuechterlein and Dawson (Nuechterlein 1984). at is, cannabis is considered one stress factor among others that may either enhance or trigger schizophrenic symptoms (Leweke 2004). A community survey on the early course and onset of schizophrenia in Germany was able to diff erentiate three approximately equal sized groups of patients with cannabis-associated psychosis: patients who had been using cannabis for several years before the fi rst (prodromal) signs of schizophrenia emerged, patients who had experienced the onset of both cannabis use and schizophrenia within one month of each other, and patients who had started to use cannabis after the onset of symptoms of schizophrenia (Hambrecht 2000). According to Lousia Degenhardt & Wayne Hall (2002) “it is unlikely that cannabis use causes psychosis among persons who would otherwise not have developed the disorder. e evidence is more consistent with the hypotheses that cannabis use may precipitate psychosis among vulnerable individuals, increase the risk of relapse among those who have already developed the disorder, and may be more likely to lead to dependence in persons with schizophrenia”. Additionally, Degenhardt, Hall and Lyndskey (2003) state that “cannabis use does not appear to be causally related to the incidence of schizophrenia, but its use may precipitate disorders in persons who are vulnerable to developing psychosis and worsen the course of the disorder among those who have already developed it”.
Difference between delta-9-tetrahydrocannabinol (THC) and cannabidiol (CBD): Regional Brain Function and Neurophysiology
Data on the underlying composition of the herbal preparations (ie. the diff erent natural cannabinoids present) or on the plasma levels of delta-9-THC, are generally not provided in retrospective clinical studies (Leweke 2004). Generally, the term ‘cannabis’ in the literature refers to all herbal cannabis preparations with a poorly defi ned mixture of diff erent natural cannabioids. Only when a concentration is given or when a precise term is used can it be certain that the eff ects of a specifi c cannabinoid are being studied (Leweke 2004). It is important to note that delta-9-THC and cannabidiol (CBD), the two most common exogenous cannabinoids, have distinct symptomatic and behavioural eff ects (Bhattacharyya 2010). Where delta-9-THC impairs reaction on movement and response-inhibition tasks, causes acute psychotic symptoms, and destabilizes brain function, CBD does not impair performance or induce psychosis, appears to reduce anxiety, and stabilizes brain function (Fusar-Poli 2009).
Functional magnetic resonance imaging (fMRI) was used in healthy volunteers to examine whether delta-9-THC and CBD had opposite eff ects on regional brain function in healthy volunteers with minimal cannabis exposure (Bhattacharyya 2010). Subjects were scanned on three occasions following oral administration of delta-9-THC, CBD or placebo while performing a verbal memory task, a response inhibition task, a sensory processing task, and when viewing fearful faces. Results revealed that delta-9-THC and CBD have opposite eff ects on activation relative to placebo (1) in the striatum during verbal recall, (2) in the hippocampus during the response inhibition task, (3) in the amygdala when subjects viewed fearful faces, (4) in the superior temporal cortex when subjects listened to speech, and (5) in the occipital cortex during visual processing (Bhattacharyya 2010). Using an eventrelated paradigm with faces that implicitly elicited diff erent levels of anxiety, Fuser-Poli et al demonstrated distinct eff ects of delta-9-THC and CBD on neural activation with fMRI and electrodermal response during emotional processing further validating their dissimilar consequences on regional brain function (Fuser-Poli 2009). CBD attenuated the neurofunctional engagement of the amygdala and cingulated cortex and reduced the electrodermal response when subjects viewed intensely fearful stimuli, verifying its anxiolytic eff ects. Alternatively, delta-9-THC modulated activation in frontal and parietal areas and augmented the electrodermal response; eff ects validating an increase in anxiety (Fusar-Poli 2009).
These diff erences in the behavioural eff ects of delta-9-THC and CBD are paralleled by diff erences in their mechanisms of action at the molecular level (Fusar-Poli 2009). Delta-9-THC binds to neuronal CB1 receptors (Devane 1992), which are found on GABAergic and glutamatergic neurons throughout the brain (Braida 2007, Herkenham 1990, Mechoulam 2003). CBD has a very low affi nity for the cannabinoid CB1 receptor (Petitet 1998).
