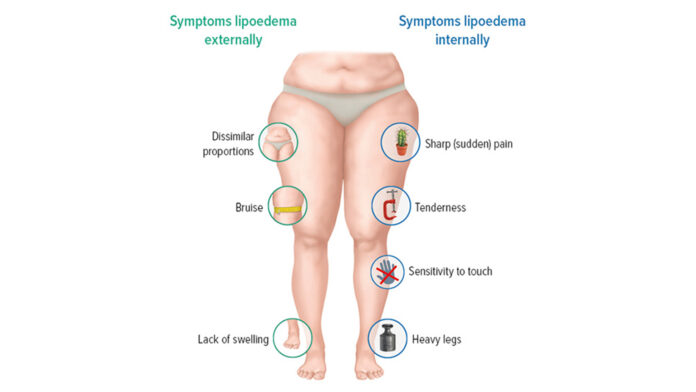
Lipedema is one of those conditions that can quietly shape a woman’s daily life—pain, swelling, heaviness, tenderness, easy bruising—yet still be mislabelled as “just weight” or confused with other fluid and fat-distribution disorders. For many women, that delay in recognition means years of frustration, body shame, and trial-and-error symptom management.
That’s why a newly published randomized controlled trial is turning heads: it suggests Pycnogenol® (a standardized French maritime pine bark extract) can meaningfully reduce lipedema-related symptoms and improve quality of life in as little as 60 days—without invasive procedures.
What the study tested
Researchers ran a double-blind, randomized, placebo-controlled trial involving 100 women aged 18–40 with a clinical diagnosis of lipedema. Participants took either Pycnogenol® 50 mg three times daily (every eight hours) or a placebo for 60 days, with assessments at baseline, 30 days, and 60 days.
The main measure: quality of life and symptom impact
The primary outcome was a validated lipedema-specific quality-of-life questionnaire called QuASiL. It captures how strongly lipedema symptoms affect day-to-day life—covering issues such as swelling, tenderness, bruising, heaviness, discomfort, and satisfaction with leg appearance.
Body composition was also tracked
Researchers also monitored body composition using bioimpedance, looking at changes such as body fat percentage.
Key findings at 60 days
The results were notable because the placebo group’s symptom burden worsened over time, while the Pycnogenol® group improved.
- Women taking Pycnogenol® saw about a 29% reduction in overall symptom burden by day 60 based on QuASiL scoring.
- The placebo group’s scores continued to trend worse across the same period.
- Improvements were reported across the symptom spectrum, with particularly meaningful gains in common complaints such as leg swelling, heavy legs, tenderness, and bruising.
- The Pycnogenol® group also showed improvements in body composition measures compared with placebo over the 60-day period.
- No adverse effects were reported in the study, supporting Pycnogenol®’s reputation as a generally well-tolerated option.
Why this matters for women’s health
Lipedema disproportionately affects women and often appears or worsens during hormonal shifts such as puberty, pregnancy, and menopause. It isn’t simply “extra weight,” and for many women it can feel like living in a body that won’t respond to the rules that work for everyone else—exercise more, eat less, try harder.
The bigger issue: there are still no approved medications and no universally accepted standard of care for lipedema. Many women rely on symptom-management strategies like compression garments, manual lymphatic drainage, physical therapy, movement routines that support lymphatic flow, and lifestyle changes that help manage inflammation and discomfort. These can be helpful, but they rarely feel like a complete solution.
In that context, a natural option showing measurable symptom improvement in a controlled trial is more than interesting—it’s potentially practice-informing.
Why Pycnogenol® is a plausible fit
Pycnogenol® is rich in plant compounds (including procyanidins and other polyphenols) and has been studied for vascular and microcirculation support. That matters because lipedema often involves chronic swelling, tissue tenderness, and a sense of heaviness that may be influenced by circulation dynamics and inflammatory signalling.
The proposed explanation is straightforward: a compound that supports microcirculation, vascular integrity, and inflammation balance could plausibly reduce the symptom “stack” that makes lipedema so physically and emotionally exhausting.
That said, the mechanism is not the same as proof. The strength of this study is clinical outcomes, not a definitive explanation of exactly how the changes occurred.
A clear-eyed look at the limitations
This trial is promising, but it’s not the final word.
- Short duration: 60 days is enough to show a signal, but not enough to confirm long-term durability or disease-modifying impact.
- Specific population: women aged 18–40, in one clinical setting. Results may not fully translate to older women or different demographics.
- Body composition nuances: changes in body fat measures are encouraging, but interpretation should be cautious, especially when baseline differences between groups can influence outcomes.
- Needs replication: the real test is whether future studies reproduce similar results across multiple sites with longer follow-up.
How to discuss it responsibly in natural health retail
If you’re in the natural products industry, this study opens a door—but it’s important to walk through it with care.
A responsible framing sounds like this:
“This clinical trial suggests Pycnogenol® may help reduce symptom burden and improve quality of life for women with lipedema. It’s not a cure, but it may be a supportive option worth discussing with a health professional.”
Practical guidance:
- Encourage customers to seek proper assessment, since lipedema is commonly misidentified.
- Treat Pycnogenol® as an add-on to foundational strategies (compression, movement, physiotherapy, lymphatic support), not a replacement.
- Advise consultation with a health care practitioner for anyone who is pregnant or breastfeeding, on anticoagulant or antiplatelet medication, managing complex health conditions, or preparing for surgery.
What comes next
For decades, lipedema has lived in the gap between “common” and “poorly served.” This trial is important because it treats women’s lived experience as measurable—and worth targeting with real clinical research.
If larger, longer studies confirm these outcomes, Pycnogenol® could earn a more central role as an evidence-led, non-invasive option for symptom support. For now, it stands as a credible early signal: not hype, not a miracle, but a meaningful step toward better tools for women who have been told, far too often, that their symptoms are “just weight.”