Brain Abnormalities and Long-term Heavy Cannabis Use
There exists confl icting evidence concerning structural consequences of long- term cannabis use on the brain. A series of preclinical investigations demonstrated induction of neurotoxic changes in the hippocampus (Chan 1998, Landfi eld 1988, Lawston 2000, Scallet 1987) leading to further investigation in human trials. Yucel et al conducted a region-of-interest-based analysis using high resolution 3 Telsa magnetic resonance imaging (3-T MRI) to assess volumetric changes in the hippocampus and the amygdala (Yucel 2008). Subjects who were non-using healthy male volunteers and those with a long history of cannabis use were carefully screened for polysubstance abuse and mental disorders as well as for subthreshold psychotic symptoms and verbal learning abilities. Findings in this study corroborate with the above cited animal research suggesting that long-term heavy cannabis use is associated with signifi cant and localized hippocampal volume reductions that relate to increasing cumulative cannabis exposure (Yucel 2008). Bilateral reduction in amygdala volume was also seen (Yucel 2008). Long-term heavy cannabis use and its association with smaller cerebellar white-matter volume similar to that observed in schizophrenia has also been shown (Solowij 2011b). Reduced volumes were more pronounced in patients with schizophrenia who use cannabis, but were apparent in healthy individuals with a cannabis use history as well (Solowij 2011b). ese fi ndings indicate that heavy cannabis use across protracted periods exert harmful effects on brain tissue, and subsequently on mental health (Yucel 2008).
Vulnerability and Genetics
Compatible with the vulnerability/stress model of schizophrenia, gene-environment interactions involving the catechol-Omethyltransferase Valine(158)Methionine polymorphism (COMT(Val158Met)) have been implicated in the causation of psychosis (van Winkel 2008). In a stress exposure study in members of the Greek army, carriers of the COMT Val158Met allele were more susceptible to the eff ects of stress on the psychosis outcome than those with the catechol-O-methyltransferase Methionine(158)Methionine genotype (Stefanis 2007). In addition, carriers of the COMT Val(158)Met allele were more likely to exhibit psychotic symptoms and to develop schizophreniform disorder if they used cannabis; whereas cannabis use had no such adverse influence on individuals with two copies of the methionine allele (COMT(Met158Met)) (Caspi 2005).
Cannabis use in adolescents is another gene-environment factor that increases the likelihood of experiencing symptoms of schizophrenia in adulthood (Arseneault 2002). After controlling for pre-existing psychotic symptoms, it was found in the Dunedin Multidisciplinary Health and Development study from New Zealand that cannabis use increased the risk of developing symptoms of schizophrenia in this cohort (Arseneault 2002). One-tenth of those in this sample who were using cannabis by the age of 15 years had developed a schizophrenia-like disorder by the age of 26 years. Onset of cannabis use prior to the age of 15 years conferred the greatest risk of subsequently developing schizophrenia. This increased risk was specific to cannabis use as opposed to the use of other drugs (Arseneault 2002). Two large studies, one out of Greece in 2004 (n=3500) (Stefanis 2004) and another out of the Netherlands in 2005 (n=2437) (Henquet 2005) found results adding credence to the hypothesis that cannabis use early in adolescents contributes to the risk of developing psychosis, particularly in those with a predisposition.
Conclusion
There is sufficient evidence that cannabinoids can induce acute transient psychotic symptoms or an acute psychosis in some individuals (Sewell 2010). It is also clear that cannabinoids can exacerbate psychosis in individuals with an established or predisposed psychotic disorder, and that these exacerbations may last beyond the period of intoxication (Sewell 2010). When these symptoms are present in a clinical setting, the practitioner should not hesitate to inquire about cannabis use, educating the patient about the possible risks involved as evidenced epidemiologically.
References:
Arseneault, L., Cannon, M., Poulton, R., Murray, R., Caspi, A., & Moffitt, T. E. Cannabis use in adolescence and risk for adult psychosis: longitudinal prospective study. British Medicial Journal. 2002; 325(7374): 1212-1213.
Ashton, C. H. Pharmacology and effects of cannabis: a brief review. The British journal of psychiatry : the journal of mental science. 2001; 178: 101-106.
Bhattacharyya, S., Morrison, P. D., Fusar-Poli, P., Martin-Santos, R., Borgwardt, S., Winton-Brown, T., McGuire, P. K. Opposite effects of delta-9-tetrahydrocannabinol and cannabidiol on human brain function and psychopathology. Neuropsychopharmacology : official publication of the American College of Neuropsychopharmacology. 2010; 35(3): 764-774.
Bisogno, T., L. Hanus, et al. Molecular targets for cannabidiol and its synthetic analogues: effect on vanilloid VR1 receptors and on the cellular uptake and enzymatic hydrolysis of anandamide. British Journal of Pharmacology. 2001; 134(4): 845-852.
Braida, D., Limonta, V., Malabarba, L., Zani, A., & Sala, M. 5-HT1A receptors are involved in the anxiolytic effect of Delta9-tetrahydrocannabinol and AM 404, the anandamide transport inhibitor, in Sprague-Dawley rats. European journal of pharmacology. 2007; 555(2-3): 156-163.
Caspi, A., Moffitt, T. E., Cannon, M., McClay, J., Murray, R., Harrington, H., Craig, I. W. Moderation of the effect of adolescent-onset cannabis use on adult psychosis by a functional polymorphism in the catechol-O-methyltransferase gene: longitudinal evidence of a gene X environment interaction. Biological Psychiatry. 2005; 57(10): 1117-1127.
Chan, G. C., Hinds, T. R., Impey, S., & Storm, D. R. Hippocampal neurotoxicity of Delta9-tetrahydrocannabinol. The Journal of neuroscience : the official journal of the Society for Neuroscience. 1998; 18(14): 5322-5332.
Degenhardt, Hall (2002). Cannabis and Psychosis. Current Psychiatry Report June; 4(3); 191-6.
Degenhardt , Hall, Lynskey (2003) Testing hypotheses about the relationship between cannabis use and psychosis. Drug and Alcohol Dependence (71) 37-48.
Devane, W. A., Dysarz, F. A., 3rd, Johnson, M. R., Melvin, L. S., & Howlett, A. C. Determination and characterization of a cannabinoid receptor in rat brain. Molecular pharmacology. 1988; 34(5): 605-613.
Devane, W. A., Hanus, L., Breuer, A., Pertwee, R. G., Stevenson, L. A., Griffin, G., Mechoulam, R. Isolation and structure of a brain constituent that binds to the cannabinoid receptor. Science. 1992; 258(5090): 1946-1949.
Di Forti, M., Morgan, C., Dazzan, P., Pariante, C., Mondelli, V., Marques, T. R., Murray, R. M. High-potency cannabis and the risk of psychosis. The British journal of psychiatry : the journal of mental science. 2009; 195(6): 488-491.
Elphick, M. R., Egertova, M. The neurobiology and evolution of cannabinoid signalling. Philosophical transactions of the Royal Society of London. Series B, Biological sciences. 2001; 356(1407): 381-408.
Fernandez-Ruiz, J. J., Munoz, R. M., Romero, J., Villanua, M. A., Makriyannis, A., Ramos, J. A. Time course of the effects of different cannabimimetics on prolactin and gonadotrophin secretion: evidence for the presence of CB1 receptors in hypothalamic structures and their involvement in the effects of cannabimimetics. Biochemical pharmacology. 1997; 53(12): 1919-1927.
Fusar-Poli, P., Crippa, J. A., Bhattacharyya, S., Borgwardt, S. J., Allen, P., Martin- Santos, R., McGuire, P. K. Distinct effects of {delta}9-tetrahydrocannabinol and cannabidiol on neural activation during emotional processing. Archives of general psychiatry. 2009; 66(1): 95-105.
Giuffrida, A., Desarnaud, F., Piomelli, D. Endogenous cannabinoid signaling and psychomotor disorders. Prostaglandins & other lipid mediators. 2001; 61(1-2): 63-70.
Giuffrida, A., Leweke, F. M., Gerth, C. W., Schreiber, D., Koethe, D., Faulhaber, J., Piomelli, D. Cerebrospinal anandamide levels are elevated in acute schizophrenia and are inversely correlated with psychotic symptoms. Neuropsychopharmacology : official publication of the American College of Neuropsychopharmacology. 2004; 29(11): 2108-2114.
Glass, M., Dragunow, M., & Faull, R. L. Cannabinoid receptors in the human brain: a detailed anatomical and quantitative autoradiographic study in the fetal, neonatal and adult human brain. Neuroscience. 1997. 77(2): 299-318.
Hall, W., Solowij, N. and Lemon, J. (1994) Health and Psychological Consequences of Cannabis Use. National Drug Strategy Monograph Series No. 25. Cranberra: Australian Government Publication Service.
Hambrecht, M., Hafner H. Cannabis, vulnerability, and the onset of schizophrenia: an epidemiological perspective. The Australian and New Zealand journal of psychiatry. 2000; 34(3): 468-475.
Henquet, C., Krabbendam, L., Spauwen, J., Kaplan, C., Lieb, R., Wittchen, H. U., & van Os, J. Prospective cohort study of cannabis use, predisposition for psychosis, and psychotic symptoms in young people. British Medical Journal. 2005; 330(7481): 11.
Herkenham, M., Lynn, A. B., Johnson, M. R., Melvin, L. S., de Costa, B. R., Rice, K. C. Characterization and localization of cannabinoid receptors in rat brain: a quantitative in vitro autoradiographic study. The Journal of neuroscience : the official journal of the Society for Neuroscience. 1991; 11(2): 563-583.
Herkenham, M., Lynn, A. B., Little, M. D., Johnson, M. R., Melvin, L. S., de Costa, B. R., Rice, K. C. Cannabinoid receptor localization in brain. Proceedings of the National Academy of Sciences of the United States of America. 1990; 87(5): 1932-1936.
Howlett, A. C. Pharmacology of cannabinoid receptors. Annual review of pharmacology and toxicology. 1995; 35: 607-634.
Koethe, D., Giuffrida, A., Schreiber, D., Hellmich, M., Schultze-Lutter, F., Ruhrmann, S., Leweke, F. M. Anandamide elevation in cerebrospinal fluid in initial prodromal states of psychosis. The British journal of psychiatry : the journal of mental science. 2009. 194(4): 371-372.
Kranaster, L., Koethe D., Cerebrospinal fluid diagnostics in first-episode schizophrenia. European archives of psychiatry and clinical neuroscience. 2011;
Kuepper, R., Morrison, P. D., van Os, J., Murray, R. M., Kenis, G., Henquet, C. Does dopamine mediate the psychosis-inducing effects of cannabis? A review and integration of findings across disciplines. Schizophrenia research. 2010; 121(1-3): 107-117.
Landfield, P. W., Cadwallader, L. B., Vinsant, S. Quantitative changes in hippocampal structure following long-term exposure to delta 9-tetrahydrocannabinol: possible mediation by glucocorticoid systems. Brain research. 1988; 443(1-2): 47-62.
Lawston, J., Borella, A., Robinson, J. K., Whitaker-Azmitia, P. M. Changes in hippocampal morphology following chronic treatment with the synthetic cannabinoid WIN 55,212-2. Brain research. 2000; 877(2): 407-410.
Leweke, F. M., Gerth, C. W., Klosterkotter, J. Cannabis-associated psychosis: current status of research. CNS drugs. 2004; 18(13): 895-910.
Leweke, F. M., Giuffrida, A., Koethe, D., Schreiber, D., Nolden, B. M., Kranaster, L., Piomelli, D. Anandamide levels in cerebrospinal fluid of first-episode schizophrenic patients: impact of cannabis use. Schizophrenia research. 2007; 94(1-3): 29-36.
Leweke, F. M., Giuffrida, A., Wurster, U., Emrich, H. M., Piomelli, D. Elevated endogenous cannabinoids in schizophrenia. Neuroreport. 1999; 10(8): 1665-1669.
Leweke, F. M., Koethe, D. Cannabis and psychiatric disorders: it is not only addiction. Addiction biology. 2008; 13(2): 264-275.
Leweke, F. M., Schneider, U., Thies, M., Munte, T. F., Emrich, H. M. Effects of synthetic delta9-tetrahydrocannabinol on binocular depth inversion of natural and artificial objects in man. Psychopharmacology. 1999; 142(3): 230-235.
Leweke, M., Kampmann, C., Radwan, M., Dietrich, D. E., Johannes, S., Emrich, H. M., Munte, T. F. The effects of tetrahydrocannabinol on the recognition of emotionally charged words: an analysis using event-related brain potentials. Neuropsychobiology. 1998; 37(2): 104-111.
Lorenzetti, V., Lubman, D.I. Structural MRI findings in long-term cannabis users: what do we know? Substance use & misuse. 2010; 45(11): 1787-1808.
Manzanares, J., Corchero, J., Romero, J., Fernandez-Ruiz, J. J., Ramos, J. A., Fuentes, J. A. Pharmacological and biochemical interactions between opioids and cannabinoids. Trends in pharmacological sciences. 1999. 20(7): 287-294.
Matsuda, L. A., Bonner, T. I., Lolait, S. J. Localization of cannabinoid receptor mRNA in rat brain. The Journal of comparative neurology. 1993; 327(4): 535-550.
Mechoulam, R., Hanus, L. Cannabidiol: an overview of some chemical and pharmacological aspects. Part I: chemical aspects. Chemistry and physics of lipids. 2002; 121(1-2): 35-43.
Mechoulam, R., Lichtman, A.H. Neuroscience. Stout guards of the central nervous system. Science. 2003; 302(5642): 65-67.
Moreira, F. A., Aguiar, D.C. Anxiolytic-like effect of cannabidiol in the rat Vogel conflict test. Progress in neuro-psychopharmacology & biological psychiatry. 2006; 30(8): 1466-1471.
Morgan, C. J., Curran H.V. Effects of cannabidiol on schizophrenia-like symptoms in people who use cannabis. The British journal of psychiatry : the journal of mental science. 2008; 192(4): 306-307.
Morgan, C. J., Schafer, G. Impact of cannabidiol on the acute memory and psychotomimetic effects of smoked cannabis: naturalistic study: naturalistic study [corrected]. The British journal of psychiatry : the journal of mental science. 2010; 197(4): 285-290.
Munro, S., Thomas, K.L. Molecular characterization of a peripheral receptor for cannabinoids. Nature. 1993; 365(6441): 61-65.
Murray, R. M., Morrison, P. D., Henquet, C., Di Forti, M. Cannabis, the mind and society: the hash realities. Nature reviews. Neuroscience. 2007; 8(11): 885-895.
Nuechterlein, K. H. Dawson, M.E. A heuristic vulnerability/stress model of schizophrenic episodes. Schizophrenia bulletin. 1994; 10(2): 300-312.
Petitet, F., Jeantaud, B., Reibaud, M., Imperato, A., Dubroeucq, M. C. Complex pharmacology of natural cannabinoids: evidence for partial agonist activity of delta9- tetrahydrocannabinol and antagonist activity of cannabidiol on rat brain cannabinoid receptors. Life sciences. 1998; 63(1): PL1-6.
Potter, D. J., Clark, P. Potency of delta 9-THC and other cannabinoids in cannabis in England in 2005: implications for psychoactivity and pharmacology. Journal of forensic sciences. 2008; 53(1): 90-94.
Scallet, A. C., Uemura, E., Andrews, A., Ali, S. F., McMillan, D. E., Paule, M. G., Slikker, W., Jr. Morphometric studies of the rat hippocampus following chronic delta- 9-tetrahydrocannabinol (THC). Brain research. 1987; 436(1): 193-198.
Schlicker, E., Kathmann, M. Modulation of transmitter release via presynaptic cannabinoid receptors. Trends in pharmacological sciences. 2001; 22(11): 565-572.
Sewell, R. A., Skosnik, P. D., Garcia-Sosa, I., Ranganathan, M., D’Souza, D. C. Behavioral, cognitive and psychophysiological effects of cannabinoids: relevance to psychosis and schizophrenia. Revista brasileira de psiquiatria. 2010; 32 Suppl 1: S15-30.
Solowij, N. Do cognitive impairments recover following cessation of cannabis use? Life sciences. 1995a; 56(23-24): 2119-2126.
Solowij, N., Jones, K. A., Rozman, M. E., Davis, S. M., Ciarrochi, J., Heaven, P. C., Yucel, M. Verbal learning and memory in adolescent cannabis users, alcohol users and non-users. Psychopharmacology. 2011a; 216(1): 131-144.
Solowij, N., Michie, P. T., Fox, A. M.Effects of long-term cannabis use on selective attention: an event-related potential study. Pharmacology, biochemistry, and behavior. 1991; 40(3): 683-688.
Solowij, N., Michie, P. T., Fox, A. M. Differential impairments of selective attention due to frequency and duration of cannabis use. Biological psychiatry. 1995b; 37(10): 731-739.
Solowij, N., Stephens, R. S., Roffman, R. A., Babor, T., Kadden, R., Miller, M., Vendetti, J. Cognitive functioning of long-term heavy cannabis users seeking treatment. JAMA : the journal of the American Medical Association. 2002; 287(9): 1123-1131.
Solowij, N., Yucel, M., Respondek, C., Whittle, S., Lindsay, E., Pantelis, C., Lubman, D. I. Cerebellar white-matter changes in cannabis users with and without schizophrenia. Psychological medicine. 2011b; 1-11.
Stefanis, N. C., Delespaul, P., Henquet, C., Bakoula, C., Stefanis, C. N., Van Os, J. Early adolescent cannabis exposure and positive and negative dimensions of psychosis. Addiction. 2004; 99(10): 1333-1341.
Stefanis, N. C., Henquet, C., Avramopoulos, D., Smyrnis, N., Evdokimidis, I., Myin- Germeys, I., Van Os, J. COMT Val158Met moderation of stress-induced psychosis. Psychological medicine. 2007; 37(11): 1651-1656.
Stella, N., Schweitzer, P., Piomelli, D. A second endogenous cannabinoid that modulates long-term potentiation. Nature. 1997; 388(6644): 773-778.
Thomas, H. A community survey of adverse effects of cannabis use. Drug and alcohol dependence. 1996; 42(3): 201-207.
Tien, A. Y., Anthony J.C. Epidemiological analysis of alcohol and drug use as risk factors for psychotic experiences. The Journal of nervous and mental disease. 1990; 178(8): 473-480.
van Winkel, R., Henquet, C., Rosa, A., Papiol, S., Fananas, L., De Hert, M., Myin-Germeys, I. Evidence that the COMT(Val158Met) polymorphism moderates sensitivity to stress in psychosis: an experience-sampling study. American journal of medical genetics. Part B, Neuropsychiatric genetics : the official publication of the International Society of Psychiatric Genetics. 2008; 147B(1): 10-17.
Wilson, R. I., Nicoll R.A. Endocannabinoid signaling in the brain. Science. 2002; 296(5568): 678-682.
Yucel, M., Solowij, N., Respondek, C., Whittle, S., Fornito, A., Pantelis, C., Lubman, D. I. Regional brain abnormalities associated with long-term heavy cannabis use. Archives of general psychiatry. 2008; 65(6): 694-701.
Meditation, Attention, and Neuroplasticity
Meditation, Attention, and Neuroplasticity
I mplications for the integrative care of attention- deficit/ hyperactivity disorder
Attention-deficit/hyperactivity disorder (ADHD) is the most common psychiatric disorder of childhood, affecting 3% to 10% of children (APA 1994, Sinha 2005, Szymanski 2001, Zylowska 2009) and may persist into adulthood (APA 1994). As defined by the DSM-IV-TR (APA 2000), essential features of ADHD include persistent inattention, hyperactivity and/or impulsivity; onset prior to age seven years; and the presence of symptoms in at least two settings, such as at school and home. Individuals with ADHD may experience difficulties in withholding attention from distractions, decision-making, regulating emotions, completing tasks or coping with a change in the requirements of a task (Baijal 2008, Sinha 2005). They are at increased risk of developing significant adverse social, academic and emotional consequences (APA 1994, Kuhlmann 2010, Sinha 2005, Szymanski 2001). While pharmacotherapy is the cornerstone of conventional ADHD care (Kuhlmann 2010, Szymanski 2001), it effectively reduces symptoms in only 70% of patients (Sinha 2005, Szymanksi 2001), may provide no functional improvement, and may cause undesirable side effects (Jensen 2004). A multimodal treatment approach including a behavioural component is generally recommended to optimize treatment success (Baijal 2008, Kuhlmann 2010, Sinha 2005). However, behaviour is rarely completely normalized even when pharmacotherapy and behavioural therapies are used in combination (Jensen 2004). Increasingly, families of children with ADHD are seeking suitable complementary and alternative care options (Chan 2003, Sinha 2005). Meditation and yoga are modalities of complementary medicine (Wahbeh 2008) for which preliminary clinical and neurobiological evidence suggests a potential therapeutic benefit in the management of pediatric and adult ADHD.
Meditation
Meditation is the ancient cognitive-spiritual practice of “stilling the fluctuations of the mind” (Baerentsen 2010). It consists of attentional self-regulatory techniques that are intended to cultivate physiological and psychological well-being (Lutz 2008, Raffone 2010, Slatger 2007, Wahbeh 2008). The various methods of meditation aim to attain deliberate, attentional self-regulation: a stable, attentive form of consciousness that differs from both ordinary wake- and sleep-states (Baerentsen 2010, Manna 2010). Two classifications of meditation are generally accepted: “focused attention” (FA) meditation and “open monitoring” (OM) meditation (Baerentsen 2010, Manna 2010). FA or “concentrative meditation” involves monitoring the focus of attention, detecting distraction, disengaging from the distraction and redirecting attention to the intended object (Manna 2010). These functions are linked with brain systems involved in conflict monitoring, selective and sustained attention (Manna 2010). OM or “mindfulness meditation” cultivates non-evaluative, non-reactive awareness of internal and external stimuli and appears to be related to brain regions involved in vigilance (Manna 2010).
Neuroplastic Effects of Meditation on the Human Brain Numerous neuroimaging studies using methods such as EEG (electroencephalography), PET (positron emission tomography), fMRI (functional magnetic resonance imaging), SPECT (single-photon emission computed tomography) and DTI (diffusion tensor imaging) have been published reporting the functional and anatomical changes that occur in the brain, including attention-related structures, with meditation practice (Baerentsen 2010, Baigal 2008, Cheisa 2010, Luders 2011, Lutz 2009, Vestergaard-Poulsen 2009). During meditation, attentional and sensory processing regions of the brain become activated, and these regions have been found to be generally more active in experienced meditators than in non-meditators (Baerentsen 2010, Chiesa 2010). Key studies identifying structural and functional changes in the brains of meditators are summarized in Table 1.
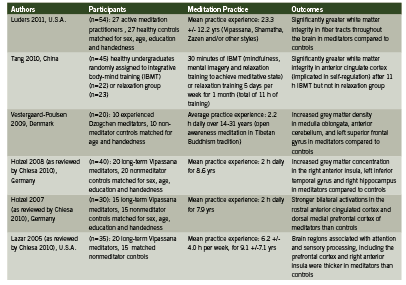
Although many brain regions and networks appear to respond neuroplastically to various methods of meditation, consistent patterns of regional brain activation and their clinical meaningfulness in the management of ADHD remain to be clearly elucidated. Preliminary research supports a potential therapeutic role in improving cognitive deficits and improving self-regulation in children and adults with ADHD. Two unpublished pilot studies (n = 23 and 24) reported behavioural improvement in ADHD children 12 years of age and younger who received individual meditation training (Moretti-Altuna 1987, Zylowska 2008, citing: Kratter 1983). In the Kratter study (1983), a graduate thesis project, male students (mean age 7-12 yrs) who met DSM-III criteria for ADHD were randomly assigned to either receive twice-weekly sessions increasing from two to eight minutes of meditation training (MT) per session for four wks; or progressive muscle relaxation training (RT) at the same frequency as MT; or were placed on a waiting list for four weeks (control group), after which they received meditation training identical to the experimental group. Both MT and RT groups showed significant improvement in impulsivity when faced with a problem-solving task, as measured by the Matching Familiar Figures Test (MFFT). Only the meditation groups showed improved selective attention ability, as assessed by the Fruit Distraction Test (FDT). Parental assessment scores also indicated significantly improved homework performance and at-home behaviour (Kratter 1983). These pilot studies were not peer-reviewed and were subject to the limitations of relatively small participant group sizes (Kratter 1983).
More recently, Zylowska and colleagues (2008) conducted an open-label study examining the feasibility of mindfulness meditation training for individuals with ADHD (n=8 adolescents and n=24 adults). Following the eight-week meditation program participants showed symptomatic and attentional process improvement (Zylowska 2008). As cited in Zylowska (2008), the meditation training was based on the work of Kabat-Zinn (1990) and Segal et al. (2002) with adaptations to address the unique challenges of ADHD. For example, sitting periods were shortened; walking meditation could be substituted for sitting meditation; and each session was concluded with a loving-kindness meditation to help address the low self-esteem issues often associated with ADHD. Participants attended six to eight weekly group sessions of 2.5 hours each, including meditation exercises and a psychoeducational component that reframed the “deficit” aspects of ADHD as “neurobiological differences”. The adolescents also practiced at home for an average of 42.6 minutes over four days per week. Assessment of neurocognitive performance and self-reported symptom scales indicated a statistically significant symptom reduction for adolescents and adults (pooled data), with 30% of participants reporting a symtomatic reduction of at least 30%. Neurocognitive findings suggested that meditation improved attention, inhibition and self-regulation, which are key deficits in ADHD (Zylowska 2008). Among the adolescent participants of this study, 75% reported a lifetime history of at least one psychiatric comorbidity, most commonly depression (25.1%). Meditation was associated with significant improvements in depression (p<0.01) and anxiety (p<0.02) scores among adults, but not among adolescents. However, the small adolescent sample size precluded meaningful statistical analysis (Zylowska 2008). This pilot study supported the feasibility of an ADHD-adapted mindfulness meditation program to reduce self-reported symptoms and improve neurocognitive performance in adults and adolescents, with 78% treatment adherence, very high participant satisfaction and no adverse events.
Supplementary, unpublished data from the same study indicated significant improvements in perceived stress and mindfulness (p<0.01) (Zylowska 2009). Participants reported additional improvements in ADHD symptoms with no changes in anxiety, depression, stress or mindfulness at three-months post-intervention follow-up (Zylowska 2009). It was noted that the social support provided by the group setting and the psychoeducational component may have contributed to the overall positive results (Zylowska 2008). According to study recruitment data, more individuals who wished to participate were prevented from doing so due to scheduling restrictions, suggesting that ease of program access and schedule flexibility may be critical when offering this type of group training (Zylowska 2008).
No adverse events have been reported with the use of meditation in the treatment of ADHD in children or adults (Krisanaprakornkit 2010).
Yoga in the Treatment of ADHD
Yoga is a non-religious, traditional practice that incorporates meditation with training in breathing techniques, physical postures and relaxation (Jensen 2004). It has been used in the treatment of ADHD (Brown 2005, Jensen 2004). In a crossover study by Jensen and Kenny (2004), boys (8-13 years of age) who had been diagnosed with ADHD by a pediatrician participated in a 20-session yoga training (n=11) or cooperative games (n=8). All but two of the boys (who did not require medication) were symptomatically stabilized by medication during school hours. (Zolpidem)
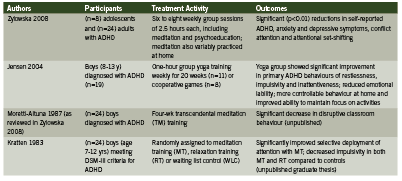
They were assessed by teachers at school, and by their parents during unmedicated periods (mornings and weekends) at home. Group yoga sessions occurred weekly for one hour and participants were also encouraged to practice at home. The yoga practice included training in concentration, breathing technique, static yoga postures and relaxation. The yoga group showed significant improvement in primary ADHD behaviours of restlessness, impulsivity and inattentiveness; and reduced mood swings, temper outbursts and crying fits as per the Conner’s Global Emotional Lability Index. Based on ADHD Index scoring, parents of children in the yoga group reported significantly more controllable behaviour and improved ability to keep focused on activities. Only the control group showed significant positive changes in anxiety, shyness and social problems, hyperactivity and perfectionism. Limitations of the study included significant differences between yoga and cooperative games groups despite randomization; inability to control the quality, duration and frequency of yoga practice at home; and the allocation of four times more contact time to the yoga group than controls, due to resource constraints (Jensen 2004).
Implications for Integrative Practice
Meditation and yoga have been used safely in conjunction with conventional care for ADHD in preliminary trials. Extensive neuroimaging data indicate meditation-induced neuroplasticity as a plausible explanation for the apparent attentional, emotional and self-regulatory benefits of meditation practice. Limitations to the currently available evidence for the use of meditation in ADHD treatment include relatively small study populations and lack of adequate statistical power (Foisy 2011, Zylowska 2008); reliance on self-reported data (Zylowska 2008); risk of bias (Krisanaprakornkit 2010); lack of standardized assessment standards (Foisy 2011); and uncertainty as to whether the observed neuroplastic effects of meditation in healthy individuals are clinically applicable to those with ADHD (Baijal 2008). Larger, controlled trials are needed to further characterize the therapeutic value of meditation and yoga in children and adults with ADHD, but these are relatively safe, inexpensive therapies that can be readily integrated into a multimodal treatment plan as supported by positive preliminary clinical and neurobiological evidence.
References
American Psychiatric Association (APA). Diagnostic and statistical manual of mental disorders (4th ed.). 1994. Washington, D.C.
American Psychiatric Association: Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Text Revision. Washington, DC, American Psychiatric Association, 2000. Accessed electronically at: http://www.cdc.gov/ncbddd/adhd/diagnosis.html
Baerentsen KB, Stodkilde-Jorgensen H, Sommerlund B, et al. An investigation of brain processes supporting meditation. Cogn Process 2010;11:57-84.
Baijal S, Gupta R. Meditation-based training: a possible intervention for attention deficit hyperactivity disorder. Psychiatry (Edgmont) 2008;5(4):48-55.
Brown KA, Patel DR. Complementary and alternative medicine in developmental disabilities. Ind J Pediatrics 2005:72:949-952.
Chan E, Rappaport LA, Kemper KJ. Complementary and alternative therapies in childhood attention and hyperactivity problems. Dev Behav Pediatr 2003;24(1):4-8.
Chiesa A, Serretti A. A systematic review of neurobiological and clinical features of mindfulness meditations. Psychological Medicine 2010;40:1239-1252.
Foisy, M. and Williams, K. The Cochrane review and non-pharmacological treatments for attention deficit hyperactivity disorder in children and adolescents: an overview of reviews. Evidence-Based Child Health: A Cochrane Review Journal, 2011;6:283–297.
Jensen PS, Kenny DT. The effects of yoga on the attention and behaviour of boys with attention-deficit/hyperactivity disorder (ADHD). J Atten Disord 2004;7(4):205-215.
Kratter J, Hogan JD. The use of meditation in the treatment of attention deficit disorder with hyperactivity. Dissertation Abstracts International 1983;44:[pages unknown]. Accessed electronically on 15 Aug 2011 at: http://www.livingwisdomschool.org/howtoapply/qs-pars-ask/research/Meditation%20in%20the%20Treatment%20of%20ADHD.pdf
Krisanaprakornkit T, Ngamjarus C, Witoonchart C, Piyavhatkul N. Meditation therapies for attention- deficit/hyperactivity disorder (ADHD). Cochrane Database of Systematic Reviews 2010, Issue 6. Art. No.: CD006507. DOI: 10.1002/14651858.CD006507.pub2.
Kuhlmann B, (ed.). Institute for Clinical Systems Improvement (ICSI). Diagnosis and management of attention deficit hyperactivity disorder in primary care for school-age children and adolescents. 2010 (Eighth Edition).
Luders E, Clark K, Narr KL, et al. Enhanced brain connectivity in long-term meditation practitioners. NeuroImage 2011;57:1308-1316.
Lutz A, Slagter HA, Dunne JD, et al. Attention regulation and monitoring in meditation. Trends Cogn Sci 2008;12(4):163-169.
Manna A, Raffone A, Perrucci MG, et al. Neural correlates of focused attention and cognitive monitoring in meditation. Brain Res Bull 2010;82:46-56.
Moretti-Altuna G. The effects of meditation versus medication in the treatment of attention deficit hyperactivity disorder. Dissertation Abstracts International 1987;47:4658. (As cited in Zylowska 2008).
Raffone A, Srinivasan N. The exploration of meditation in the neuroscience of attention and consciousness. Cogn Process 2010:11:1-7.
Sinha D, Efron D. Complementary and alternative medicine use in children with attention deficit hyperactivity disorder. J Paediatr Child Health 2005;41(23-26).
Slatger HA, Lutz A, Greischer LL, et al. Mental training affects distribution of limited brain resources. PLoS Biol 2007;5(6):1228-1235.
Szymanski M, Zolotor A. Attention-deficit/hyperactivity disorder: management. Am Fam Physician 2001;64:1355-1362.
Tang Y, Lu Q, Geng X, et al. Short-term meditation induces white matter changes in the anterior cingulate. Proc Nat Acad Sci 2010;107(35):15649-15652.
Vestergaard-Poulsen P, van Beek M, Skewes M, et al. Long-term meditation is associated with increased gray matter density in the brain stem. NeuroReport 2009;20:170-174.
Wahbeh H, Elsas S-M, Oken BS. Mind-body interventions: applications in neurology. Neurology 2008;70:2321-2328.
Zylowska L, Ackerman DL, Yang MH, et al. Mindfulness meditation training in adults and adolescents with ADHD: a feasibility study. J Atten Disord 2008;11(6):737-746.
Zylowska L, Smalley SL, Schwartz JM. Mindful awareness and ADHD. Clinical Handbook of Mindfulness 2009;43:319-338.













